Abstract
Objectives
This case–control study explored the association of sirtuin 1 polymorphisms (rs10997868 and rs730821) with metabolic parameters, smoking duration, sirtuin 1 levels, and the oxidative stress biomarker 8-hydroxy-2′-deoxyguanosine in healthy smokers in Saudi Arabia.
Methods
Genomic DNA was isolated from peripheral blood samples, and rs10997868 and rs730821 polymorphism genotyping was performed for 43 healthy smokers and 33 healthy controls using real-time polymerase chain reaction. Plasma sirtuin 1 and 8-hydroxy-2′-deoxyguanosine levels were measured using enzyme-linked immunosorbent assay.
Results
Sirtuin 1 levels were significantly higher in healthy smokers than in controls, while 8-hydroxy-2′-deoxyguanosine levels were not significantly lower in healthy smokers. There was no significant association of sirtuin 1 protein expression and 8-hydroxy-2′-deoxyguanosine levels with rs10997868 and rs730821. Our findings showed a significant association between smoking duration and rs10997868 single nucleotide polymorphism, which was highest in those with the C/C genotype and lowest in those with the A/C genotype (p = 0.035). In addition, the serum cholesterol level was highest in the C/C group and lowest in the A/C group (p = 0.029). None of the studied factors (smoking duration, serum cholesterol, fasting blood sugar, or serum triglycerides) was significantly associated with sirtuin 1 rs730821.
Conclusions
Our study showed that sirtuin 1 upregulation and polymorphisms are associated with smoking duration and lipid levels in healthy smokers.
Introduction
Cigarette smoking is harmful and the leading cause of death due to cardiovascular disease. 1 Owing to the widely known health risks associated with smoking and secondhand smoke, the World Health Organization has collaborated with different countries to implement policies and programs that motivate and assist people to stop smoking. This global action plan aims to reduce the prevalence of smoking worldwide by 30% by 2025. 2 Tobacco use and its associated risks to both users and secondhand smokers are serious public health issues in Saudi Arabia. The number of smokers and smoking-related mortality in Saudi Arabia are gradually increasing, with over 70,000 deaths due to smoking-related illnesses being reported each year. 3 Cigarette smoke promotes the production of reactive oxygen species and oxidative stress by activating nicotinamide adenine dinucleotide phosphate hydrogen oxidase in rat vascular smooth muscle cells. 4 It affects antioxidant systems by compromising the balance between antioxidant consumption and production.5,6 In addition to exaggerating oxidative stress, smoking is considered to contribute significantly to DNA damage; mutations in the genes associated with carcinogenesis; and cardiovascular, pulmonary, and neurological diseases.7,8
Among proteins of the sirtuin family, sirtuin 1 (SIRT1) has been the most extensively studied. 9 SIRT1 localizes to the nucleus and cytoplasm, participating in epigenetic regulation via histone deacetylation. 10 Furthermore, SIRT1 is involved in apoptosis, inflammation, aging, oxidative stress, glucose metabolism, glycolysis, gluconeogenesis, fat metabolism, and insulin secretion.11–15 Few studies exploring SIRT1 single nucleotide polymorphisms (SNPs) have revealed their associations with inflammation, obesity, type 2 diabetes, blood pressure, dyslipidemia, cancer, and coronary artery disease.16,17 SIRT1 upregulation exerts anti-aging effects. 15 However, SIRT1 upregulation may enhance addiction predisposition, anxiety, depression, and hyperactivity.18–20 Notably, SIRT1 polymorphisms have also been associated with depression. 21
Oxidative DNA damage results from the production of 8-hydroxy-2′-deoxyguanosine (8-OHdG), a reliable biomarker of DNA damage. 22 Although oxidative stress has been associated with cigarette smoking, 23 its association with antioxidant levels and oxidative DNA damage markers remains inconsistent. To date, only a few studies have investigated 8-OHdG levels in the serum or plasma of healthy smokers. Furthermore, reports on 8-OHdG levels in healthy smokers have been inconsistent. A study indicated that cigarette smokers exhibited lower average 8-OHdG expression than nonsmokers, likely due to the presence of effective repair mechanisms for oxidative damage in smokers. 24 In contrast, several studies have reported increased levels of 8-OHdG in smokers.24,25 Joshi et al. reported increased serum 8-OHdG and MDA levels along with the downregulation of antioxidant enzymes, including superoxide dismutase, catalase, and glutathione peroxidase, in long-term healthy cigarette smokers without systemic diseases compared with those in nonsmokers. 5 Chen et al. showed that in vivo exposure of rats to cigarette smoke extract twice daily for 45 days increased 8-OHdG expression. 26
To date, no studies have investigated the association of SIRT1 polymorphisms with SIRT1 protein expression and 8-OHdG levels in healthy smokers. Most studies have focused on SIRT1 levels in different types of cancer. This case–control study aimed to examine the association of SIRT1 polymorphisms (rs10997868 and rs730821) with SIRT1 and 8-OHdG levels as well as smoking duration among healthy smokers in Saudi Arabia.
Materials and methods
Sample collection
Healthy cigarette smokers in Saudi Arabia were recruited from a smoking clinic at the Ibn Sena Hospital in Makkah City, Saudi Arabia, between November 2023 and April 2024. The study population included 43 healthy smokers and 33 healthy participants aged 20–60 years. Smokers who smoked water pipes or electronic cigarettes, had been smoking for <1 year, did not smoke daily, or were undergoing smoking cessation treatment while taking cessation medication were excluded. Individuals with chronic diseases (such as hypertension and diabetes mellitus) and heart disease as well as those aged <20 or >60 years were excluded. Individuals exposed to secondhand smoke were also excluded. The control group comprised randomly selected individuals from other outpatient clinics who met the following criteria: healthy Saudi Arabian individuals who provided consent to participate in the study and had no chronic illnesses, similar to the case group. Ethical approval for this study was obtained from the Research Ethics Committee (Institutional Review Board Committee) of the Directorate of Health Affairs, Makkah Region (No.: H-02-K-076-1023-1007, October 30, 2023). All participants provided written informed consent. This study was conducted in accordance with the Declaration of Helsinki 1975, as revised in 2024.
This study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 27 No identifiable patient information was utilized in the study, and all participant details were thoroughly de-identified prior to the analyses.
Biochemical measurements
Peripheral blood samples (2 mL) extracted from the participants and collected in ethylenediaminetetraacetic acid–containing tubes were utilized for genotyping and enzyme-linked immunosorbent assay (ELISA). In addition, 2-mL samples of fasting venous blood were collected in lithium heparin–containing tubes (green tops) and analyzed for cholesterol, triglyceride, and fasting blood glucose levels using an automated Dimension® EXL200 Clinical Chemistry System (Siemens, Munich, Germany).
SIRT1 (SEKH-0540) and 8-OHdG (SEKSM-0038) levels were determined using ELISA kits (Human Immunoassay; Solarbio, China) as per the manufacturer’s instructions. Briefly, the samples (100 μL) and standards were placed onto a 96-well plate and incubated for 90 min at 37°C. Subsequently, a detection antibody (100 µL) was introduced, and the samples were incubated for 1 h at 37°C, followed by four washing steps. An avidin–horseradish peroxidase conjugate (100 μL) was then added, and the samples were incubated for 30 min at 37°C in the dark, after which it was washed six times. Next, 90 μL of the substrate reagent was added, and the samples were incubated for 15 min at 37°C in the dark. Finally, 50 μL of stop solution was added, and the optical density of the samples at 450 nm was measured immediately using a microplate spectrophotometer as per the manufacturer’s instructions.
DNA extraction and genotyping
Genomic DNA was extracted from whole blood samples using a HiGene™ genomic DNA preparation kit (Biofact, Daejeon, Republic of Korea) following the manufacturer’s protocol. The quality and integrity of the DNA were determined using a NanoDrop spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). Isolated DNA samples with 260/280 ratios between 1.8 and 2.1 were used.
SNP genotyping of SIRT1 (rs10997868 and rs730821) was performed via real-time polymerase chain reaction using TaqMan allelic discrimination assays (Thermo Fisher Scientific, USA). The total volume of each well of a 96-well plate was 10 μL, and each well contained 1 μL of DNA, 5 μL of TaqMan Genotyping Master Mix (2×), 3.75 μL of distilled water, and 0.25 μL of TaqMan SNP genotype assay. Once inside the thermal cycler, enzyme activation was initiated at 95°C for 10 min, followed by 40 cycles of denaturation at 95°C for 15 s, annealing at 60°C for 1 min, and extension at 60°C for 1 min as per the manufacturer’s instructions.
Statistical analyses
Data entry and statistical analyses were performed using the Statistical Package for Social Sciences (SPSS, version 28.0; SPSS Inc., Chicago, IL, USA). Normality was tested using the Shapiro–Wilk test. Data were described using frequency and percentages for qualitative variables and using arithmetic mean and SD values for continuous numerical variables. One-way analysis of variance and Kruskal–Wallis tests were used to compare normally and non-normally distributed continuous variables between more than two groups, respectively. The chi-square test, crude odds ratio (OR), and 95% confidence interval (CI) were used to compare qualitative variables. Illustrative charts and graphs of the diagnostic workups and genetic testing are presented (Tables 1 to 4, Figure 1). Statistical significance was set at p < 0.05.
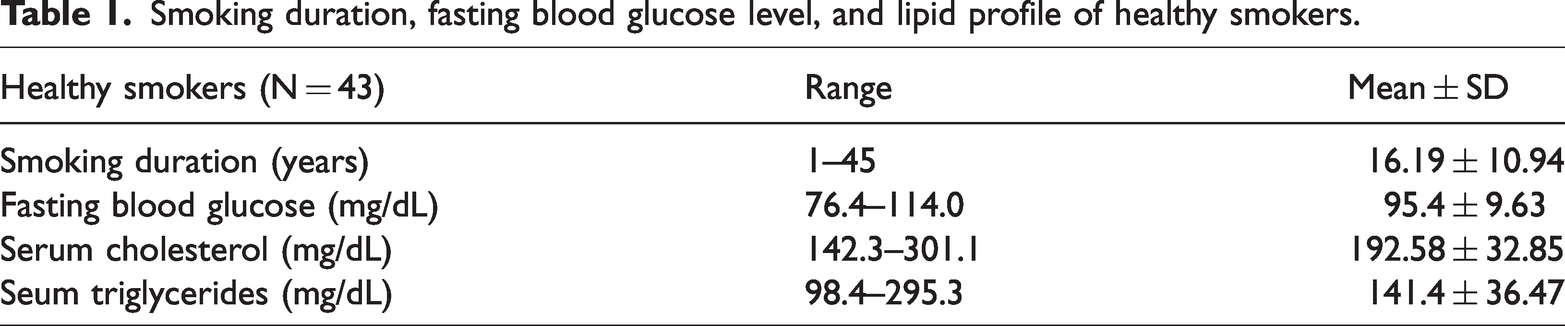
Smoking duration, fasting blood glucose level, and lipid profile of healthy smokers.
Comparison of SIRT1 genotype and allele frequencies between healthy smokers and controls.

chi-square test.
SIRT1: sirtuin 1; OR: odds ratio; CI: confidence interval.
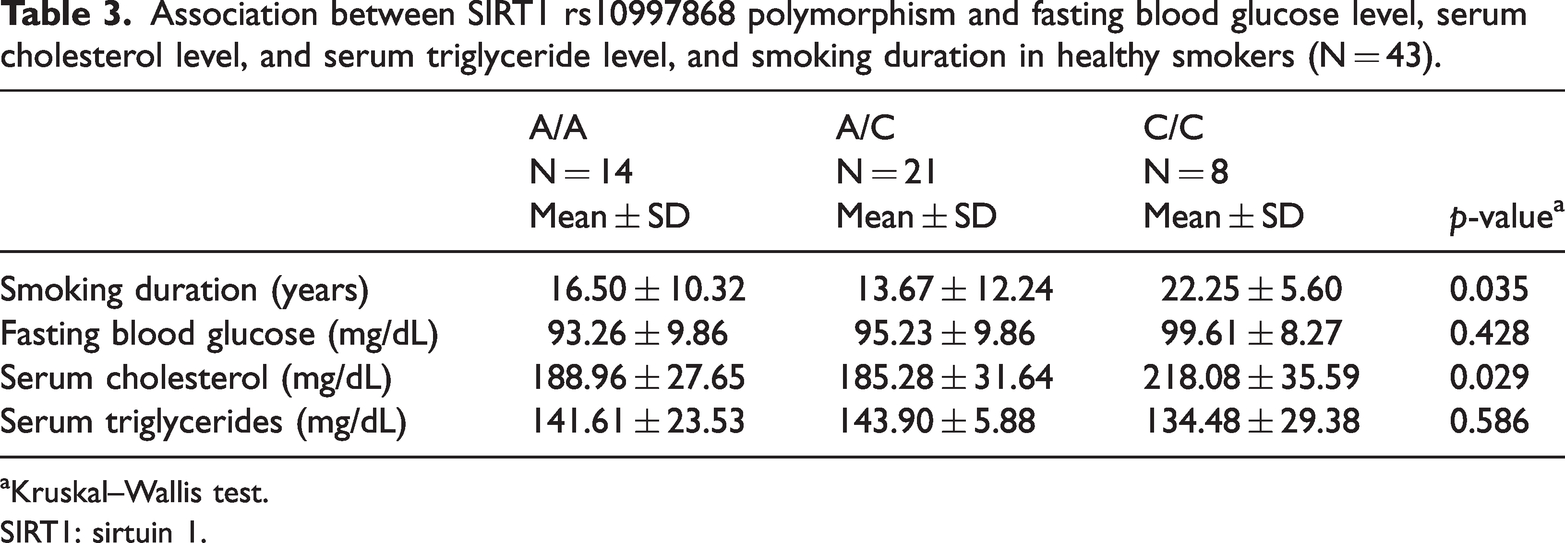
Association between SIRT1 rs10997868 polymorphism and fasting blood glucose level, serum cholesterol level, and serum triglyceride level, and smoking duration in healthy smokers (N = 43).
Kruskal–Wallis test.
SIRT1: sirtuin 1.
Comparison between healthy smokers and controls regarding SIRT1 and 8-OHdG protein expression levels.

Independent t-test.
SIRT1: sirtuin 1; 8-OHdG: 8-hydroxy-2′-deoxyguanosine.
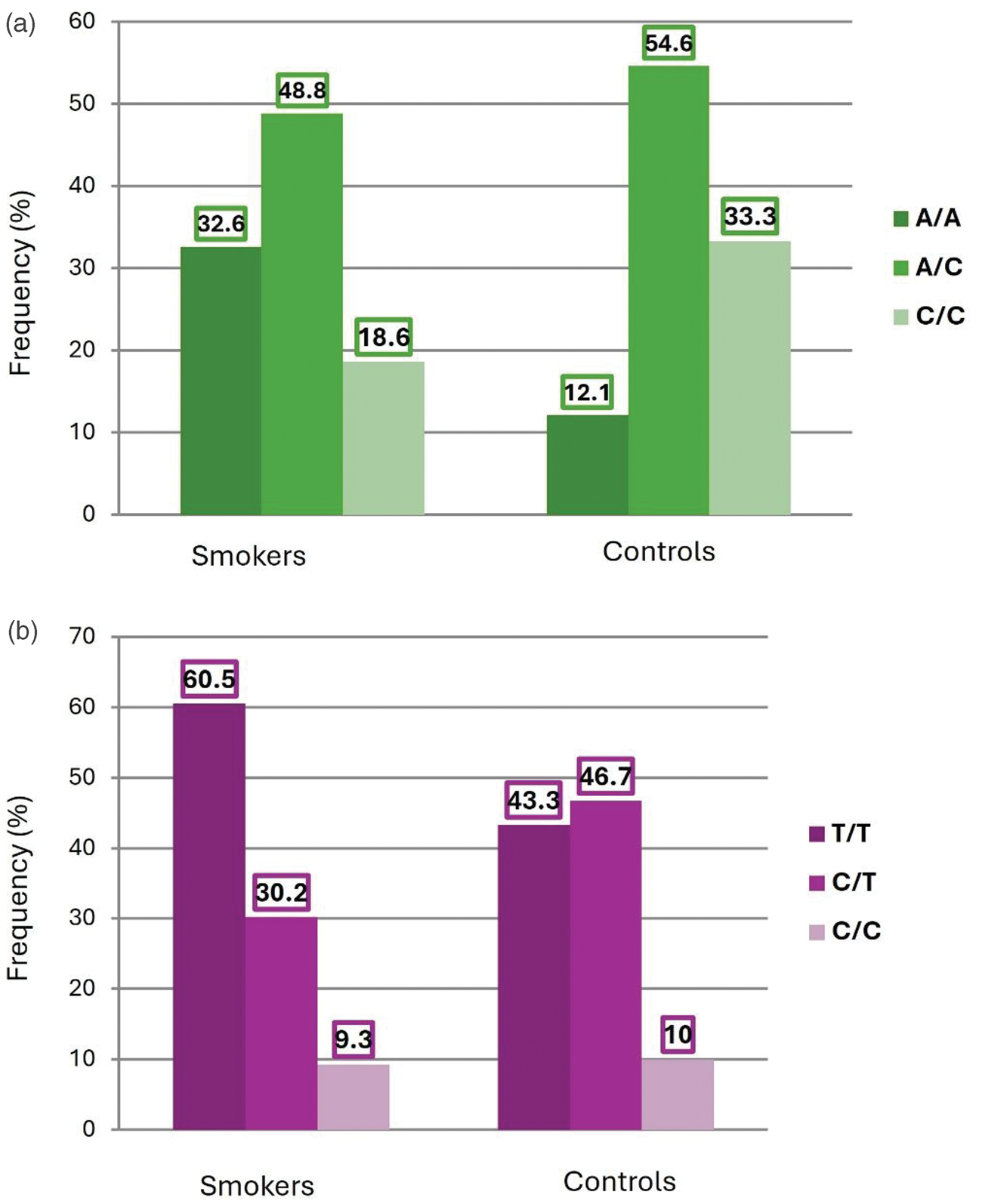
Genotype frequencies of SIRT1 SNPs rs10997868 (a) and rs730821 (b) in controls and healthy smokers. Frequencies (percentage of individuals) are shown for each genotype in both groups. SIRT1: sirtuin 1.
Results
In total, 43 healthy smokers and 33 controls were included in this study. The mean ages (± SD) of the healthy smokers and controls were 38.42 ± 9.15 and 31.8 ± 5.90 years, respectively (p < 0.001). Among healthy smokers, the smoking duration ranged from <5 to >5 (mean ± SD: 16.19 ± 10.94) years. The fasting blood glucose level was 76.4–114 (95.4 ± 9.63) mg/dL, serum cholesterol level was 142.3–301.1 (192.58 ± 32.85) mg/dL, and serum triglyceride level was 98.4–295.3 (141.4 ± 36.47) mg/dL) Table 1.
Comparison of SIRT1 genotype and allele frequencies between healthy smokers and controls
As shown in Table 2 and Figure 1(a), 32.6% of the healthy smokers had a SIRT1 rs10997868 A/A genotype compared with 12.1% of the controls. In addition, 33.3% of the controls had a C/C genotype compared with 18.6% of the healthy smokers. However, this difference was not statistically significant (p = 0.081). Considering genotype A/A as a reference, the C/C genotype was less common among healthy smokers (OR = 0.21; 95% CI = 0.05–0.87; p = 0.321).
As shown in Table 2 and Figure 1(b), there were no statistically significant differences between healthy smokers and controls with respect to SIRT1 rs730821 frequency.
Factors associated with SIRT1 rs10997868
As shown in Table 3, there was a significant association between smoking duration and SIRT1 rs10997868 SNP, which was highest (22.25 ± 5.60 years) in those with the C/C genotype and lowest (13.67 ± 12.24 years) in those with the A/C genotype (p = 0.035). The serum cholesterol level was highest (218.08 ± 35.59 mg/dL) in those with the C/C genotype and lowest (185.28 ± 31.64 mg/dL) in those with the A/C genotype (p = 0.029). The levels of fasting blood glucose and triglycerides were not significantly associated with SIRT1 rs10997868.
Factors associated with SIRT1 rs730821
None of the studied factors (smoking duration, serum cholesterol, fasting blood sugar, or serum triglycerides) were significantly associated with SIRT1 rs730821 (Supplementary Table S1).
SIRT1 and the oxidative stress marker 8-OHdG
SIRT1 levels were significantly higher in healthy smokers than in controls (12.16 ± 2.79 vs. 5.79 ± 2.41 pg/mL, p < 0.001). The levels of the oxidative stress marker 8-OHdG were lower in healthy smokers than in controls (39.08 ± 11.50 vs. 33.83 ± 8.77 pg/mL). However, the difference was not statistically significant (Table 4).
The levels of SIRT1 or 8-OHdG were not associated with rs10997868 (Supplementary Table S2) or rs730821 (Supplementary Table S3).
Discussion
Cigarette smoking triggers oxidative stress by impairing the balance between antioxidant consumption and production, contributing to DNA damage and disease-associated gene polymorphisms.5,6 Among the sirtuins, SIRT1 has been extensively studied because of its involvement in many important pathways that regulate oxidative stress and enhance antioxidant defenses. 28 Population-based cohort studies have identified significant links between prevalent SNPs in SIRT1 and type 2 diabetes, 29 obesity,17,30 and atherosclerosis, 31 all of which are characterized by increased oxidative stress.32,33 In our study, SIRT1 rs10997868 showed a significantly stronger association with smoking duration and cholesterol levels in healthy smokers than in nonsmokers. However, no such association was found for SIRT1 rs730821. SIRT1 protein expression was higher in healthy smokers than in controls. The oxidative DNA damage biomarker 8-OHdG was not significantly upregulated in healthy smokers compared with that in controls. These findings reflect the alterations induced by cigarette smoking, which are implicated in disease pathogenesis.
Only a few human genetic association studies on SIRT1 have been reported to date. SIRT1 rs10997868 is associated with lung function decline, 30 diabetes, and diabetic nephropathy. 34 However, no studies have assessed SIRT1 rs10997868 in healthy smokers. Therefore, this is the first study to report that rs10997868 is more strongly associated with smoking duration and cholesterol levels in healthy smokers than in controls, while no such associations with these factors or other clinical parameters were found for SIRT1 rs730821. Notably, cholesterol levels and smoking duration were significantly higher in healthy smokers with the CC genotype for SIRT1 rs10997868, indicating that they are at high risk of hypercholesterolemia. Our results are consistent with those of a previous study using different models, which showed that other SIRT1 SNPs, such as rs7069102 and rs2273773, were associated with abnormal cholesterol metabolism in Japanese hemodialysis patients. 35 SIRT1 rs730821 and rs10997868 are reportedly associated with increases in body mass index (BMI) and weight, 30 suggesting that differences in SIRT1 expression influence the impact of abdominal adipose tissue on lung function decline and might regulate systemic low-grade inflammation resulting from increased abdominal fat tissue. 30 Another study reported that SIRT1 polymorphisms are associated with BMI and obesity.17,36 Thus, we can infer from these results that genetic variations in SIRT1 may increase the risk of hypercholesterolemia and obesity in healthy smokers. In addition, previous findings have indicated that cigarette smoking is associated with increased dyslipidemia prevalence, BMI, central obesity, von Willebrand factor functional activity, and high-sensitivity cardiac troponin I level. 37 Collectively, these results suggest cardiovascular disease vulnerability in healthy smokers.
Recent international studies have increasingly highlighted the role of SIRT1 gene polymorphisms and expression in modulating metabolic and oxidative stress responses, particularly in populations exposed to cigarette smoke. Shimoyama et al. reported that certain SIRT1 polymorphisms, including rs7895833, are associated with metabolic syndrome components such as dyslipidemia in a Japanese cohort. This suggests that SIRT1 variations influence lipid metabolism and susceptibility to metabolic disturbances. 38 Similarly, in a Chinese population, Zhang et al. found elevated SIRT1 expression levels in healthy smokers, which was hypothesized to represent an adaptive mechanism against smoking-related oxidative stress, with some SIRT1 SNPs correlating with metabolic biomarkers. 39 These observations support our findings that SIRT1 upregulation and specific genotypes are linked to smoking duration and lipid parameters. Our data contribute to this growing body of evidence by indicating population-specific genetic associations, emphasizing the crucial influence of genetic background on SIRT1 variants. Thus, our results support the hypothesis that SIRT1 genetic variations and expression levels are significant contributors to metabolic alterations observed in healthy smokers. Further studies with larger, multi-ethnic cohorts are warranted to validate these associations and clarify the potential of SIRT1 as a biomarker or therapeutic target in smoking-related metabolic disorders.
SIRT1 acts as an antioxidant, making cells resistant to oxidative stress. 40 The effect of cigarette smoke on SIRT1 expression has been studied in vitro using alveolar epithelial type 2 cells exposed to cigarette smoke extract. These cells exhibit SIRT1 downregulation and greater mitochondrial oxidative stress-related DNA damage. 41 In the current study, serum SIRT1 protein levels were higher in healthy smokers than in controls. Similarly, Beane et al. found that compared with nonsmokers, SIRT1 activity was significantly elevated in cytologically normal bronchial airway epithelial cells from active smokers. However, its levels were downregulated in those with non–small cell lung cancer. 42 The authors suggested that this upregulation of SIRT1 activity acted as a protective shield against oxidative stress and DNA damage caused by smoking, proposing a tumor-suppressor role of SIRT1. 43 In contrast, a study showed decreased levels of SIRT1 in the peripheral blood mononuclear cells of healthy smokers compared with those in nonsmokers. 44 This discrepancy could be attributed to variations in patient samples and techniques.
Moreover, a recent study showed that increased SIRT1 expression is responsible for addiction, anxiety, and depression.18–20 Consistently, nicotine and lipopolysaccharide stimulation were shown to increase the expression of SIRT1 mRNA and protein in a time- and concentration-dependent manner in human gingival fibroblasts. 45 In the context of cigarette smoking, we can infer from these studies that the observed upregulation of SIRT1 might induce depression, anxiety, and addiction to cigarette smoking among healthy smokers, thereby aggravating this harmful habit. However, this needs further investigation.
8-OHdG, a widely recognized biomarker of oxidative DNA damage, can be quantitatively measured using DNA hydrolysis. 22 Although previous studies have generally reported elevated 8-OHdG levels in leukocytes from chronic smokers, 46 our preliminary findings—characterized as results of an exploratory pilot—indicate lower circulating 8-OHdG levels in healthy smokers than in controls. However, the difference was not statistically significant. Given the heterogeneity and inconsistency of results across the existing literature—likely attributable to variations in sample types, populations, and analytical methods—these results should be interpreted with caution. Notably, some studies, including the current study, have indicated a lower mean expression of 8-OHdG in cigarette smokers, which could be attributed to enhanced repair mechanisms for oxidative damage in smokers. 47 These findings underscore the need for further research with larger sample sizes and standardized methodologies to elucidate the true relationship between tobacco exposure and oxidative DNA damage.
Limitations
This study has several limitations that should be considered when interpreting the findings. First, the relatively small sample size, primarily due to time and budget constraints, directly impacts the reproducibility and generalizability of our results. Although we observed statistically significant correlations between SIRT1 genetic variants, oxidative stress biomarkers, and clinical parameters, these findings require validation in larger cohorts. Second, a significant age difference existed between the healthy smoker and control groups; although previous research suggests that age does not directly affect 8-OHdG levels under normal conditions, 48 the potential confounding effect of age-related differences on SIRT1 expression cannot be entirely excluded, especially given the known downregulation of SIRT1 during senescence via autophagy.49,50 However, the fact that we found increased SIRT1 expression in healthy smokers compared with that in nonsmokers suggests that the observed effects on oxidative stress markers are primarily attributable to cigarette smoking. The lack of previous studies reporting SIRT1 levels in healthy smokers underscores the novelty of our work, positioning it as a pilot study for future research. We recommend that future investigations employ larger, age-matched, multi-ethnic cohorts to assess these biomarkers across diverse populations and determine their predictive value for the development and progression of smoking-related diseases.
Conclusion
Our findings showed that SIRT1 rs10997868 influences smoking duration and cholesterol metabolism in healthy smokers, while SIRT1 rs730821 is not associated with these parameters. Additionally, the present study indicates that the SIRT1 expression levels are higher in healthy smokers than in controls. These findings provide strong evidence regarding the involvement of SIRT1 polymorphisms in the pathophysiology of diseases caused by tobacco exposure. Therefore, in-depth investigations are necessary to elucidate the precise roles of SIRT1 polymorphisms and expression in healthy smokers.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251387499 - Supplemental material for Association of sirtuin 1 rs10997868 and rs730821 polymorphisms with sirtuin 1 and hydroxy-2′-deoxyguanosine levels in healthy smokers: A case–control study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251387499 for Association of sirtuin 1 rs10997868 and rs730821 polymorphisms with sirtuin 1 and hydroxy-2′-deoxyguanosine levels in healthy smokers: A case–control study by Samar Sultan in Journal of International Medical Research
Footnotes
Acknowledgements
We would to thank Princes Al-Jawhara Center of Excellence in Research of Hereditary Disorders, King Abdulaziz University, Jeddah, Saudi Arabia.
Author contributions
Conceptualization: Samar Sultan
Investigation: Samar Sultan
Methodology: Samar Sultan
Supervision: Samar Sultan
Writing–original draft: Samar Sultan
Writing–review & editing: Samar Sultan
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author declares no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
