Abstract
Objective
To investigate the relationship between smoking exposure and cervical cancer risk by integrating observational and genetic evidence.
Methods
We analyzed data from the National Health and Nutrition Examination Survey (1999–2018) and performed a genetic instrumental variable analysis using genome-wide association studies data. Smoking exposure was assessed using self-reported status and serum cotinine concentrations. Logistic regression models were used to evaluate observational associations, while inverse variance weighting was used for genetic analysis.
Results
Observational analysis showed that current smokers had a significantly higher cervical cancer risk than nonsmokers and former smokers (adjusted odds ratio = 3.05, 95% confidence interval: 1.61–5.78, p < 0.001). Higher serum cotinine concentrations were also positively associated with cervical cancer risk. Genetic analysis further supported a causal link between smoking exposure and cervical cancer.
Conclusions
Smoking exposure significantly increases cervical cancer risk, emphasizing the importance of smoking cessation for prevention of cervical cancer. Incorporating tobacco control into cervical cancer prevention strategies could reduce disease burden.
Keywords
Introduction
Cervical cancer is one of the leading causes of cancer-related deaths among women globally, ranking fourth in prevalence and mortality.1,2 Each year, over 120,000 women are newly diagnosed with cervical cancer, and approximately 77,000 succumb to this disease. 3 It is most commonly caused by persistent infection with high-risk human papillomavirus (HPV), particularly types 16 and 18. Other risk factors that reportedly contribute to cervical cancer development include early sexual activity, multiple sexual partners, and smoking.2,4 The introduction of screening programs and HPV vaccination has reportedly reduced the prevalence and mortality rates in many developed countries.5,6 However, significant disparities remain, especially in low- and middle-income nations, which have limited access to prevention and treatment services. Smoking is one of the major modifiable risk factors for cervical cancer as it can exacerbate the persistence of HPV infection and compromise the strength of the immune system.6,7 In 2019, over 30,000 cervical cancer–related deaths were attributed to smoking, underlining the substantial impact of this lifestyle factor. 8 Smoking is widely acknowledged as a significant cause of various health conditions, including cancer, cardiovascular diseases, and respiratory illnesses.9,10 It is known to introduce harmful substances such as reactive oxygen species (ROS) into the body, which induce oxidative stress, inflammation, and cellular damage.11,12 This cascade of events significantly increases the risk of developing cancers at various sites, including the lungs, pancreas, and cervix.9,13 Smoking also plays a role in the alteration of DNA methylation patterns, further contributing to carcinogenesis. 13 Despite a global decline in smoking prevalence, tobacco use continues to present a significant public health burden. Recent studies have demonstrated that smoking not only elevates the risk of cancers but also impairs the immune response, thereby facilitating the progression of diseases such as cervical cancer.11,14,15 In addition to its cancer-related effects, smoking has been linked to other chronic conditions, including type 2 diabetes 16 and age-related macular degeneration, 17 as revealed by Mendelian randomization (MR) studies. This complex relationship underscores the need for continued research on the multifaceted role of smoking in disease development. Cotinine, a primary metabolite of nicotine, acts as a biomarker for tobacco exposure, with its levels in the body closely reflecting tobacco consumption.18,19
Clinical research on the link between smoke exposure and cervical cancer risk is limited; therefore, this study aimed to examine how smoking status and serum cotinine concentrations are related to cervical cancer development using data from the National Health and Nutrition Examination Survey (NHANES). MR, an advanced analytical method that utilizes single nucleotide polymorphisms (SNPs) from genome-wide association studies (GWAS) as instrumental variables (IVs), provides a means to assess causality between exposure (e.g. serum cotinine) and outcome (e.g. cervical cancer). Unlike traditional observational studies, MR is less prone to confounding or reverse causation. By combining the NHANES data with MR analysis, our study offers a more robust approach to explore the relationship between smoke exposure and cervical cancer risk, strengthening the reliability of our findings.
Methods
Study design and population in NHANES
The NHANES database is a health and nutrition survey dataset for the US population, managed by the Centers for Disease Control and Prevention (CDC). 20 Data are released every 2 years and include demographic information, methodology details, examination and dietary data, questionnaire responses, and laboratory results. Further details can be found on the website: https://www.cdc.gov/nchs/nhanes. The NHANES study protocol was approved by the Ethics Review Board of the National Center for Health Statistics (NCHS).
In this study, we identified 8339 women from the NHANES dataset who had voluntarily participated in the survey between 1999 and 2018. We examined the relationship between tobacco smoke exposure and cervical cancer risk, focusing on smoking status and serum cotinine concentrations. The exclusion criteria were as follows: (a) lack of pregnancy-related data (n = 1753); (b) age <20 years (n = 46,029); (c) male sex (n = 26,473); and (d) missing data on smoking status, cervical cancer, cotinine concentrations, or other covariates (n = 18,722). The cervical cancer questionnaire was administered only to individuals aged ≥20 years. Consequently, our study population comprised only adults. Of the 101,316 participants in the NHANES, 8339 met our inclusion criteria for screening. The inclusion and exclusion process is illustrated in Figure 1.

Flowchart of sample selection from NHANES (1999–2018). NHANES: National Health and Nutrition Examination Survey.
Assessment of smoke exposure and cervical cancer risk in the NHANES database
Smoking status was defined based on participants’ responses to a series of questions. First, they were asked the following: “Have you ever smoked more than 100 cigarettes in your lifetime?” Those who answered “No” were classified as “Nonsmokers.” Participants who answered “Yes” were then asked the following: “Do you currently smoke?” Those who responded “Sometimes” or “Every day” were categorized as “Current smokers,” while those who answered “Not at all” were classified as “Former smokers.” In total, participants were grouped into the following three categories: nonsmokers, former smokers, and current smokers.
Cotinine, a key biomarker of nicotine, is the preferred indicator for assessing tobacco exposure through measurements in the blood, urine, and saliva. 21 Compared with other diagnostic tools, cotinine offers advantages such as high sensitivity, good specificity, and a long half-life, making it the optimal marker for tobacco use. 22 Serum cotinine concentrations were measured using liquid chromatography/tandem mass spectrometry with atmospheric pressure ionization. 23 Based on previous studies,24,25 we defined critical values of 0.05 and 3.00 ng/mL to categorize cotinine concentrations into the following three groups: <0.05 ng/mL, 0.05–2.99 ng/mL, and ≥3.00 ng/mL. Due to the skewed distribution of cotinine concentrations in the serum, a logarithmic transformation was applied, and the transformed values were analyzed as continuous variables.
The “Medical condition” section of the NHANES questionnaire is used to collect information about participants’ health status and medical history. It includes questions such as “Have you ever been told by a doctor that you have cancer or a malignancy?” (MCQ220), followed by “What type of cancer was it?” (MCQ230A-D). Women diagnosed with cervical cancer were identified as those with MCQ220 coded as 1 and MCQ230A coded as 15; missing codes for MCQ230C-D indicated cervical cancer. Women without cervical cancer were identified as those with MCQ220 coded as 2. 26
Covariates
Based on previously published studies, we identified factors associated with tobacco smoke exposure, cotinine concentrations, and cervical cancer risk. These factors include age, race, marital status, poverty-to-income ratio (PIR), educational level, body mass index (BMI), waist circumference, alcohol consumption, blood glucose, insulin levels, glycated hemoglobin (HbA1c), albumin, creatinine, hypertension, and diabetes mellitus.26–28 In Table S1, the covariate HPV was additionally added.
Genetic IVs for serum cotinine in MR
This study adheres to the three core assumptions of MR. First, the genetic IV is strongly associated with the smoking variable as the exposure factor. Second, due to the random assortment of SNPs during gametogenesis, the genetic variation serving as an IV for exposure (smoking) is presumed to be independent of any confounding factors. Third, the genetic IV for exposure (smoking) is hypothesized to influence the risk of the outcome (cervical cancer) primarily through smoking exposure rather than via alternative pathways. The study design is shown in Figure 2.

Mendelian randomization study design: smoking and cervical cancer.
Genetic summary data on cervical cancer in MR
In this study, the smoking variables included cotinine levels, ever versus never smokers, and smoking a cigarette or pipe within the last hour. The GWAS summary data for these three smoking variables were sourced from the European Bioinformatics Institute GWAS Catalog (https://www.ebi.ac.uk/gwas). Specifically, the cotinine levels dataset (ID: GCST009965) included 5185 samples; ever versus never smokers dataset (ID: GCST007327) included 518,633 samples; and smoking a cigarette or pipe within the last hour dataset (ID: GCST90043724) included 42,609 samples. The summary statistics for cervical cancer were obtained from the Integrative Epidemiology Unit (IEU) OpenGWAS project (https://gwas.mrcieu.ac.uk/), comprising 199,086 samples, including 563 cases and 198,523 controls.
Detailed information regarding the GWAS summary data is presented in Table 1.
Summary of the GWAS data.

GWAS: genome-wide association studies; IEU: Integrative Epidemiology Unit.
All participants in these cohorts were of European ancestry. It is noteworthy that all the original studies had obtained formal ethical approval and informed consent from all their participants. Therefore, our study did not require any additional ethical approval because it relied solely on summary-level statistics.
IV selection
All IVs for further analysis were carefully selected based on the three key assumptions of MR. 29 To ensure an adequate number of IVs, the significance threshold for smoking-related instruments was set at 5 × 10−6. To address potential bias from linkage disequilibrium (LD) among the selected SNPs, we performed a clumping process (r2 < 0.001, physical window = 10,000 kb) using the 1000 Genomes Project as the reference panel for calculating LD r2. Candidate SNPs were then matched with the GWAS data of the outcome variable based on chromosome and genomic position. The F-statistic was derived using the Cragg–Donald statistic, calculated as F = βexposure2/(standard error (SE)exposure)2. 30 IVs with F-statistic < 10 were excluded from the final selection. Subsequently, we harmonized the exposure and outcome datasets to ensure that the effect alleles were aligned. We also removed palindromic SNPs with intermediate allele frequencies.
Statistical analyses
To ensure national representativeness, we accounted for the complex sampling design of the NHANES dataset, using sample weights in our analyses. We described the baseline characteristics of the overall population and stratified subgroups by smoking status and serum cotinine concentrations using weighted statistics. Categorical variables were presented as percentages, and continuous variables as mean ± SE. p values were calculated using weighted chi-square tests for categorical variables and weighted linear regression for continuous variables. When analyzing serum cotinine concentrations as a continuous variable, log transformation was applied to achieve normality. Univariate analyses were performed based on smoking status and serum cotinine concentrations to examine the associations between covariates and cervical cancer. Logistic regression models were then used to explore the relationship between smoke exposure and cervical cancer risk. The crude model was an unadjusted model, model 1 was adjusted for age and race, and model 2 included additional covariates. Building on model 2, restricted cubic splines (RCS) were applied to assess the potential nonlinear relationship between log-transformed serum cotinine concentrations and cervical cancer risk.
Before imputation, the proportion of missing data for variables included in the NHANES analysis ranged from 0% to 8.98%. The highest proportion of missing data was observed for family PIR (8.98%), followed by alcohol consumption (8.28%), waist circumference (3.82%), and insulin level (1.49%). Other variables had <1.5% missing data, and several variables had no missing values (Table S2). Missing values were addressed using multiple imputation with chained equations (MICE), generating five imputed datasets. This approach ensured the robustness of the results by accounting for missing values in the dataset.
In this MR analysis, inverse variance weighting (IVW) regression was used as the primary method to assess the relationship between smoking and cervical cancer. This method provides a consistent and efficient estimate by synthesizing Wald estimates for each SNP using a meta-analytic approach, yielding the initial MR estimate. 31 To assess the robustness of the results in the presence of potential horizontal pleiotropy among SNPs, we also conducted MR-Egger regression, weighted median method, and simple mode analyses.
MR-Egger regression serves a dual purpose. It assesses horizontal pleiotropy across all genetic instruments and provides an estimate corrected for pleiotropy. 31 The weighted median method provides a consistent estimate of the causal effect when at least 50% of the selected genetic instruments are valid. This method calculates the median of ratio estimates after weighting the SNP estimates 32 ; however, it may produce biased estimates if >50% of the SNPs influence the outcome through pathways other than exposure. The weighted mode method typically generates consistent estimates when a substantial proportion of the individual instrumental effect estimates are derived from valid instruments. 33 Similar to the weighted mode method, the simple mode method may yield biased estimates if the majority of the causal effect estimates are driven by invalid SNPs. 34
For sensitivity analyses, Cochran’s Q test 35 was used to assess the heterogeneity of associations using both MR-Egger and IVW methods. Horizontal pleiotropy was detected using the MR-Egger intercept and MR-PRESSO test. 36 The MR-PRESSO method evaluates overall heterogeneity by regressing SNP–exposure associations against SNP–outcome associations. It then compares the observed distances of each SNP from the regression line to the expected distances under the null hypothesis of no pleiotropy. Outliers identified using the MR-PRESSO global test were excluded, and robustness was further assessed using leave-one-out analysis to evaluate the stability of MR results against any outlier SNPs. Additionally, graphical assessments via funnel plots and scatter plots provided supplementary evaluations of the robustness and symmetry of effect estimates.
All statistical analyses were performed using R version 4.2.3, with the survey, MICE, TwoSampleMR, and MR-PRESSO packages. A two-tailed p value <0.05 was considered statistically significant.
Results
Baseline characteristics in the NHANES dataset
The weighted baseline characteristics of the study population based on smoking status are presented in Table 2. Nonsmokers, former smokers, and current smokers comprised 14.96% (n = 1640), 14.59% (n = 1422), and 70.45% (n = 5277) of the study population, respectively. Significant statistical differences were observed among these smoking groups in terms of age, BMI, waist circumference, blood glucose, HbA1c, race, educational level, marital status, PIR, alcohol consumption, hypertension history, diabetes history, and cervical cancer history (all p < 0.001). However, no significant differences were observed in their insulin, albumin, and creatinine levels. The average age of the participants was 47.64 years, with the majority being non-Hispanic White (70.73%). Compared with other groups, participants in the current smoker group were younger and exhibited lower blood glucose, insulin, HbA1c, and creatinine levels. They generally had lower educational levels; a higher proportion of participants in this group were unmarried, had lower PIR, exhibited higher alcohol consumption, and did not have a history of hypertension or diabetes. Notably, the current smoker group group had a higher prevalence of cervical cancer (9.72%).
Weighted baseline characteristics of participants categorized as per smoking status.
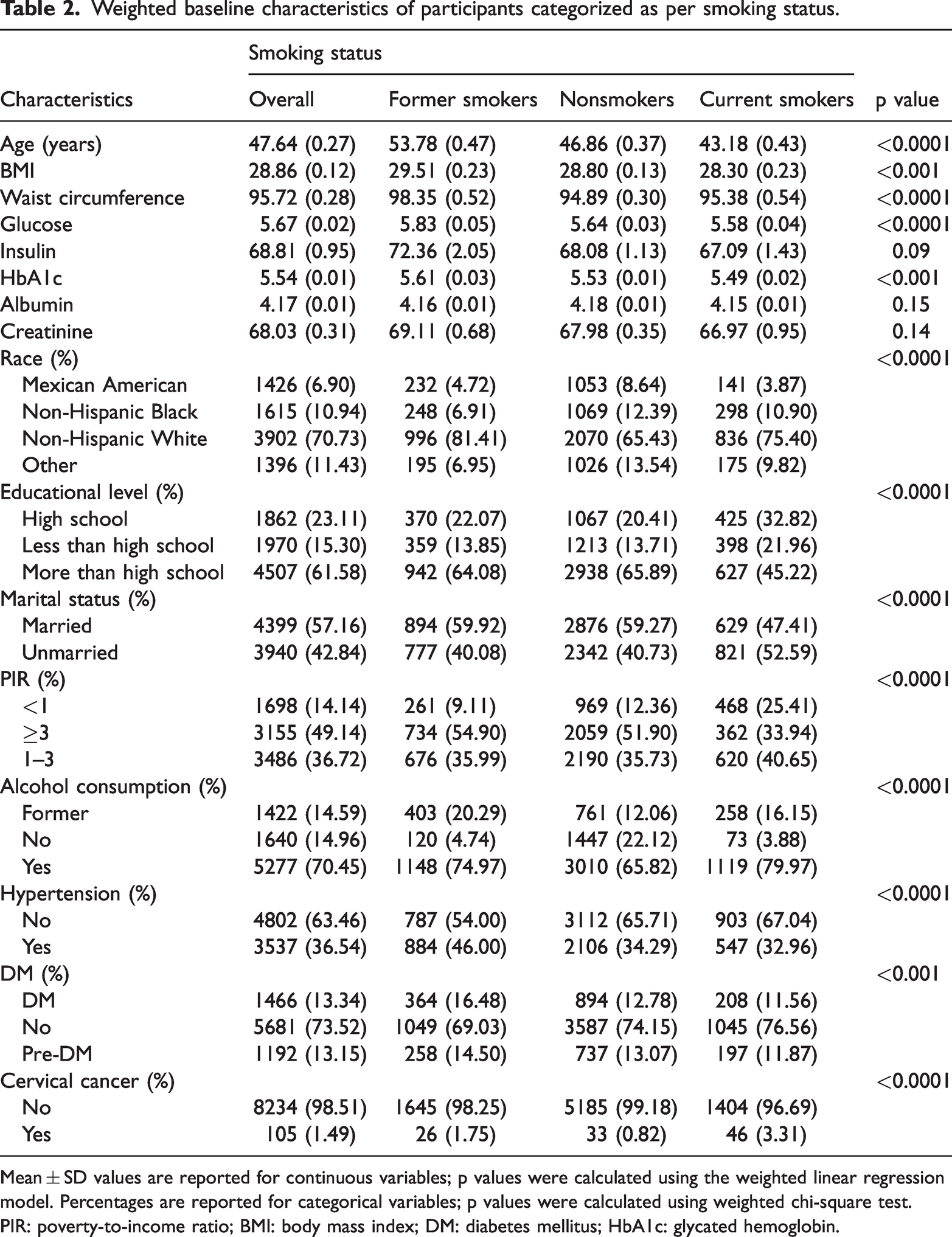
Mean ± SD values are reported for continuous variables; p values were calculated using the weighted linear regression model. Percentages are reported for categorical variables; p values were calculated using weighted chi-square test.
PIR: poverty-to-income ratio; BMI: body mass index; DM: diabetes mellitus; HbA1c: glycated hemoglobin.
Regarding the grouping based on serum cotinine concentrations, the weighted baseline characteristics are presented in Table 3. No significant differences were observed in terms of participants’ diabetes history (p = 0.08). The characteristics of participants with serum cotinine concentrations ≥3.00 ng/mL were similar to those of current smokers. In this group, the majority of the participants were non-Hispanic White (74.02%), had lower educational levels, and showed a relatively higher prevalence of cervical cancer (3.22%).
Weighted baseline characteristics of participants categorized as per serum cotinine concentrations.
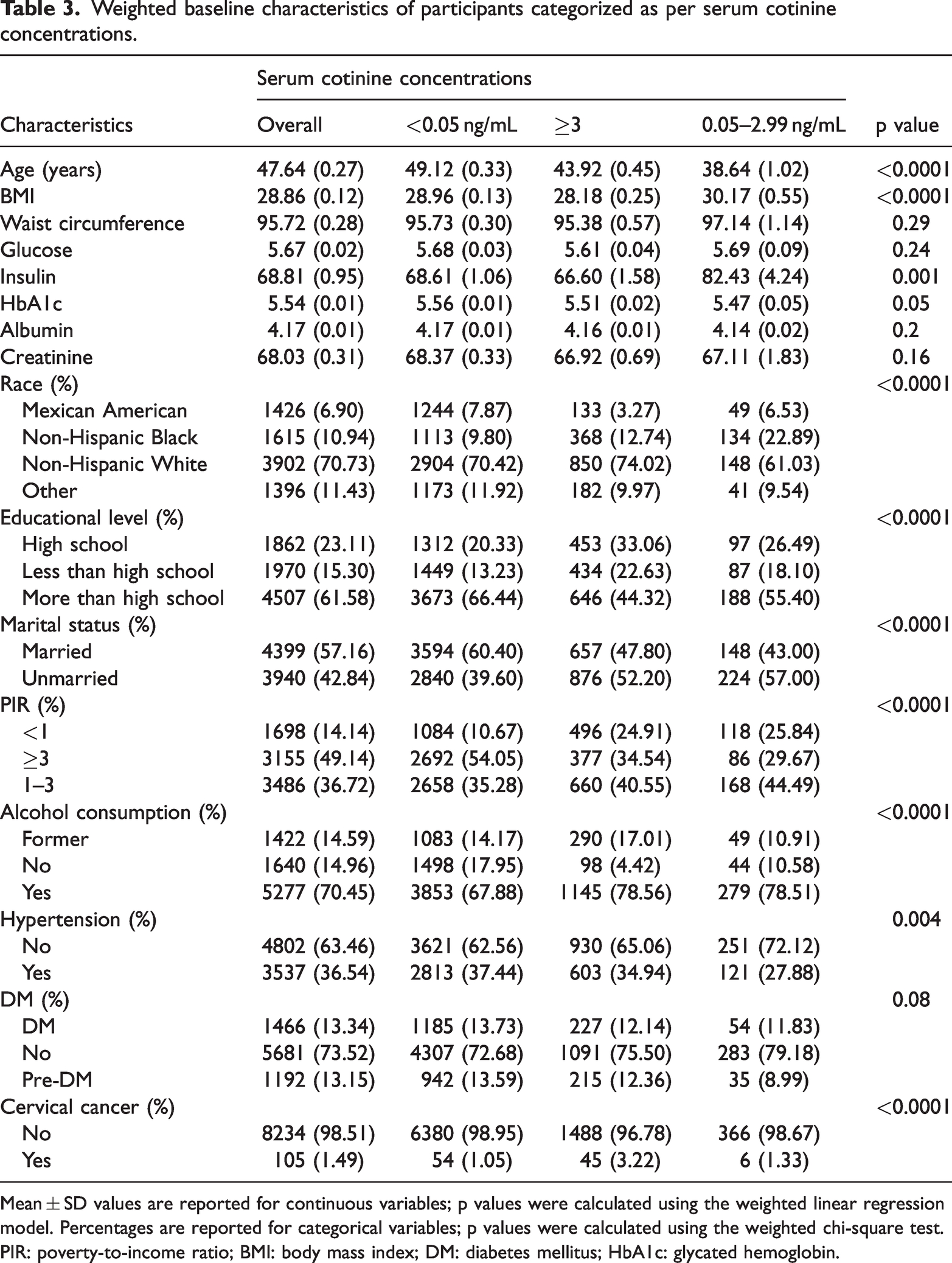
Mean ± SD values are reported for continuous variables; p values were calculated using the weighted linear regression model. Percentages are reported for categorical variables; p values were calculated using the weighted chi-square test.
PIR: poverty-to-income ratio; BMI: body mass index; DM: diabetes mellitus; HbA1c: glycated hemoglobin.
Relationships between smoking status, serum cotinine concentrations, and cervical cancer prevalence in the NHANES dataset
The association between smoking status and cervical cancer is shown in Table 4. Compared with nonsmokers, we found a stronger association between current smokers and the occurrence of cervical cancer, regardless of the adjustment model (crude model: odds ratio (OR) = 4.12 (2.34, 7.25), p < 0.0001; model 1: OR = 3.85 (2.17, 6.82), p < 0.0001; model 2: OR = 3.05 (1.61, 5.78), p < 0.001). However, no significant association was found for former smokers in model 2 (p = 0.09). Table 4 presents the relationship between serum cotinine concentrations, both continuous and categorical, and cervical cancer prevalence. Log2-transformed serum cotinine was associated with an increased prevalence of cervical cancer, with consistent effect sizes across models (p < 0.001). Regardless of the adjustment model, individuals with serum cotinine concentrations of 0.05–2.99 ng/mL and ≥3.00 ng/mL had a higher prevalence of cervical cancer than those with serum cotinine concentrations of <0.05 ng/mL; the highest prevalence was observed in those with serum cotinine concentrations of ≥3.00 ng/mL (model 1: OR = 3.03 (1.85, 4.96), p < 0.0001; model 2: OR = 2.35 (1.38, 4.01), p = 0.002). All results were statistically significant. RCS analysis revealed a nonlinear relationship between log2-transformed serum cotinine concentrations and cervical cancer prevalence (Figure 3; nonlinear p = 0.0407). When serum cotinine concentrations were below the reference value (0.05 ng/mL), cervical cancer prevalence increased with an increase in serum cotinine concentrations. However, when cotinine concentrations exceeded the reference value, the prevalence rose rapidly after a brief fluctuation.
Association between smoking status, serum cotinine concentrations, and risk of cervical cancer.

Crude model: unadjusted model.
Model 1: adjusted for age and race.
Model 2: further adjusted for age, race, marital status, PIR, educational level, BMI, waist circumference, alcohol consumption, glucose level, insulin level, HbA1c level, albumin level, creatinine level, hypertension, and presence of DM
OR: odds ratio; CI: confidence interval; PIR: poverty-to-income ratio; BMI: body mass index; HbA1c: glycated hemoglobin; DM: diabetes mellitus.

RCS analysis of log2-transformed serum cotinine concentrations versus OR for cervical cancer (model 2). RCS: restricted cubic splines; OR: odds ratio.
IV selection in MR
After a series of stringent selections, 5, 7, and 300 SNPs were identified as IVs that are highly correlated with smoking a cigarette or pipe within the last hour, cotinine levels, and ever versus never smokers, respectively. These SNPs exhibited strong statistical associations with smoking (p < 5 × 10−6, r2 < 0.001, 10,000 kb). Subsequent statistical analyses were conducted based on these selected SNPs, all of which had F-statistics >10, indicating the absence of weak instruments. Detailed information on the selected SNPs is provided in Table S3.
Results of MR analysis
In MR studies, we found a significant genetic causal relationship between smoking and cervical cancer risk.
IVW regression was employed as the primary method for causal effect estimation. The results indicated that cotinine levels (OR = 1.0011, 95% CI: 1.0001–1.0021, p = 0.0304), ever versus never smokers (OR = 1.0020, 95% CI: 1.0003–1.0038, p = 0.0192), and smoking a cigarette or pipe within the last hour (OR = 1.0017, 95% CI: 1.0003–1.0032, P = 0.0204) were all positively associated with cervical cancer. These associations were further assessed using additional methods, including MR-Egger regression, weighted median, weighted mode, and simple mode analyses. The results of the five MR analyses are presented in Table 5 and illustrated using scatter plots.
MR analysis of the causal relationship between smoking and cervical cancer.

MR: Mendelian randomization; OR: odds ratio; CI: confidence interval; nSNPs: number of single nucleotide polymorphisms.
Sensitivity analyses
Sensitivity analyses, including tests for pleiotropy and heterogeneity, were performed to strengthen the reliability of the results. MR-Egger intercept tests and MR-PRESSO global tests for all three smoking variables showed no evidence of horizontal pleiotropy (p > 0.05). Additionally, no statistical heterogeneity was found in the MR-Egger and IVW analyses for the positive associations. Detailed results of sensitivity analysis are shown in Table 6. Leave-one-out sensitivity analyses indicated that the causal associations between smoking and cervical cancer risk were not influenced by individual SNPs, confirming the robustness of the findings. Funnel plots were largely symmetrical, further supporting the reliability of the results. Scatter charts, leave-one-out charts, and funnel charts are presented in the supplementary figures.
Results of sensitivity analysis of the causal relationship between smoking and cervical cancer.
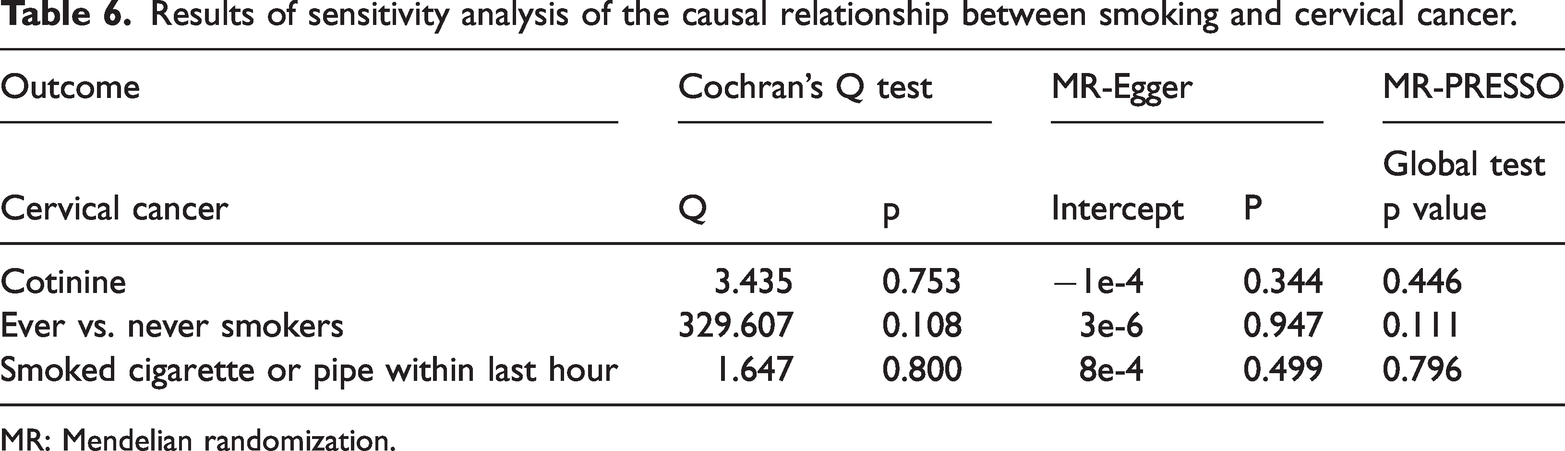
MR: Mendelian randomization.
Discussion
In this study, our findings indicate that smoking exposure is a risk factor for cervical cancer. Our observational analysis revealed an increased prevalence of cervical cancer among current smokers. Furthermore, both observational and MR analyses consistently support a positive association between smoking exposure and cervical cancer risk. To the best of our knowledge, this is the first study to combine observational and MR approaches to examine the relationship between smoking and cervical cancer risk. This study aimed to evaluate this relationship using publicly available datasets. Specifically, the NHANES dataset and publicly accessible GWAS databases were leveraged to integrate observational analyses with MR to assess whether smoking exposure increases cervical cancer prevalence.
We explicitly compared our findings with those of previous studies in the literature. For example, a 2018 meta-analysis 37 demonstrated that passive smoking increased cervical cancer risk; this is consistent with our findings showing that both active and passive smoking are associated with cervical cancer prevalence. Moreover, our results align with those of more recent studies that indicate that smoking exacerbates HPV persistence, a known risk factor for cervical cancer development. These studies emphasize the relevance of our findings and provide context for our research. Our observational study was conducted in a US population, while the GWAS data for serum cotinine concentrations and cervical cancer risk used in the MR analysis were derived from European populations. This cross-population consistency suggests that smoking exposure contributes to cervical cancer risk across diverse ethnic groups. Nicotine has been demonstrated to promote tumor progression in various cancers, including lung, 38 head and neck, 39 and breast cancers. 39 Finally, the pharmacokinetic interactions of tobacco smoke may significantly impact the efficacy and toxicity of anticancer therapies. However, the underlying biological mechanisms linking smoking to cervical cancer remain incompletely understood and warrant further investigation.
HPV infection is the primary etiological factor in cervical cancer. In our fully adjusted models, HPV infection status was included as a covariate to account for this major confounder. In a previous study, smoking contributed to cervical dysplasia and enhanced the persistence of high-risk HPV infections, thereby facilitating the development of malignant lesions in the cervix and uterus. 40
The biological mechanisms underlying the association between smoking and cervical cancer risk remain incompletely understood. However, several potential mechanisms have been proposed.
Immune suppression and HPV persistence: Chronic smoking may impair immune function,
41
thereby increasing susceptibility to HPV infection, which is considered the primary etiological factor in cervical cancer.42,43 Studies suggest that smoking suppresses the immune system, reducing the body’s ability to clear HPV infections, thereby increasing the risk of HPV-induced cervical intraepithelial neoplasia and cervical cancer.4,37 Moreover, smoking may further promote persistent HPV infection by modulating local immune responses and enhancing oxidative stress at the cellular level.
44
Oxidative stress and inflammation induced by smoking: Smoking generates a significant amount of free radicals and ROS, which can cause oxidative damage to cellular DNA, lipids, and proteins. Oxidative stress not only disrupts the cellular structure and function but also triggers chronic inflammatory responses, further promoting cancer development. Research has shown that smoking-induced oxidative stress may contribute to carcinogenesis by increasing intracellular free radicals, leading to genetic mutations and genomic instability, thereby creating a favorable environment for malignant transformation.
9
Smoking-induced DNA methylation alterations: Smoking also promotes carcinogenesis by altering DNA methylation patterns. Smoking-related DNA methylation changes have been closely associated with cervical cancer development. Studies have shown that smoking can lead to hypermethylation in specific genomic regions, particularly in genes involved in cell growth and signal transduction, thereby driving tumor progression.
45
These epigenetic modifications are not merely transient responses; as per a previous study, certain methylation markers can serve as predictive indicators of cancer development, especially during the precancerous stages.
45
Smoking and immune suppression: Smoking may also contribute to cervical cancer progression by suppressing immune system function. Chemical compounds in tobacco, such as nicotine and other carcinogens, can impair immune cell activity, reducing the effectiveness of antitumor immune responses. This immune evasion mechanism allows cancer cells to survive and proliferate within the body, further facilitating tumor progression and metastasis.39,46 Impact of smoking on cervical cancer treatment response: Smoking not only plays a role in cervical cancer development but also negatively affects treatment outcomes. It has been associated with poor therapeutic response, increased recurrence rates, and reduced quality of life in cervical cancer patients. The detrimental effects of smoking on chemotherapy and radiotherapy may be attributed to smoking-induced oxidative stress and immune suppression, which can diminish treatment efficacy and increase therapy-related complications.
47
Smoking influences the initiation and progression of cervical cancer through multiple biological pathways. It increases cancer risk by enhancing HPV infection persistence, promoting oxidative stress, altering DNA methylation patterns, and suppressing immune function. Additionally, smoking exacerbates the burden of cervical cancer by impairing treatment response and increasing therapy-related complications. Therefore, smoking prevention and cessation strategies are crucial in cervical cancer prevention and management. Further research is needed to explore the underlying mechanisms by which smoking contributes to cervical carcinogenesis.
Our study has several strengths. First, we used a large and representative sample from the NHANES database, which enhanced the generalizability of our findings. Second, we combined observational data with MR analysis. Although observational studies such as NHANES cannot establish causality due to confounding factors and reverse causality, MR overcomes these issues by utilizing large-scale GWAS data, providing the statistical power needed to explore the link between tobacco exposure and cervical cancer. Importantly, our study is the first to combine NHANES data with MR analysis to investigate the causal relationship between smoking and cervical cancer. MR helps address confounding and reverse causality, strengthening our causal inference. This innovative approach improves our understanding of smoking as a cause of cervical cancer and has important implications for public health and prevention strategies.
However, the following limitations should be considered while interpreting our results:
Cross-sectional design: First, our cross-sectional analysis excluded participants with incomplete data, which may have introduced selection bias. Additionally, the cross-sectional nature of the NHANES dataset prevents us from establishing a temporal relationship between smoking initiation and cervical cancer diagnosis. Therefore, our findings should be interpreted as associations rather than causal evidence. Self-reported data: The reliance on self-reported smoking status may have introduced reporting bias, which is a common issue in large-scale surveys. This limitation could have affected the accuracy of our data. Genetic data: Although MR strengthened our findings, it relies on genetic summary data, which may not capture all sources of variation or pleiotropy. Moreover, since the genetic data used in the MR analysis are predominantly from European and American populations, the generalizability of our findings to other ethnic groups may be limited. Hypothesis-generating nature: The primary goal of our study was hypothesis generation, and our findings were derived from bioinformatics analysis. Although the results are promising, they require clinical validation and in vivo experiments to confirm the causal relationship between smoking and cervical cancer.
Conclusion
Our study provides important insights into the relationship between smoking exposure and cervical cancer risk, using both observational data and MR analysis. Although our findings suggest a significant association, further clinical studies and in vivo experiments are necessary to confirm this causal relationship.
The next steps involve validating these results through clinical trials and exploring the mechanisms by which smoking affects cervical carcinogenesis. Our study may guide future clinical research by identifying smoking as a modifiable risk factor for cervical cancer, potentially informing prevention strategies and therapeutic approaches.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251383687 - Supplemental material for Smoking exposure and cervical cancer risk: Integrating observational and genetic evidence
Supplemental material, sj-pdf-1-imr-10.1177_03000605251383687 for Smoking exposure and cervical cancer risk: Integrating observational and genetic evidence by Luyang Su, Ren Xu, Yanan Ren, Cuiqiao Meng, Pei Wang, Qi Wu, Liyun Song and Zeqing Du in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251383687 - Supplemental material for Smoking exposure and cervical cancer risk: Integrating observational and genetic evidence
Supplemental material, sj-pdf-2-imr-10.1177_03000605251383687 for Smoking exposure and cervical cancer risk: Integrating observational and genetic evidence by Luyang Su, Ren Xu, Yanan Ren, Cuiqiao Meng, Pei Wang, Qi Wu, Liyun Song and Zeqing Du in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605251383687 - Supplemental material for Smoking exposure and cervical cancer risk: Integrating observational and genetic evidence
Supplemental material, sj-pdf-3-imr-10.1177_03000605251383687 for Smoking exposure and cervical cancer risk: Integrating observational and genetic evidence by Luyang Su, Ren Xu, Yanan Ren, Cuiqiao Meng, Pei Wang, Qi Wu, Liyun Song and Zeqing Du in Journal of International Medical Research
Supplemental Material
sj-pdf-4-imr-10.1177_03000605251383687 - Supplemental material for Smoking exposure and cervical cancer risk: Integrating observational and genetic evidence
Supplemental material, sj-pdf-4-imr-10.1177_03000605251383687 for Smoking exposure and cervical cancer risk: Integrating observational and genetic evidence by Luyang Su, Ren Xu, Yanan Ren, Cuiqiao Meng, Pei Wang, Qi Wu, Liyun Song and Zeqing Du in Journal of International Medical Research
Footnotes
Acknowledgments
We thank all the participants, staff, and institutions who contributed to the NHANES database and the publicly available GWAS database.
Author contributions
Writing–original draft: SLY, XR, and DZQ. Formal analysis: RYN. Writing–review and editing: RYN, MCQ, WP, WQ, and SLY. Investigation and data visualization: WP and WQ. Software: SLY. Supervision: SLY.
Consent for publication
All authors have read the manuscript and have agreed to its publication.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary material; further inquiries can be directed to the corresponding author.
Declaration of conflicting interests
The authors declare no conflict of interest.
Ethics approval and consent to participate
The Centers for Disease Control and Prevention (CDC) and the National Center for Health Statistics (NCHS) are responsible for conducting NHANES. All participants provided informed consent forms. The NHANES study protocol was approved by the NCHS Ethics Review Board. The genome-wide association studies (GWAS) data used in this study were derived from publicly published original studies that had obtained informed consent from participants and ethics committee approval at the time of publication.
Funding
The authors declare that financial support was received for the research, authorship, and/or publication of this article. The study was funded by the Medical Science Research Project of Hebei (NO. 20250293).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
