Abstract
Objective
To determine the 3-month heart disease–related mortality rate among patients with lung cancer who have undergone lobectomy and identify the common risk factors for mortality to provide a basis for clinical risk assessment and intervention strategies.
Methods
We conducted a retrospective cohort study using the Surveillance, Epidemiology, and End Results Program database (2010–2015), including 35,094 patients with stage I–III lung cancer who underwent surgery. Logistic regression analyzed mortality at 3, 7, and 11 months, focusing on non-cancer causes, particularly heart diseases.
Results
Among the 35,094 patients, 3.24% (1138) died within 3 months, with heart disease accounting for 8.96% (102) of the deaths, a significantly higher percentage than that at 4–7 months (p = 0.017). Logistic regression analysis showed that several factors, including sex, age, postoperative radiotherapy, and postoperative systematic treatment were independent factors contributing to mortality owing to heart diseases within 3 months of surgery.
Conclusions
In this study, we analyzed the causes of early death in patients who had undergone lung cancer surgery and determined the independent risk factors associated with 3-month mortality due to heart disease in these patients.
Introduction
For an extended period, lung cancer has ranked among the most prevalent malignancies worldwide, posing a severe threat to human life and impeding longevity. 1 Surgical treatment that aims to improve the survival rate and quality of life of patients by removing the tumor tissue has long been recognized as one of the most crucial treatment methods for patients with stage I–IIIa non–small cell lung cancer (NSCLC) in clinical practice.2–4 In addition, video-assisted thoracoscopic surgery for lung resection has garnered extensive acclaim within the medical community; concomitantly, the use of lobectomy and sub-lobar resection in early-stage management of pulmonary pathologies has witnessed a remarkable upsurge.5,6 Despite undergoing surgical treatment with curative intent, some patients with lung cancer die in the early postoperative phase. 7
Numerous factors influence the survival and mortality rates of patients with lung cancer undergoing resection surgery to varying degrees. Among various factors, cardiovascular events have gradually attracted increasing attention.8–10 In patients with lung cancer, diverse risk factors are associated with the occurrence and development of cardiovascular diseases. First, prior to undergoing surgical procedures, patients with lung cancer are likely to present with pre-existing cardiovascular disorders. 11 Second, many patients with lung cancer are long-term smokers. Cigarette smoking is a well-established risk factor for cardiovascular damage that could increase the incidence of postoperative cardiovascular complications.12,13
The early postoperative period is a crucial phase for patients who have undergone lung resection. 14 A former study reported a mortality rate of 17.2% within the first 90 days after pneumonectomy, and cardiovascular complications were a major risk factor. 15 In another study, within 3 months of surgery, the patients' bodies were in a stage of recovery and adaptation; however, the onset of heart diseases severely impacted the overall postoperative prognosis of these patients. 16 Previous studies have extensively explored aspects such as the 1-month and 3-month mortality rates as well as postoperative risk in patients with lung cancer.17,18 However, studies reporting risk factors for heart disease–related mortality within 3 months of lung cancer surgery remain relatively scarce.
It is crucial to precisely determine the risk factors contributing to death due to heart disease within the first 3 months following lung cancer surgery. This knowledge empowers clinicians to perform highly accurate risk assessments for patients with a high susceptibility to heart disease and devise customized monitoring and intervention strategies, thereby enhancing the long-term survival rates and improving the overall prognoses of patients with lung cancer.
In this retrospective study, we analyzed data from the Surveillance, Epidemiology, and End Results (SEER) database, which includes a large number of cases of patients with postoperative lung cancer. The aim of this study was to determine the 3-month heart disease–related mortality rate among patients with lung cancer who have undergone lobectomy and identify the common mortality risk factors.
Patients and methods
Ethics statement
We conducted our study in accordance with the Helsinki Declaration of 1975 as revised in 2024. Our study does not involve any specific information of patients. Moreover, we have followed the Enhancing the QUAlity and Transparency Of health Research (EQUATOR) guidelines for observational studies. 19 Individual, identifiable patient information was excluded in the SEER database; therefore, no ethical approval from the Institutional Review Board was required for the study.
Patient selection
We obtained data from the SEER 17 Regs Custom Data (Incidence), November 2023 submission (2010–2015) and used the SEER*Stat software (version 8.4.0) for data extraction (https://seer.cancer.gov/datasoftware/documentation/seerstat/). The process of patient selection is shown in Figure 1. Our research filtered 152,457 patients with malignant lung tumor. It was requisite that the patient satisfy the following inclusion criteria: (a) age ≥18 years; (b) the number of lymph nodes dissected during the operation should be clearly recorded; (c) no distant organ metastases or N3 metastases; and (d) clear staging information was available, and each patient was commended with stage I–III diseases (International Association for the Study of Lung Cancer, 8th edition). Finally, we included 35,094 patients for the analyses.

The process of patient enrollment and the ultimate outcomes of enrolled patients.
Variables
Data on general demographic characteristics (Table 1), including ethnicity, sex, age, marital status, year of disease diagnosis, and survival months were extracted. Information about tumor characteristics such as grade, stage, location, laterality, and histology as well as cause of death was included. In addition, information about treatment regimens for lung cancer was also included, which comprised preoperative radiotherapy, preoperative systematic treatment, surgery, lymph node dissection, postoperative radiotherapy, and postoperative systematic treatment.
Baseline information of the study cohort.

NSCLC: non–small cell lung cancer; SCLC: small cell lung cancer.
8th classification: based on the International Association for the Study of Lung Cancer [IASLC] staging system.
Statistical analyses
In this research, we performed all statistical analyses using R (version 4.4.1), RStudio (version 2024.09.1 + 394), and GraphPad prism (version 9.5). Initially, we employed the chi-square test to examine if there were any disparities in the mortality rates categorized by cause of death at the 3rd, 7th, 11th, and 15th month after surgery. Continuous variables are presented as median with interquartile range values. Logistic regression analysis was performed using the “autoReg” package; we estimated the relationship between death due to heart diseases within 3 months and other affecting factors. We incorporated general demographic characteristics, tumor characteristics, and treatment progress into the univariable logistic analysis and screened out factors with a significant (p < 0.05) influence on the 3-month mortality rate; these factors were then used in the multivariable logistic regression model. Odds ratios with 95% confidence intervals of all the research factors were compared. Subsequently, we conducted similar analyses for the 7-month and 11-month mortality rates associated with heart diseases. To control for confounding variables and improve the robustness of the findings, we focused on the key variables identified in the study and refined the statistical analysis of the research using the propensity score matching (PSM) method. We analyzed the risk factors for cardiac death within 3 months after lung cancer surgery. To address potential selection bias in the existing logistic regression, we used the “MatchIt” package to perform PSM with variables that may influence exposure allocation or heart disease–related death as matching covariates.
Results
Table 1 presents the traits of the research population. In total, 35,094 patients were included in the analysis. Majority of the subjects were Caucasian (83.7%, 29,390/35,094). The patients were classified into three groups based on the following age groups: ≤65, >65 to <85, and ≥85 years. Among them, 14,622 patients were aged ≤65 years, accounting for 41.7% of the total study population. Considerably more patients were diagnosed with NSCLC (98.5%, 34,568/35,094) than with small cell lung cancer (1.2%, 422/35,094). Out of all surgical approaches, the most preferred was lobectomy (77.5%, 27,207/35,094) followed by sublobectomy (17.9%, 6267/35,094), and pneumonectomy (4.6%, 1620/25,094). In addition, 2.5% (869/35,094) and 8.9% (3133/35,094) of the patients underwent preoperative radiotherapy and postoperative radiotherapy, respectively. The proportion of patients who underwent preoperative systematic treatment was 4.5% (1569/35,094) and that of those who underwent postoperative systematic treatment was 22.6% (7929/35,094).
In this cohort, 3.24% (1138) patients died within 3 months, 2.93% (1028) died at 4–7 months, 3.28% (1152) died at 8–11 months, and 3.01% (1055) died at 12–15 months (Figure 2(a) and (b)) after the surgery. The postoperative mortality rates associated with heart disease at 3, 7, 11, and 15 months were 8.96% (102/1138), 7.30% (75/1028), 5.38% (62/1152), and 5.97% (63/1055), respectively (Figure 3). The mortality rate for patients who died from heart diseases within 3 months was significantly higher compared with that for those who died at 4–7 months (p = 0.017). The median survival time of the study population was 76 months.

Postoperative mortality in the enrolled patients. (a) Overall proportion of postoperative deaths in the enrolled patients and (b) a comparison of number of deaths among the enrolled patients at various time points within 15 months of surgery using chi-square test.
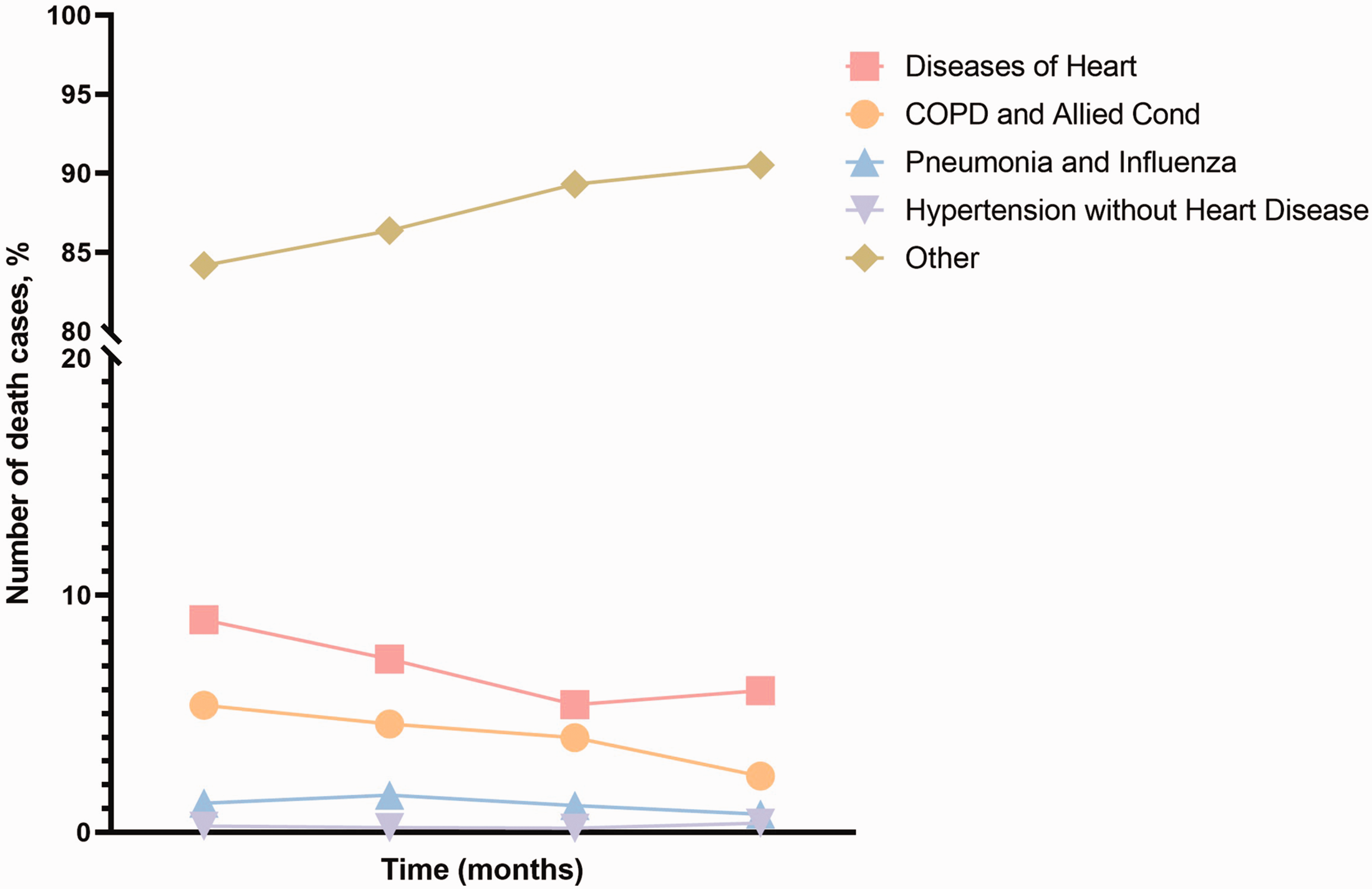
Overall proportion of postoperative deaths due to different causes within 15 months of surgery.
In the univariable logistic analysis, age, sex, radiotherapy following surgery, and systematic treatment after surgery were related to heart disease mortality in patients within 3 months postoperatively (Table 2). In addition, race and disease staging classification were also associated with mortality at 4–7 months (Table 3). Data revealed that race, age, sex, marital status, preoperative systematic treatment, postoperative systematic treatment, and grade were corelated to the mortality rate at 8–11 months (Table 4). Moreover, we performed PSM to control the confounding factors (Supplement Table 1–3). After PSM, we re-evaluated the relationship between postoperative systemic treatment and the 3-month mortality. The results demonstrated that postoperative systemic treatment remained significantly associated with a lower risk of death within 3 months of surgery.
Results of univariable and multivariable logistic regression analyses to identify risk factors for death due to heart disease within 3 months of surgery.
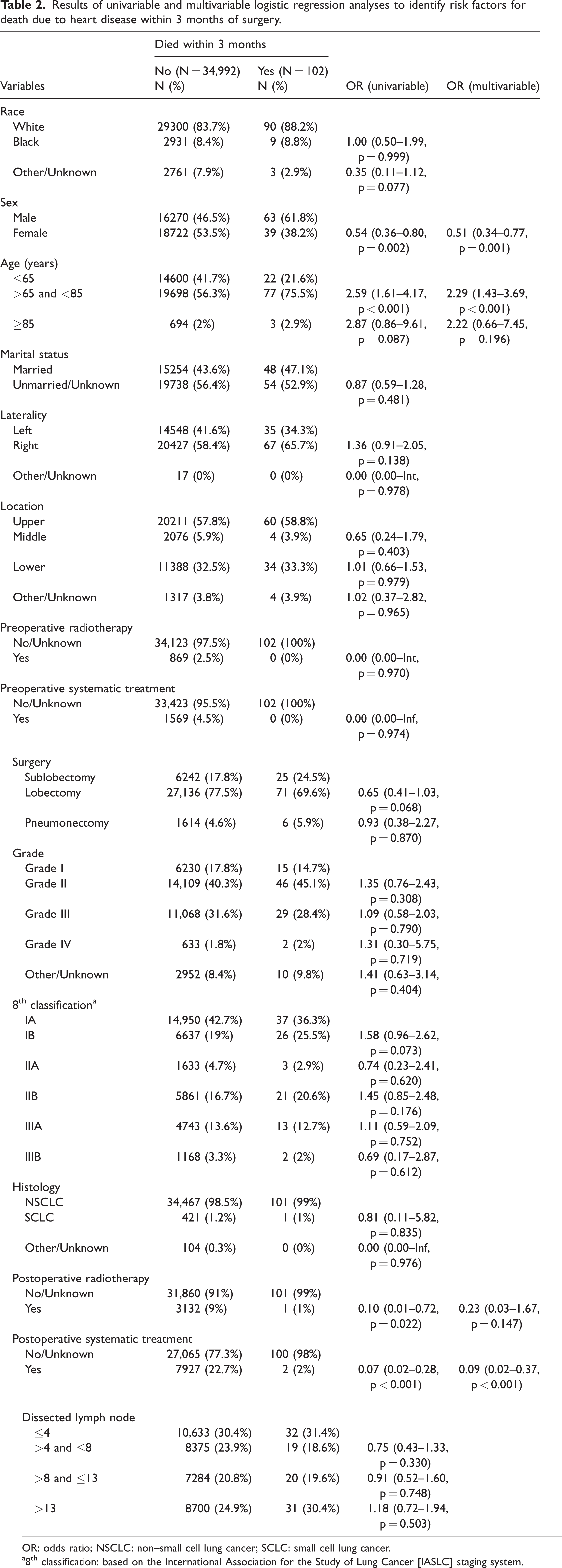
OR: odds ratio; NSCLC: non–small cell lung cancer; SCLC: small cell lung cancer.
8th classification: based on the International Association for the Study of Lung Cancer [IASLC] staging system.
Results of univariable and multivariable logistic regression analyses to identify risk factors for death due to heart disease at 4–7 months after the surgery.

OR: odds ratio; NSCLC: non–small cell lung cancer; SCLC: small cell lung cancer.
8th classification: based on the International Association for the Study of Lung Cancer [IASLC] staging system.
Results of univariable and multivariable logistic regression analyses to identify risk factors for death due to heart disease within 11 months of surgery.
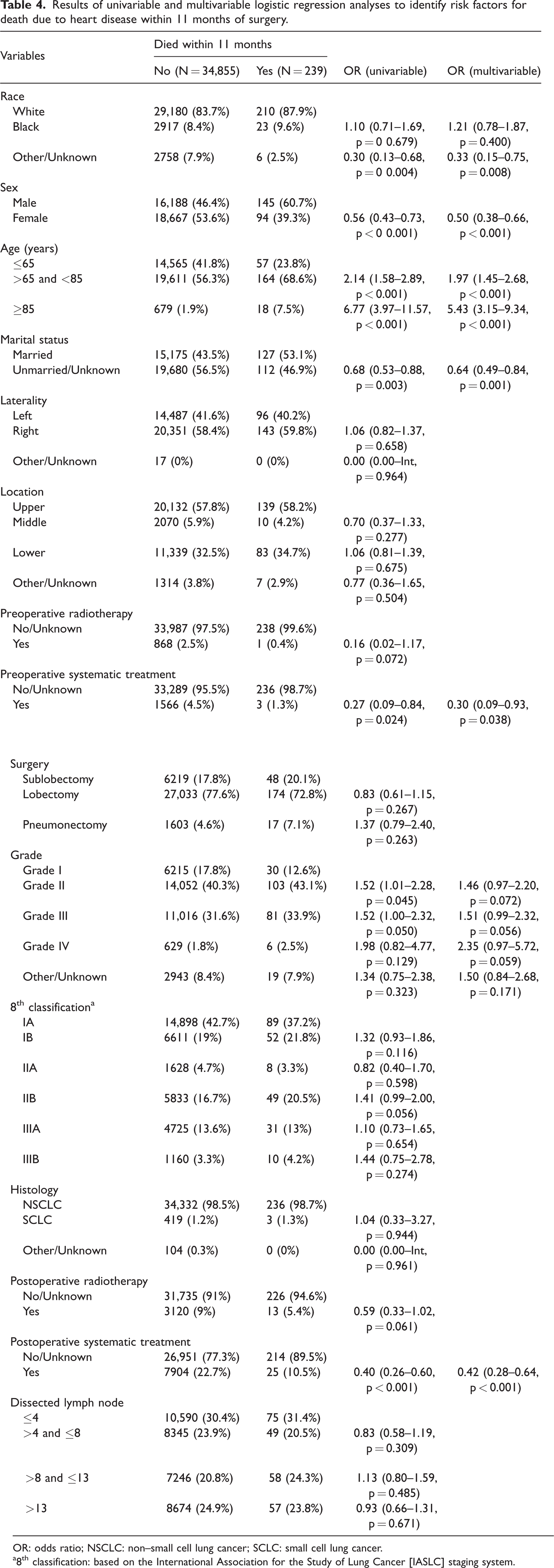
OR: odds ratio; NSCLC: non–small cell lung cancer; SCLC: small cell lung cancer.
8th classification: based on the International Association for the Study of Lung Cancer [IASLC] staging system.
Discussion
As a crucial treatment modality for lung cancer, lung cancer surgery aimed to extend patient survival. Nevertheless, in the aftermath of the operation, patients frequently encounter a multitude of risks. 20 In this study, we endeavored to explore the risk factors associated with heart disease–related mortality within 3 months of lung cancer surgery in patients. This matter holds pivotal significance for enhancing the postoperative management of patients with lung cancer and curbing the mortality rate.
We used the SEER database to access the available case-related information, establishing a robust data groundwork for our study. A retrospective research approach was employed, and 35,094 patients diagnosed with stage I–III lung cancer from 2010 to 2015 were included. To conduct an in-depth assessment of the factors influencing mortality, we employed univariable and multivariable logistic regression analysis, statistical methodologies, to systematically analyze the mortality rates of patients at 3, 7, and 11 months after surgery. The research findings revealed that the 3rd month after surgery was a time-point with a relatively high mortality rate (3.24%, 1138/35,094) among patients with lung cancer, during which the proportion of deaths due to heart diseases was particularly high (8.96%, 102/1138). Further analysis revealed that factors such as sex, age, postoperative radiotherapy, and postoperative systematic treatment were independent influencing factors for heart disease–related mortality within 3 months of surgery.
Compared with those of previous studies, the results of this study show consistency in some aspects. For example, as per a previous study, age constitutes a crucial risk factor for the occurrence of cardiovascular events subsequent to surgery. 21 We also found that age (>65 years) is an independent factor associated with mortality after lung surgery. As age increases, the cardiac function of the human body gradually degenerates. Specifically, the sensitivity of the heart to β-adrenergic receptors reduces, leading to a decline in the heart rate and ejection fraction. Meanwhile, the end-diastolic and end-systolic pressures of the ventricle rise. All these changes make elderly individuals more susceptible to arrhythmia and heart failure. 22 Moreover, pre-existing chronic diseases, such as chronic obstructive pulmonary disease, hypertension, and diabetes, can significantly increase the risk of postoperative heart disease. 23 Furthermore, patients receiving postoperative systematic treatment had a relatively lower risk of developing heart diseases within 3 months of lung surgery. As per previous research, postoperative adjuvant therapies, such as targeted therapy and immunotherapy, may reduce the occurrence of cardiac events by improving tumor control. 24 However, the specific impact of these therapies on cardiotoxicity requires further investigation. It is noteworthy that undergoing systematic treatment prior to surgery may lower the risk of mortality due to heart disease at 8–11 months after the surgery. Preoperative therapies may exert positive effects on tumor progression and the cardiovascular system despite the lack of explicit evidence. In addition, the impact of postoperative radiotherapy on the incidence of heart disease was not significant (p > 0.05). Nonetheless, Banfill et al. 25 has reported a dose-volume dependent relationship between radiotherapy dose and cardiotoxicity. Specifically, when the heart is subjected to higher radiotherapy doses, the risk of cardiac events increases significantly. This discrepancy might stem from the fact that the radiation dose absorbed by the heart in the short term fails to reach the injury threshold; alternatively, it could be due to the delayed effects of cardiac damage after radiotherapy. Heart disease–related mortality in patients at 4–7 months after surgery was associated with the tumor stage. Multivariable analysis showed that compared with that in stage IA, the risks of occurrence of events of concern was higher in stages IIB, IIIA, and IIIB. It has been reported that the incidence of postoperative heart disease is relatively high due to the increased risk of cardiovascular diseases caused by progression in tumor stage.26,27
It is important to acknowledge that retrospective cohort studies, owing to their inherent design, are subject to certain limitations that warrant careful consideration. As an observational study relying on existing data from the SEER database, this research is susceptible to selection bias, as the inclusion of patients and documentation of variables were not randomized or controlled. Observational data, while valuable for generating hypotheses, may inherently contain unmeasured or residual confounding factors. For instance, detailed information on pre-existing cardiovascular comorbidities or intraoperative hemodynamic parameters, which could influence postoperative cardiac outcomes, were not fully captured in the database. Additionally, the temporal relationships between treatments (such as postoperative radiotherapy or systemic therapy) and cardiac events cannot be definitively established, limiting causal inference. These constraints are inherent to retrospective analyses and highlight the need for future prospective, controlled studies to validate the present findings.
We must acknowledge certain limitations of our study. The potential affecting factors included in this study, such as preoperative and postoperative systematic treatment, might involve subtle but significant differences that may have contributed to disparities in the mortality rate. Moreover, we did not focus on the long-term mortality rate resulting from heart diseases and need to conduct further research on this subject.
Our findings should be interpreted with caution regarding causal inference. As this is an observational study based on retrospective data, the identified associations between factors (e.g. postoperative systematic treatment and age) and 3-month heart disease–related mortality do not establish definitive causal relationships. Rather, they highlight potential correlations that warrant further investigation. For instance, the observed reduction in heart disease–related mortality among patients receiving postoperative systematic treatment may be influenced by unmeasured confounding factors (e.g. baseline cardiac function and adherence to concurrent cardiovascular medications) not captured in the SEER database. To validate these associations and explore the underlying causal mechanisms, prospective controlled studies, preferably those with rigorous randomization of treatments and detailed monitoring of intermediate cardiac endpoints, are required.
Overall, this study furnishes crucial reference for risk assessment and clinical intervention of mortality induced by heart diseases within 3 months of lung cancer surgery. By identifying these associated factors, targeted monitoring and management strategies can be developed to explore potential reductions in postoperative mortality owing to heart disease in patients with lung cancer, pending validation in prospective studies, thereby improving their prognosis and quality of life.
Conclusions
In summary, the probability of patients with lung cancer dying from heart disease after surgery was relatively high within 0–3 months, exceeding those at 4–7 and 8–11 months postoperatively. We determined that postoperative radiotherapy and systematic treatment may be independent affecting factors affecting the heart disease–related mortality at 0–3 months. Nevertheless, the mortality rate started to rise at 12–15 months postoperatively.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251378947 - Supplemental material for Risk analysis of cardiac disease mortality within 3 months in patients with lung cancer after surgery: A real-world cohort study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251378947 for Risk analysis of cardiac disease mortality within 3 months in patients with lung cancer after surgery: A real-world cohort study by Yimin Wu, Yun Cheng, Shuang Fu, Junbo Yuan, Jue Shen and Shunv Cai in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251378947 - Supplemental material for Risk analysis of cardiac disease mortality within 3 months in patients with lung cancer after surgery: A real-world cohort study
Supplemental material, sj-pdf-2-imr-10.1177_03000605251378947 for Risk analysis of cardiac disease mortality within 3 months in patients with lung cancer after surgery: A real-world cohort study by Yimin Wu, Yun Cheng, Shuang Fu, Junbo Yuan, Jue Shen and Shunv Cai in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605251378947 - Supplemental material for Risk analysis of cardiac disease mortality within 3 months in patients with lung cancer after surgery: A real-world cohort study
Supplemental material, sj-pdf-3-imr-10.1177_03000605251378947 for Risk analysis of cardiac disease mortality within 3 months in patients with lung cancer after surgery: A real-world cohort study by Yimin Wu, Yun Cheng, Shuang Fu, Junbo Yuan, Jue Shen and Shunv Cai in Journal of International Medical Research
Footnotes
Acknowledgments
All the participating authors are thankful for the SEER database that provided very useful data.
Author contributions
Jue Shen and Shunv Cai contributed to the study conception and design. Yun Cheng and Yimin Wu collected and analyzed the data as well as wrote the manuscript drafts. Shuang Fu, Junbo Yuan, and Shunv Cai contributed to data interpretation and manuscript review. Shunv Cai revised the manuscript. All authors provided final approval for the manuscript.
Consent to participate
Not applicable.
Data availability statement
Declaration of conflicting interests
The authors declare no competing interests.
Human ethics and consent to participate declarations
The human data in the manuscript were handled in accordance with the Declaration of Helsinki, with the need for informed consent waived for this retrospective study.
Funding
This work was supported by Zhejiang Provincial Medical Health Science and Technology Project (No. 2021KY103 and No. 2024KY845). The design of the study; data collection, analysis, and interpretation; and manuscript writing were not influenced by the funding bodies in any way.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
