Abstract
Background
Lung cancer remains the leading cause of cancer-related deaths globally. Patients with postoperative recurrence typically have limited treatment options. This study evaluated the safety and feasibility of computed tomography–guided radiofrequency ablation for treating high-risk pulmonary nodules in patients who have undergone lung cancer surgery and are ineligible for reoperation.
Methods
We retrospectively analyzed 40 patients (16 men and 24 women, aged 41–78 years) with radiologically suspicious recurrent nodules following lung cancer resection who were treated with radiofrequency ablation at West China Hospital (July 2023–August 2024). Outcomes included procedural success, occurrence of complications, hospitalization duration, 30-day mortality, and imaging response.
Results
All procedures were technically successful. Five minor complications occurred in 12.5% of the patients, including fever (n = 2) and pneumothorax (n = 3). No deaths occurred within 30 days, and no major adverse events (e.g. hemothorax) were observed. The median postoperative length of hospital stay after ablation was 2.0 days. Furthermore, 82.5% of the patients were followed up and had achieved complete ablation at the 3-month mark.
Conclusion
Computed tomography–guided radiofrequency ablation is a minimally invasive, safe, and effective option for managing high-risk pulmonary nodules in postoperative lung cancer patients who are deemed ineligible for reoperation. Its low complication rate, rapid recovery, and promising local control support its integration into multimodal treatment algorithms. Larger prospective studies are needed to validate long-term outcomes.
Keywords
Introduction
Globally, there are approximately 600,000 new cases of lung cancer each year. Lung cancer is the malignancy with the highest mortality rate, posing a serious threat to public health. 1 Surgical resection remains the standard treatment for patients with resectable primary lung cancer or solitary metastatic nodules. 2 However, approximately two-thirds of non–small cell lung cancer (NSCLC) patients are ineligible for radical resection due to cardiopulmonary comorbidities, other extra thoracic diseases, or advanced age. 3 The lung is also the second most common site for metastasis, and local therapy may be effective for patients with solitary pulmonary metastatic lesions. 4
In recent years, radiofrequency ablation (RFA) has been extensively evaluated as an alternative minimally invasive treatment for inoperable malignant pulmonary nodules, with major advantages including shorter length of hospital stay, lower costs, and fewer complications. Therefore, as a minimally invasive surgical technique, RFA plays an increasingly important role in comprehensive tumor treatment strategies. A retrospective study showed that RFA significantly prolonged progression-free survival and overall survival in patients with liver-only recurrence after pancreatic resection while reducing surgical trauma and associated risks; 5 RFA has recently been used for managing tumors of the kidneys, adrenal glands, thyroid gland, and breasts.6–9 RFA involves the insertion of an electrode to deliver radiofrequency energy, generating localized high temperatures that cause necrosis of the target tissue. However, there are limited reports on the prognosis of NSCLC patients at different stages who underwent computed tomography (CT)–guided percutaneous RFA.10,11
The clinical safety and efficacy of RFA in treating lung cancer patients remain controversial. Although RFA demonstrates utility for local tumor control in medically inoperable cases, current evidence does not support its equivalence to surgical resection for early-stage disease, 12 and it is contraindicated for central tumors due to technical limitations. 13 Numerous studies have evaluated its effectiveness and safety in treating malignant pulmonary nodules.14,15 It remains highly controversial whether it can be used for high-risk pulmonary nodules in postoperative recurrent lung cancer patients who are ineligible for surgery.14,15 Therefore, the aim of our study was to assess the complication rate and 30-day perioperative survival rate in patients with high-risk pulmonary nodules after lung cancer surgery who were treated with RFA, thereby evaluating the effectiveness and safety of RFA in patients with suspicious lung nodules who are ineligible for surgery. At present, there is a lack of such studies internationally.
Materials and methods
Clinical data of patients
From July 2023 to August 2024, a total of 40 patients (16 men and 24 women) aged between 41 and 78 (mean age: 58.2 ± 9.4) years underwent CT-guided RFA for pulmonary nodules at West China Hospital, Sichuan University. All patients had previously undergone a single surgical resection for primary lung cancer between 2017 and 2022. At least 6 months after the surgery, they developed a single high-risk pulmonary nodule indicating ipsilateral recurrence, and the interval between the initial operation and subsequent ablation was more than 1 year in all cases. Due to poor cardiopulmonary function, the presence of extra cardiopulmonary diseases, refusal of surgery, or advanced age, surgery could not be performed in these patients. Regarding comorbidities, 11 patients had diabetes, 1 had coronary heart disease, 9 had hypertension, and 26 had other comorbidities; in addition, 25 of these patients had more than 2 concurrent conditions. The ablation sites were as follows: (a) upper lobe of the right lung (n = 13); (b) upper lobe of the left lung (n = 11); (c) lower lobe of the left lung (n = 7); (d) lower lobe of the right lung (n = 5); and (e) middle lobe of the right lung (n = 4). The characteristics of the enrolled patients are presented in Table 1. All patient data were deidentified prior to analysis to ensure confidentiality and protect personal information. The data were sourced from West China Hospital, Sichuan University.
Patient characteristics.
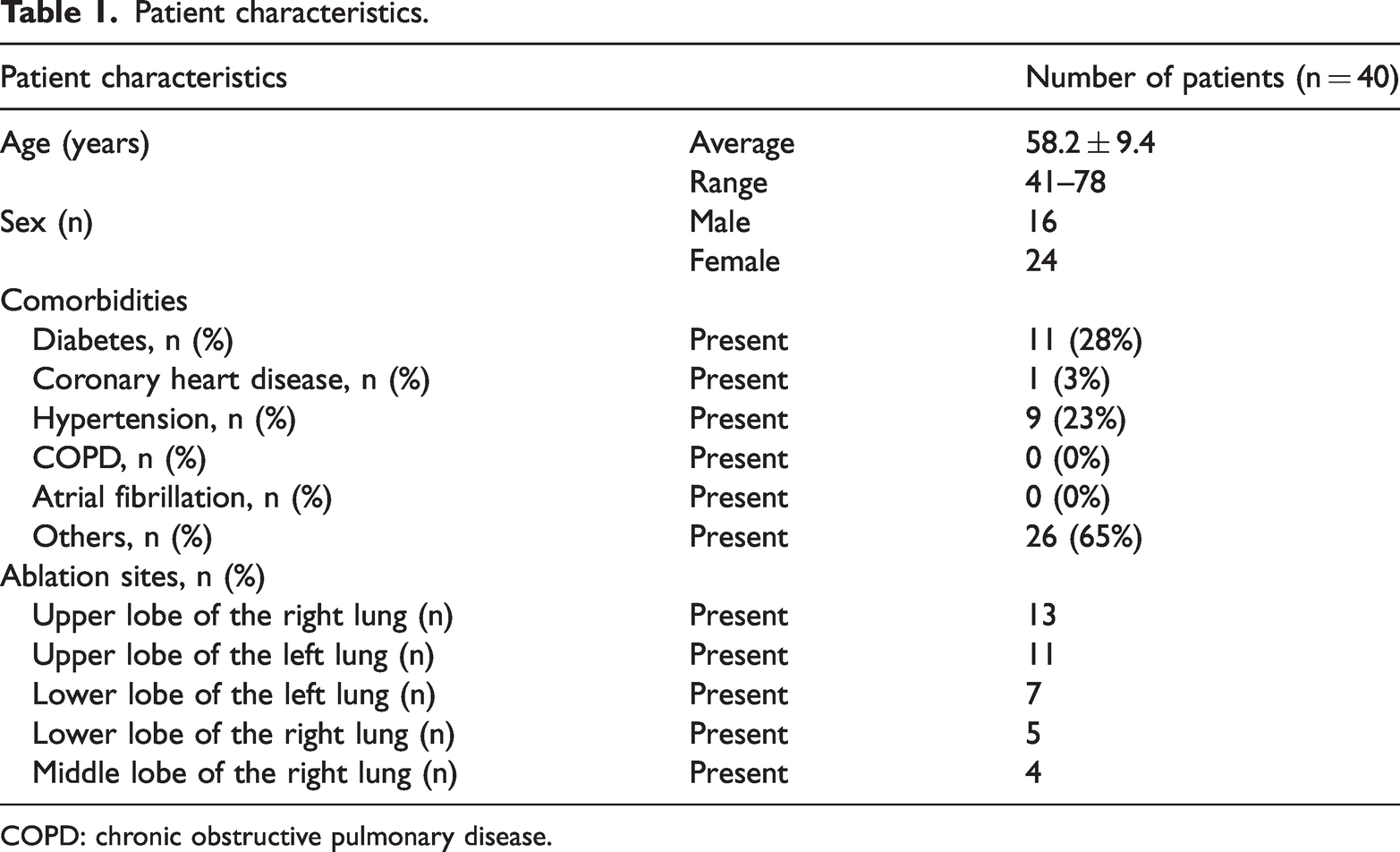
COPD: chronic obstructive pulmonary disease.
Patient eligibility was determined through comprehensive clinical evaluation, comprising a meticulous review of medical histories, physical examinations, and recent imaging studies. Thoracic contrast-enhanced CT (typically acquired within 2 weeks preoperatively) served as the cornerstone imaging modality for assessing tumor dimensions, location, and spatial relationships with vital structures. Tumors demonstrating perihilar proximity or immediate adjacency to major vascular territories were routinely excluded from therapeutic consideration based on the established safety criteria. When clinically indicated, staging investigations, including whole-body bone scintigraphy and cerebral magnetic resonance imaging, were supplemented with positron emission tomography (PET)–CT, where available, to exclude distant metastases, with priority given to candidates demonstrating curative ablation potential for precise tumor, node, metastasis staging.
The study protocol was in accordance with the principles of the Helsinki Declaration of 1975, as revised in 2024, and was approved by the Institutional Ethics Committee (approval number: (Review No. 2024-2070)). As a retrospective chart review study, the Ethics Committee waived the requirement for informed consent. This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 16 We included 40 patients with high-risk pulmonary nodules suggestive of recurrence after lung cancer surgery, all of whom had been pathologically diagnosed with NSCLC prior to surgery and were comprehensively diagnosed with high-risk pulmonary nodules before undergoing ablation. The mean diameter of the high-risk pulmonary nodules was 1.18 ± 0.34 (0.5–2.5) cm, and the diagnoses were confirmed cytologically or histologically for all enrolled patients.
Treatment methods
Prior to ablation, patients routinely undergo enhanced chest CT, electrocardiography, pulmonary function test, complete blood count, tumor marker assessment, liver and kidney function tests, arterial blood gas analysis, and coagulation profile analysis. During the RFA procedure, the patient’s position is determined based on the shortest distance from the lesion to the body surface. For instance, if the patient is in the supine position, after CT localization of the pulmonary nodule, routine disinfection and draping are performed. Successful local infiltration anesthesia is achieved using 1% lidocaine hydrochloride. The ablation needle is then inserted along the CT-guided point, advancing 1 cm beyond the tumor. After setting the target temperature, ablation is maintained for 2 min per cycle and is performed consecutively for two cycles. A repeat CT scan is conducted to confirm that both the tumor and the surrounding 2-cm margin have been ablated before withdrawing the needle. The puncture site is dressed and secured with sterile materials. After ensuring that the patient has no significant discomfort, they are safely shifted to the ward.
RFA procedures were performed using the RF 3000™ Radiofrequency Ablation System Generator (Boston Scientific Corporation, Natick, MA, USA) and LeVeen SuperSlim™ Needle Electrodes (model: 2.0-cm array diameter; cannula length: 15/25 cm; Order Numbers 26-226/26-227).17,18 Ablations utilized pulsed-RF mode with impedance-controlled energy delivery. The generator’s real-time impedance feedback system (baseline: 40–80 Ω) automatically modulated power output (maximum 200 W) until achieving the procedural endpoint—an abrupt impedance rise to ≥220 Ω—indicating complete tissue desiccation. Electrode deployment was monitored under CT guidance, utilizing the device’s echogenic tip and 1-cm shaft markers for precise placement. During puncture, the shortest pleural-to-lesion trajectory was selected while avoiding major pulmonary arteries, aortic branches, and critical vasculature. Ablation parameters included temperature adjustment (65°C–95°C) and power modulation (30–100 W), with the entire procedure typically lasting 12–20 min. We performed CT evaluations immediately after the ablation and then at 3 months, 6 months, and 1 year after the procedure. Post-ablation assessment of the treatment zone was performed according to the following criteria:
Radiological assessment criteria following ablation 19
Complete ablation was characterized by the following:
Lesion disappearance; Complete cavity formation; Fibrosis or scar formation; Solid nodule involution or stability (no contrast-enhanced signs on CT and/or no fluorodeoxyglucose (FDG) uptake on PET/CT); Lesion within atelectasis (no contrast-enhanced signs on CT and/or no FDG uptake on PET/CT).
Incomplete ablation was characterized by the following:
Partial cavity formation with residual solid/liquid components (irregular peripheral/internal enhancement on CT and/or intense FDG uptake on PET/CT); Partial fibrosis with solid residues (irregular peripheral/internal enhancement on CT and/or intense FDG uptake on PET/CT); Solid nodule with unchanged or increased size (irregular peripheral/internal enhancement on CT and/or intense FDG uptake on PET/CT); Lesion within atelectasis (contrast-enhanced signs on CT and/or intense FDG uptake on PET/CT).
Local progression was characterized by the following:
Enlargement ≥10 mm with expanded irregular/internal enhancement on CT and/or increased FDG uptake on PET/CT; Newly developed local lesion (new enhancement signs on CT and/or new intense FDG uptake on PET/CT).
Observational indicators
After ablation, we applied the criteria shown in Table 2 for evaluation.
Observational indicators.

Statistical analyses
We performed descriptive statistical analyses of the patients’ sex, age, comorbidities, ablation extent, 30-day postoperative mortality, length of hospital stay, postoperative complications, and intraoperative safety, defined by the success rates of localization, intraoperative bleeding, and other intraoperative safety events. All statistical analyses were performed using Python version 3.12.5 and the ‘pandas’ package. Calculations included frequency, percentage, mean, SD, minimum, and maximum values. All statistical results were saved in an Excel file (2021 version) for further reporting and tabular presentation.
Results
All 40 enrolled patients successfully underwent the intervention; complete ablation was achieved in all cases, and no major intraoperative complications or mortality occurred. None of the patients experienced severe postoperative pain. Among the patients who received the intervention, the accuracy rate was 100%; the average blood loss during the ablation was 5–10 mL, and no adverse events occurred. Postoperatively, two patients developed fever, and three experienced pneumothorax. Fever was managed with physical cooling or antibiotics, while pneumothorax was treated using closed thoracic drainage, with two patients requiring drainage for more than 7 days. The 30-day postoperative mortality rate was 0%. Perioperative complications that occurred in the enrolled patients are presented in Table 3. The median length of hospital stay following ablation was 2.0 days, with the first quartile at 1.0 day and the third quartile at 2.25 days, resulting in an interquartile range of 1.25 days. Although most patients had advanced age and severe comorbidities, among those who were successfully followed up, 82.5% were confirmed to have achieved complete ablation by the 3-month mark. Moreover, only a few patients completed the full series of CT follow-up at 1, 3, and 6 months after ablation.
Incidence of complications after RFA.
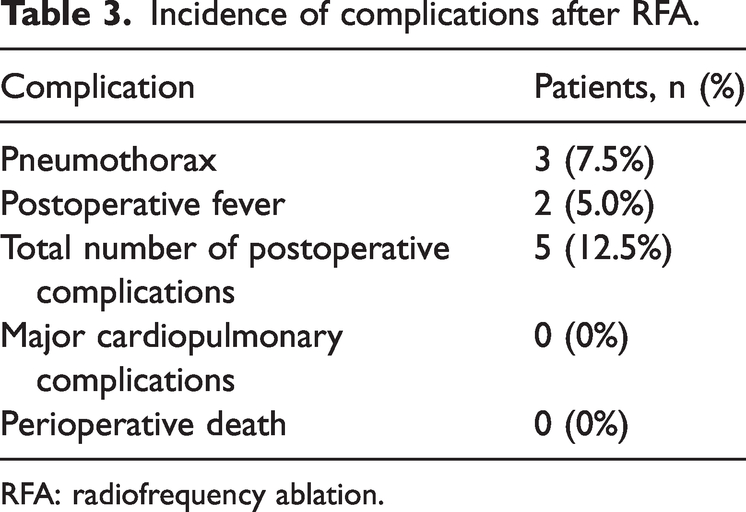
RFA: radiofrequency ablation.
In this subset, we observed the progression of imaging changes following RFA. Initially, mild-to-moderate inflammatory changes were noted at the puncture site on CT after successful ablation. By 3 months after ablation, cavitation typically formed at the ablation site, followed by a marked reduction in the size of the nodules at the treated site. The classic manifestations are illustrated in Figure 1(a) to (f). This phenomenon is consistent with our radiological assessment criteria.

Computed tomography results (a) before the puncture; (b) after the puncture; (c) after ablation; (d) 3 months after ablation; (e) 6 months after ablation; and (f) 12 months after ablation.
Discussion
In this study, we performed CT-guided percutaneous RFA in 40 patients with high-risk nodules suspected of recurrence after lung cancer surgery. The overall incidence of postoperative complications was 12.5%, with complications observed in five cases. Given that all ablations were conducted >12 months after the initial surgery and considering the temporal onset of the observed complications, these events are unlikely to be attributable to the previous surgical procedure. The median postoperative length of hospital stay after ablation was 2.0 days. Most importantly, the 30-day postoperative mortality rate was 0%. Our findings suggest that RFA may be a safe and effective alternative for local control of NSCLC and metastatic lung nodules in patients who are ineligible for curative surgery due to cardiopulmonary comorbidities, extra cardiopulmonary diseases, or advanced age.
Image-guided percutaneous RFA was first introduced in 1990 for the treatment of liver tumors and has since become the preferred ablative technique worldwide for treating tumors of the kidneys, adrenal glands, thyroid gland, lungs, and breasts.5,15,20,21 Undoubtedly, surgical resection is the first-line treatment for early-stage NSCLC, with lobectomy being the gold standard, which has been shown to be more effective than sub-lobar resection. A large randomized controlled trial involving early-stage NSCLC patients demonstrated that lobectomy achieves better resection margins, improves patient survival, and reduces local recurrence rates. 22 For patients with locally advanced stage II or III NSCLC who are ineligible for surgery, concurrent chemoradiotherapy or immunoradiotherapy is considered a more appropriate treatment option. 23 However, it is important to note that prolonged chemoradiotherapy may lead to drug development and seriously affect the clinical outcome in advanced NSCLC patients. 24 Therefore, it is essential to adopt more effective treatment strategies to improve the quality of life of patients with NSCLC.
Pre-ablation, high-risk pulmonary nodules were identified based on imaging characteristics and patients’ surgical history, as evaluated by a specialized clinical team. In this context, “high-risk pulmonary nodules” refers to nodules highly suspicious for the recurrence of primary NSCLC after the initial surgery, for which pathological confirmation is not feasible due to patient-related limitations. Conventional solid tumor response criteria are not suitable for assessing local efficacy following ablation. Imaging during the first 3 months after ablation primarily reflects inflammatory changes and edema rather than true tumor control. Early post-ablation changes (within 1 week) typically include the following: (a) intralesional solid, honeycomb, or low-density cavitary alterations; (b) surrounding ground-glass opacity (GGO) extending ≥5 mm beyond the tumor margin, indicative of potentially complete ablation; and (c) a slightly hyperdense reactive zone outside the GGO, forming the characteristic “fried egg” or “cockade” appearance. During the intermediate phase (1 week to 3 months), the ablation zone may exhibit slight enlargement, resolution of GGO, and the development of a sharply defined enhancing ring (“egg shell” sign), with pleural thickening frequently observed in lesions adjacent to the chest wall. Beyond 3 months, lesions generally stabilize or gradually regress, with potential evolution patterns including fibrosis, cavitation, nodular formation, atelectasis, or enlargement, the latter potentially representing recurrence or fibrotic proliferation. The presence of the characteristic “egg shell” sign facilitates effective assessment of post-ablation imaging. 19
A meta-analysis indicated that pneumothorax is the most common complication following RFA, with an incidence of 3.70%–59.00% in lung cancer patients; this aligns with our results. 25 Among the five patients with postoperative complications, three had more than two comorbidities, and each of these patients had at least one comorbidity. Previous studies have identified several risk factors for pneumothorax, including the lower location of lung tumors, advanced age, smaller tumor size, and presence of emphysema. Specifically, the trajectory length through the aerated lung has been shown to be an independent risk factor for pneumothorax. 26 Since its first application in lung tumors in 2000, 20 the clinical safety and efficacy of RFA have garnered increasing attention due to its widespread use. Yamagami et al. reported that the 30-day mortality rate associated with CT-guided RFA is between 0.4% and 2.6%.27–30 In our study, no major life-threatening complications or deaths occurred within 30 days postoperatively, consistent with the low perioperative mortality rates reported by other similar studies.6,15 These results suggest that RFA is safe for use in clinical practice. In another study, the most common postoperative complication was chest pain, possibly caused by nerve burning 14 ; however, no such complication was observed in our study. The median postoperative length of hospital stay after ablation was 2.0 days in our study; this differed from the average length of hospital stay of 6–8 days reported in a previous retrospective study involving lung cancer patients. 31 At our institution, the average length of stay after video-assisted thoracic surgery (VATS) lobectomy is 2–5 days, demonstrating that the results of RFA can be achieved within a significantly shorter duration of hospital stay than those of surgery. In patients with poor baseline conditions, RFA facilitates early postoperative recovery, contributing to an overall favorable post-intervention outcome.
Limitation
This study has several limitations that must be considered when interpreting the results. First, the relatively small sample size of 40 patients may limit the statistical power and generalizability of the findings. Second, the absence of a control group makes it difficult to contextualize outcomes and draw definitive conclusions regarding treatment efficacy. Furthermore, as we could not obtain pathological specimens both before and after ablation, the initial diagnosis and assessment of complete tumor necrosis was based on imaging criteria rather than the gold standard of histological verification. Additionally, the potential existence of micro-metastases beyond the detection threshold of CT imaging could not be evaluated, introducing uncertainty regarding long-term oncological outcomes. Finally, although collaborative multi-institutional studies exist beyond those cited here, the specific metrics used in this study possess characteristics that are unique and differ from those used in established reporting frameworks. This novelty underscores the need for future validation through standardized comparative studies and the incorporation of supplementary assessment scales.
Conclusion
This study demonstrated that RFA is a safe and effective alternative treatment for patients with high-risk suspicious pulmonary nodules who are ineligible for surgery. It offers a new therapeutic option for patients with recurrent high-risk pulmonary nodules after lung cancer surgery who are unable to undergo surgery owing to poor baseline conditions or other reasons. In our study population, the accuracy rate of the intervention was 100%, the amount of blood loss during ablation was 5–10 mL, and no adverse events occurred. The 30-day postoperative mortality rate was 0%, the incidence of postoperative complications was low, and the length of hospital stay was notably short. These findings suggest that CT-guided percutaneous RFA is a safe and promising treatment option for patients with high-risk suspicious pulmonary nodules after lung cancer surgery. Further research is warranted to confirm broader clinical applications.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251382006 - Supplemental material for Safety and feasibility of computed tomography–guided radiofrequency ablation for treating high-risk pulmonary nodules in postoperative lung cancer patients
Supplemental material, sj-pdf-1-imr-10.1177_03000605251382006 for Safety and feasibility of computed tomography–guided radiofrequency ablation for treating high-risk pulmonary nodules in postoperative lung cancer patients by Xiang Lin, Jian Zhou, Beinuo Wang, Zhenghao Dong, Junjie Zhang, Fan Liu and Hu Liao in Journal of International Medical Research
Footnotes
Acknowledgments
We acknowledge the use of DeepSeek AI, a large language model developed by DeepSeek, for English language editing and refinement of the manuscript. The tool assisted in improving grammatical accuracy, sentence structure, and overall readability. It is important to note that all content processed using the AI tool was thoroughly reviewed, revised, and approved by the authors, who assume full responsibility for the final version of the manuscript.
Author contributions
Xiang Lin was responsible for writing the original draft, study methodology, and formal analysis. Jian Zhou was responsible for providing suggestions for important changes. Beinuo Wang was responsible for conceptualization. Zhenghao Dong was responsible for data validation. Junjie Zhang was responsible for data collection. Hu Liao was responsible for supervision, visualization, writing–review and editing. All authors have read and approved the final version of the manuscript.
Availability of data and material
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors declare no competing interests.
Ethical approval
This research was performed in accordance with the Helsinki Declaration of 1975, as revised in 2024, approved by the Medical Ethics Committee of West China Hospital, Sichuan University (NO.2024-2070). All procedures were performed in compliance with relevant laws and institutional guidelines.
Funding
No funding was received.
Informed consent
This retrospective study was conducted after obtaining permission from the Ethics Committee of West China Hospital; informed consent from the patients was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
