Abstract
Objective
The sodium–glucose cotransporter-2 inhibitor dapagliflozin reduces proteinuria and slows the decline in estimated glomerular filtration rate in patients with chronic kidney disease. Dapagliflozin is reported to be effective in patients with immunoglobulin A nephropathy; however, there have been only a few real-world clinical trials involving patients with immunoglobulin A nephropathy. Therefore, we aimed to evaluate the effect of dapagliflozin in Japanese patients with immunoglobulin A nephropathy.
Method
We performed a multicenter, nonblinded, single-arm, prospective observational study involving 44 patients with immunoglobulin A nephropathy. Patients who had completed corticosteroid treatment and were currently undergoing renin–angiotensin system inhibitor therapy were primarily enrolled.
Results
At baseline, the mean estimated glomerular filtration rate was 56.2 ± 29.4 mL/min/1.73 m2, and the median urinary protein-to-creatinine ratio was 0.63 g/g (interquartile range: 0.32–1.22 g/g). The estimated glomerular filtration rate decreased significantly after 1 month and plateaued thereafter. The urinary protein-to-creatinine ratio significantly decreased in patients with higher proteinuria (>0.5 g/g), but not in those with less proteinuria (<0.5 g/g). The post-treatment estimated glomerular filtration rate slope was significantly attenuated compared with the pretreatment slope, especially in patients with a relatively rapid decline in estimated glomerular filtration rate.
Conclusion
Dapagliflozin reduces residual proteinuria, even after treatment with corticosteroids and renin–angiotensin system inhibitors, in patients with biopsy-confirmed immunoglobulin A nephropathy. These findings may guide future treatment strategies for immunoglobulin A nephropathy.
Keywords
Introduction
Immunoglobulin A (IgA) nephropathy is the most common type of biopsy-confirmed primary glomerular diseases worldwide. 1 Racial and ethnic variations in the prevalence of IgA nephropathy have been identified; for example, there is a significantly higher prevalence of IgA nephropathy in east and southeast Asia than in Europe or North America.2,3 The Kidney Disease Improving Global Outcomes (KDIGO) guidelines, published in 2021, 4 emphasize the importance of blood pressure control and the administration of the maximum tolerated dose of a renin–angiotensin system (RAS) inhibitor for the treatment of IgA nephropathy. The guidelines also stress the need to evaluate the individual risks and benefits associated with corticosteroid therapy, although the specific treatment options differ regionally. 5
Multiple studies in Japan have shown improvements in renal survival as well as higher rates of partial or complete remission of hematuria and proteinuria following tonsillectomy combined with pulsed corticosteroid therapy.6–8 Urinalysis performed during annual check-ups in Japan facilitates the early diagnosis of IgA nephropathy, which permits timely intervention. In such instances, the administration of immunosuppressive agents to patients with early-stage disease can result in clinical remission. 9 However, proteinuria can persist in some patients, despite treatment with RAS inhibitors.
The DAPA-CKD trial demonstrated that the sodium–glucose cotransporter-2 (SGLT2) inhibitor dapagliflozin (DAPA) reduces the relative risk of kidney failure in a broad range of patients with chronic kidney disease (CKD), including those with and without type 2 diabetes, when used in conjunction with an RAS inhibitor. 10 In addition, a subanalysis of DAPA-CKD trial data revealed a substantial effect of DAPA in patients with IgA nephropathy. Over half of the patients contributing data to this subanalysis were Asian, but the effects of DAPA may vary by race and nationality. Furthermore, it was unclear whether the patients were receiving concurrent immunosuppressive treatment. 11
In the present study, we aimed to evaluate the effects of DAPA on proteinuria and the slope of estimated glomerular filtration rate (eGFR) in patients with IgA nephropathy and residual proteinuria despite prior treatment with corticosteroids and RAS inhibitors.
Methods
Study design
We performed a multicenter, nonblinded, single-arm, prospective observational study. Patients with biopsy-confirmed IgA nephropathy who were regularly attending clinics at Juntendo University Hospital (Tokyo, Japan) or Juntendo Urayasu Hospital (Chiba, Japan) were selectively enrolled. We obtained signed consent from all participants. The study was approved by the Ethics Committee of Juntendo University Hospital (approval number: E21-0177) and was conducted in accordance with the tenets of the Helsinki Declaration of 1975, as revised in 2024. Details of all participants were deidentified.
Patients with persistent proteinuria despite prior treatment, including corticosteroids and/or RAS inhibitors, were recruited. Patients were not administered corticosteroid or RAS inhibitor treatment if this was deemed to be clinically inappropriate by their physician. Patients who did not have significant proteinuria (<0.15 g/g) were excluded from the study.
The eGFRs of patients were calculated using the following equation, which is specific to the Japanese population: eGFR (mL/min/1.73 m2) = 194 × (age (years))−0.287 ×(serum creatinine (mg/dL))−1.904 × 0.739 (for women). 12 The urinary protein-to-creatinine ratio (UPCR) was calculated from measurements recorded using spot urine samples. Spot urine samples were collected at each outpatient visit, and the timing of collection was consistent for each participant to reduce variability due to physical activity or diurnal fluctuations. Responders were defined as patients who showed a >30% reduction in UPCR from baseline after 1 month of treatment, whereas nonresponders were defined as those whose UPCR did not decrease by at least this amount. This difference in eGFR between the initiation of DAPA treatment and 1 month later was termed the “initial decline.”
In addition, eGFR data obtained from at least seven examinations over the year preceding treatment and three examinations performed 1, 6, and 12 months following the initiation of treatment were used to calculate the pre- and post-treatment slopes of the eGFR.
This study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 13 Adverse events were defined as any occurrences leading to the discontinuation of DAPA, including volume depletion, sustained decline in eGFR of ≥30%, major hypoglycemia, bone fractures, amputations, and suspected diabetic ketoacidosis.
Statistical analysis
The results were expressed as the mean ± standard deviation or median (interquartile range (IQR)), as appropriate. Datasets were compared using Student’s t-test, Mann–Whitney U test, or repeated-measures analysis of variance, depending on the distribution and nature of the data. Linear regression analysis using the least squares method was employed to calculate the annual rate of change in eGFR before and after the initiation of SGLT2 inhibitor treatment. Statistical analyses were conducted using JMP Pro v.16.0.0 (SAS, Cary, NC, USA) and GraphPad Prism v. 9.2.0 (GraphPad, San Diego, CA, USA). p-values <0.05 were considered to indicate statistical significance.
Results
Characteristics of the patients
A total of 50 patients were initially enrolled in the study. Of these, 4 were transferred to other hospitals and 2 did not have proteinuria, leaving 44 patients eligible for inclusion in the analysis. A flowchart demonstrating the inclusion and exclusion of patients is presented in Figure 1. The median duration from the initiation of DAPA treatment to the first collection of blood and urine samples was 35 days (IQR: 28–56 days). The baseline clinical characteristics of the patients are summarized in Table 1.

Flowchart of the study design.
Characteristics of the study patients.

BP: blood pressure; eGFR: estimated glomerular filtration rate; RAS: renin–angiotensin system; UPCR: urinary protein-to-creatinine ratio.
The mean age of the participants was 46.7 ± 13.1 years. Overall, 87% of the participants had a history of hypertension, and 9% had diabetes. Furthermore, 82% were prescribed RAS inhibitors, and 18% were not, because of low blood pressure or adverse effects associated with these drugs during the course of treatment. In addition, 96% of the participants had received corticosteroid therapy, and 4% had not, because of severe impairment in renal function or comorbid diabetes. In total, 34% of the participants were taking statins in combination with DAPA. The mean eGFR was 56.2 ± 29.4 mL/min/1.73 m2, and the median UPCR was 0.63 g/g (IQR: 0.32–1.22 g/g). No adverse events were observed in any participants during the study period.
Change in eGFR during treatment with DAPA
Compared with baseline values, the eGFRs of the patients decreased significantly after 1 month of DAPA treatment, which plateaued thereafter (Figure 2(a)). Thirty-two percent of the patients experienced an initial decline in eGFR of >10% (Figure 2(b)). Among those with a baseline UPCR >0.5 g/g, the proportion (32%) of patients who experienced an initial decrease in eGFR of >10% was similar to that among the entire sample. No interaction was observed between the time course of the change in eGFR and the presence of hematuria (p = 0.92; Figure S1).

Effect of dapagliflozin on the changes in eGFR, including the initial decline in eGFR. (a) Changes in eGFR before and after treatment with dapagliflozin (n = 44). p-values were calculated using repeated-measures analysis of variance and (b) Proportions of the entire sample (n = 44) and patients with UPCR >0.5 g/g (n = 25) at baseline showing a decline in eGFR over the first month of treatment. **p < 0.01, ***p < 0.001. eGFR: estimated glomerular filtration rate; UPCR: urinary protein-to-creatinine ratio.
Change in UPCR during treatment with DAPA
Individual UPCRs in response to DAPA treatment varied substantially. The mean UPCR decreased between baseline and both 1 and 6 months of treatment (Figure 3(a)), and 41% of the patients experienced a reduction in UPCR of >30%.
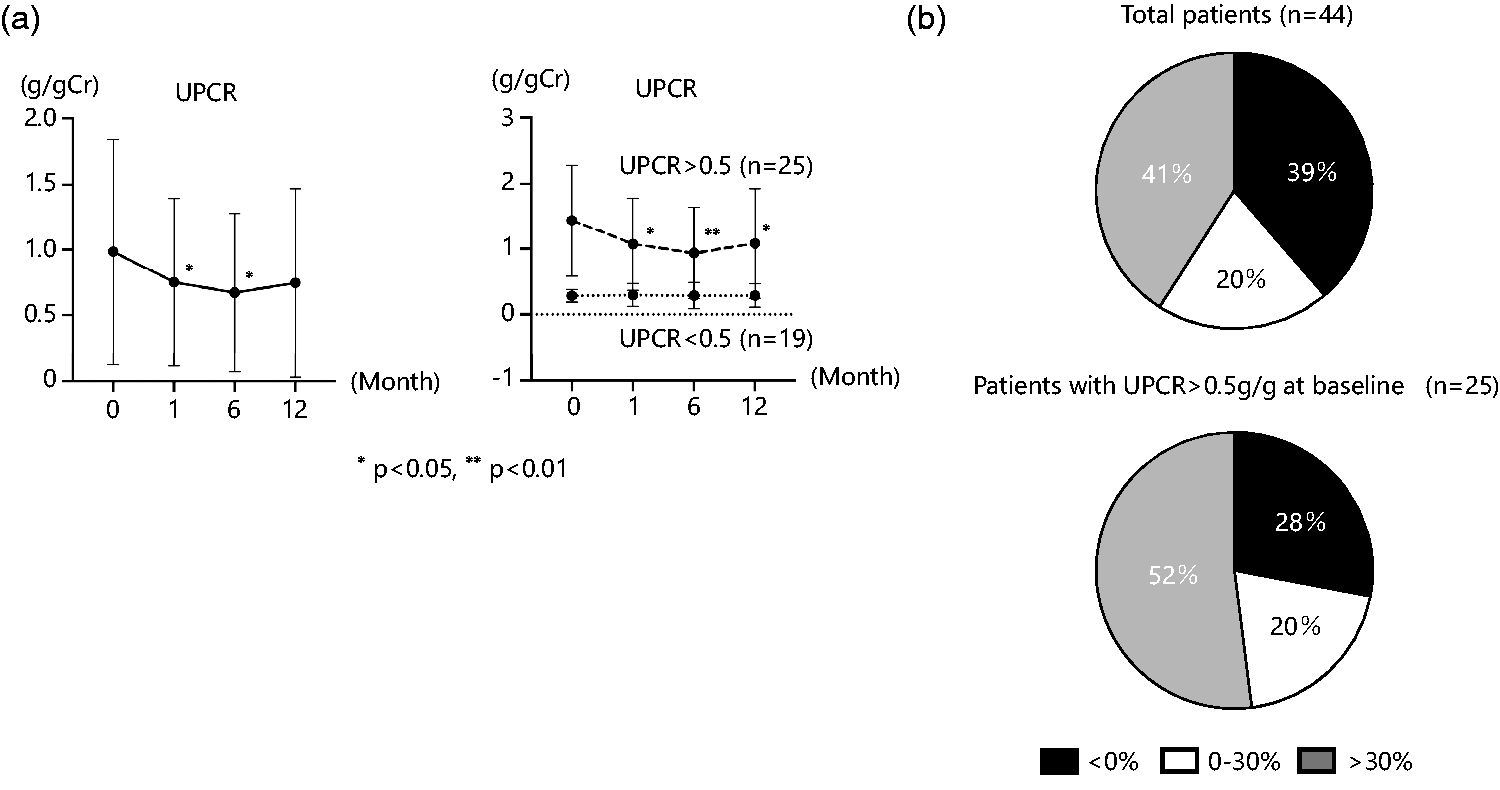
Effect of dapagliflozin on the change in UPCR. (a) Changes in eGFR before and after treatment with dapagliflozin in the entire sample and in patients with UPCR > 0.5 or <0.5 g/g at baseline and (b) proportions of patients showing a reduction in UPCR during the first month of DAPA treatment in the entire sample and in patients with UPCR >0.5 g/g at baseline. *p < 0.05, **p < 0.01. p-values were calculated using repeated-measures analysis of variance. eGFR: estimated glomerular filtration rate; UPCR: urinary protein-to-creatinine ratio; DAPA: dapagliflozin.
When stratified by baseline UPCR, no change was observed in patients with UPCR <0.5 g/g (n = 19). Conversely, patients with baseline UPCR >0.5 g/g showed significant decreases during treatment, and a higher proportion of patients in this group achieved >30% reduction in UPCR than that in the entire sample (52% vs. 41%, respectively) (Figure 3(b)). There was no interaction between the time course of the change in UPCR and the presence of hematuria (p = 0.81; Figure S1).
Clinical characteristics of UPCR responders and nonresponders who had UPCR >0.5 g/g at baseline
Among patients with a baseline UPCR >0.5 g/g, 13 (52.0%) showed a reduction in UPCR during DAPA treatment, and 12 (48.0%) did not show a significant response (Table 2). The UPCR responders were older than the nonresponders (52.8 ± 8.0 vs. 41 ± 12 years, p = 0.01).
Baseline characteristics of responders and nonresponders among patients with baseline UPCR >0.5 g/g.

BP: blood pressure; eGFR: estimated glomerular filtration rate; RAS: renin–angiotensin system; UPCR: urinary protein-to-creatinine ratio.
Relationship between the initial decline in eGFR and UPCR and the change in the slope of eGFR
The percentage change in eGFR during the first month of DAPA treatment was associated with the percentage change in UPCR (Figure 4). Although the post-treatment eGFR slope (after 1 month) tended to be less steep than the pretreatment slope in the overall population, this difference was not statistically significant (pretreatment: −3.23 (−6.37, +1.25) vs. post-treatment: −0.96 (−2.91, +2.70) mL/min/1.73 m2; p = 0.07).

Correlation between the initial percentage changes in eGFR and UPCR. Scatter plots showing a positive correlation between the percentage changes in eGFR and UPCR during the first month of treatment. p-values were calculated using Spearman’s rank correlation coefficient analysis. eGFR: estimated glomerular filtration rate; UPCR: urinary protein-to-creatinine ratio.
However, in a subgroup of patients with a relatively rapid decline in eGFR before DAPA initiation (defined as ΔeGFR <−1.0 mL/min/1.73 m2/year, n = 22), the post-treatment eGFR slope was significantly attenuated compared with the pretreatment slope (p < 0.01; Figure 5), which suggests a renoprotective effect of DAPA in patients with progressive eGFR decline.

Slopes of eGFR before and after dapagliflozin treatment in the entire sample and in patients who experienced relatively a rapid decline in eGFR before treatment. **p < 0.01. p-values were calculated using Student’s t-test (unpaired). eGFR: estimated glomerular filtration rate.
Changes in hemoglobin and uric acid concentrations during treatment with DAPA
Given that SGLT2 inhibitors have been reported to exert pleiotropic effects, including increases in hemoglobin and reductions in uric acid concentrations, we next evaluated these parameters during DAPA treatment. The hemoglobin concentrations of the patients increased during treatment, while those of uric acid decreased; the concentrations of both then plateaued (Figure 6).

Effects of dapagliflozin on the changes in hemoglobin and uric acid concentrations. **p < 0.01, ***p < 0.001. p-values were calculated using repeated-measures analysis of variance.
Discussion
In the present study, we demonstrated that treatment with DAPA significantly reduces the UPCR of patients with IgA nephropathy who have a baseline UPCR >0.5 g/g, whereas it has no significant effect on the UPCR of patients with IgA nephropathy who have a UPCR <0.5 g/g. UPCR responders exhibited a larger initial decline in eGFR. In patients who were experiencing a relatively rapid decline in eGFR, DAPA treatment slowed this decline. These results suggest that DAPA may help prevent renal function decline in high-risk patients with IgA nephropathy, particularly those with modest levels of proteinuria or a relatively rapid decline in eGFR.
In the previous subanalysis of DAPA-CKD trial data that focused on patients with IgA nephropathy, key details such as their history of immunosuppressive treatment and hematuria status were not provided. 11 In contrast, the present findings support the fact that DAPA exerts beneficial effects in patients who have previously received steroid-based immunosuppressive therapy. In addition, the present findings indicate that DAPA induces an initial decline in eGFR, ameliorates anemia, and reduces the uric acid concentrations of patients with IgA nephropathy, consistent with the outcomes of CKD of other etiologies.
Our finding that DAPA reduces proteinuria in patients with UPCR ≥0.5 g/g is consistent with the growing evidence that even modest levels of proteinuria are associated with significant long-term risk. In a recent large-scale cohort study, Pitcher et al. 14 reported that approximately 30% of patients with time-averaged proteinuria between 0.44 and <0.88 g/g (i.e. <1 g/day) and 20% of those with proteinuria <0.44 g/g progressed to kidney failure within 10 years. These results emphasize the importance of achieving significant proteinuria reduction, ideally below 0.5 g/g, to improve long-term renal outcomes.
To the best of our knowledge, this is the first study to demonstrate the effects of SGLT2 inhibitors in Japanese patients with IgA nephropathy. A previous study performed in Chinese patients with this condition demonstrated a significant reduction in proteinuria 6 months after the start of SGLT2 inhibitor administration. 15 In contrast, the patients in the present cohort had previously received steroid treatment, but both studies showed a significant reduction in proteinuria during the administration of an SGLT2 inhibitor.
SGLT2 inhibitors reduce glomerular hyperfiltration through mechanisms distinct from those of RAS inhibitors.16–18 A key mechanism in the progression of IgA nephropathy is the stress induced on the remaining nonsclerotic glomeruli. Consistent with previous studies on SGLT2 inhibitors in CKD, the present findings indicate a positive association between the initial decline in eGFR and the subsequent reduction in proteinuria during DAPA treatment. 19 This suggests that DAPA may help mitigate glomerular hyperfiltration in patients with IgA nephropathy.
The baseline eGFR of the patients in the present study (46.5 mL/min/1.73 m2) was comparable to that of the patients with IgA nephropathy who contributed data to the subanalysis of the DAPA-CKD trial (43.8 mL/min/1.73 m2). Although there was an initial acute decline in eGFR of >10% in 49.4% of patients with CKD in the DAPA-CKD trial, 20 we identified a lower prevalence (37.0%). This difference may be attributable to variations in the timing of initial eGFR assessment (2 weeks in the DAPA-CKD trial vs. 5 weeks in our study) or differing CKD etiologies. Our previous cohort study on the effects of DAPA in patients with CKD also showed a lower prevalence of acute eGFR decline, owing to differences in the timing of assessments. 19
In the CANVAS study, mediation analysis showed that renoprotective effects were linked to a decrease in the urinary albumin-to-creatinine ratio, amelioration of anemia, and a reduction in uric acid concentration. 21 The similar effects identified in the present study imply that DAPA may also be beneficial for patients with IgA nephropathy. Although it remains unclear whether the identified improvements in UPCR and hemoglobin and uric acid concentrations are specific to patients with IgA nephropathy, the effects of DAPA were consistent, regardless of the presence of hematuria, suggesting that it may have common renoprotective effects in patients with various CKD etiologies.
The present study had several limitations. First, the study lacked a control arm, which limits the ability to draw causal inferences from the results. Although a randomized controlled trial would provide stronger evidence, such a design was not feasible in this real-world observational setting because of ethical and practical constraints. Second, the follow-up period was relatively short, and the sample size was modest, which may have limited the statistical power of our findings and their generalizability. Third, although our initial inclusion criteria specified patients on RAS inhibitors, some patients—particularly nonresponders—were not receiving them at baseline at the discretion of their nephrologist. This heterogeneity in pre-trial standard therapies may have affected treatment responses. Fourth, multivariable adjustments were not performed for key potential confounders such as changes in blood pressure, concomitant medications, and renal histological severity. Consequently, residual confounding cannot be excluded. Despite these limitations, the study provides valuable prospective, real-world evidence supporting the potential utility of SGLT2 inhibitors in Japanese patients with IgA nephropathy who are already receiving standard therapy.
In conclusion, RAS inhibitors have been the only therapeutic option for patients with IgA nephropathy and residual proteinuria following steroid therapy. However, DAPA currently represents a potential new treatment option.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251376895 - Supplemental material for Effect of dapagliflozin on proteinuria and the slope of the estimated glomerular filtration rate in patients with immunoglobulin A nephropathy: A multicenter prospective study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251376895 for Effect of dapagliflozin on proteinuria and the slope of the estimated glomerular filtration rate in patients with immunoglobulin A nephropathy: A multicenter prospective study by Takeo Koshida, Hitoshi Suzuki, Maki Murkoashi, Hisatsugu Takahara, Yusuke Suzuki and Tomohito Gohda in Journal of International Medical Research
Footnotes
Author contributions
TK and TG were responsible for study conception, design, and protocol drafting. TK, HS, MM, and TG were responsible for data acquisition and analysis. TK, HS, MM, and TG interpreted the results of the analyses. Each author contributed important intellectual content in drafting the manuscript and approved the accuracy and integrity of the final version of the manuscript.
Data availability statement
The data will be made freely available upon request to the corresponding author.
Declaration of conflicts of interest
TG received honoraria from AstraZeneca and Ono Pharmaceutical. Other authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
There was no funding provided for the conduction or publication of this research.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
