Abstract
Introduction
Smoking is a major contributor to the development and progression of chronic respiratory diseases. Although cessation is known to improve general health, its direct impact on pulmonary function remains debated. This meta-analysis aimed to evaluate the effects of various smoking cessation interventions on lung function outcomes in patients with chronic respiratory diseases.
Methods
A systematic search of PubMed, Scopus, and Web of Science was conducted to identify eligible clinical trials. Standardized mean differences with 95% confidence intervals were calculated to compare changes in forced expiratory volume in one second (FEV1), forced vital capacity (FVC), and the FEV1/FVC ratio.
Results
Fourteen studies were included in the study. Smoking cessation was associated with a marginal improvement in FEV1 (standardized mean difference = 0.23; 95% confidence interval: −0.00 to 0.47; P = 0.054). However, no significant effects were observed on FVC (standardized mean difference = 0.08; 95% confidence interval: −0.17 to 0.23; P = 0.53) or the FEV1/FVC ratio (standardized mean difference = 2.26; 95% confidence interval: −2.25 to 6.78; P = 0.32). Subgroup analyses indicated that participant age may influence the heterogeneity of FEV1 outcomes.
Conclusion
Smoking cessation may be associated with small, nonsignificant changes in FEV1 and shows no significant short-term effects on FVC or the FEV1/FVC ratio. Although these results highlight the modest short-term impact of cessation on lung function, particularly in patients with established disease, they do not negate the substantial long-term clinical benefits of smoking cessation, including reductions in exacerbations, disease progression, and mortality.
Introduction
Chronic obstructive pulmonary disease (COPD) is a major global health concern and a leading contributor to morbidity and mortality. According to the Global Burden of Disease Study, COPD is the third leading cause of death worldwide, emphasizing the critical need for effective prevention and management strategies. 1 Smoking is a key contributor to the development and progression of COPD, with substantial evidence linking tobacco use to the disease’s underlying pathophysiology. Smoking cessation is widely recognized as the most effective intervention for slowing disease progression and preserving lung function in affected individuals. 2 Various smoking cessation methods are available, including face-to-face and telephone counseling, behavioral training programs, and pharmacological interventions such as nicotine replacement therapy (NRT) and varenicline. These approaches have demonstrated effectiveness across diverse populations attempting to quit smoking.3,4 Counseling offers psychological support, varenicline targets nicotinic receptors to alleviate cravings and withdrawal symptoms, and NRT delivers a controlled dose of nicotine to facilitate the cessation process. 5
Lung function is commonly assessed using spirometry, with key parameters including forced expiratory volume in one second (FEV1), forced vital capacity (FVC), and the FEV1/FVC ratio. These indices are clinically significant as they quantify the degree of airflow obstruction, with reductions indicative of disease progression. Moreover, they play a critical role in distinguishing between obstructive and restrictive pulmonary disorders. 6
In addition to COPD, smoking is a major modifiable risk factor for other chronic respiratory conditions. In asthma, tobacco smoke exposure contributes to increased airway hyperresponsiveness, persistent inflammation, and reduced responsiveness to inhaled corticosteroids. Clinical studies have shown that smoking cessation in asthma patients can lead to improvements in FEV1, symptom control, and quality of life, with some benefits apparent within weeks.7,8 In lung cancer, ongoing smoking after diagnosis is associated with poorer treatment response, increased risk of recurrence, and higher mortality. Evidence suggests that cessation at or after diagnosis can improve survival outcomes; enhance the effectiveness of therapies such as surgery, chemotherapy, and targeted agents; and reduce treatment-related complications. 9 These benefits underscore the importance of smoking cessation interventions across the full spectrum of chronic respiratory diseases, not solely in COPD.
A comprehensive literature review revealed a spectrum of outcomes regarding the efficacy of smoking cessation interventions on lung function in patients with COPD. 10 Studies consistently show that quitting smoking can lead to improvements in FEV1 decline rates, even among individuals with established COPD, suggesting a potential for lung function recovery postcessation.11–13 Both varenicline and NRT are effective pharmacological aids for smoking cessation and may contribute to improvements in lung function among patients with COPD. However, despite the well-established benefits of quitting smoking, the direct effects of specific cessation strategies on spirometric indices remain a subject of ongoing research. 14 Furthermore, various clinical trial studies have been conducted in this field, with varying results. Some studies have shown the significant effect of smoking cessation methods on improving the lung function,8,15,16 but several other studies have not reported significant results.17–25 This variability may stem from differences in study populations, intervention protocols, and follow-up durations. To address these inconsistencies, this systematic review and meta-analysis aimed to synthesize the available evidence on the effects of various smoking cessation strategies on lung function, specifically focusing on FEV1, FVC, and the FEV1/FVC ratio. By clarifying the extent to which cessation influences these key respiratory outcomes, this study sought to inform and optimize COPD management strategies and improve patient quality of life.
Methods
This investigation was conducted and recorded per the guiding tenets of the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA). 26
Search strategy
The search was conducted in PubMed, Scopus, and Web of Science databases until January 2025 using the following keywords: (“Smoking cessation”[Title/Abstract] OR “stopping Smoking”[Title/Abstract] OR “Quitting Smoking”[Title/Abstract] OR “smoking abstains”[Title/Abstract] OR “vape cessation”[Title/Abstract] OR “vape stopping”[Title/Abstract] OR “vape quitting” [Title/Abstract] OR “vape abstain”[Title/Abstract] OR “electronic cigarettes cessation”[Title/Abstract] OR “electronic cigarettes stopping”[Title/Abstract] OR “electronic cigarettes quitting”[Title/Abstract] OR “electronic cigarettes abstain”[Title/Abstract] OR “Giving Up Smoking”[Title/Abstract]) AND (“Chronic respiratory disease”[Title/Abstract] OR “asthma”[Title/Abstract] OR “chronic obstructive pulmonary disorder”[Title/Abstract] OR COPD[Title/Abstract] OR “cystic fibrosis”[Title/Abstract] OR “lung cancer”[Title/Abstract] OR “sleep apnea”[Title/Abstract].
Study selection and inclusion and exclusion criteria
The PICO (Patient/Population, Intervention, Comparison, and Outcome) framework for this meta-analysis was defined as follows—population (P): patients with lung-related diseases; intervention (I): various smoking cessation methods; comparison (C): control group; outcome (O): spirometric indices including FEV1, FVC, and the FEV1/FVC ratio.
The inclusion criteria were as follows: (a) clinical trials; (b) studies assessing the impact of smoking cessation interventions on spirometry outcomes; (c) publications in English; (d) studies involving adults aged ≥18 years; and (e) studies reporting sufficient data on baseline and postinterventional spirometry values for both intervention and control groups.
The exclusion criteria included the following: (a) in vitro or animal studies, duplicate publications, reviews, letters, conference abstracts, and case reports; (b) observational studies; and (c) studies involving co-supplementation with other agents.
Data extraction
Studies that met the inclusion criteria were subsequently reviewed independently by two reviewers. The following data were extracted: (a) author’s name; (b) date of publication and the country of origin where the study was conducted; (c) the number of participants in both the intervention and control groups; (d) type of intervention; (e) duration of supplementation; (f) age, sex, and body mass index (BMI) of the study participants; and (g) baseline concentrations of the variables studied.
Methodological quality
The Cochrane Collaboration Risk of Bias tool was used to systematically assess the risk of bias in the included studies. 27 Two reviewers independently evaluated the quality of the studies based on the following criteria: (a) adequacy of sequence generation; (b) allocation concealment; (c) blinding; (d) handling of dropouts; (e) selective outcome reporting; and (f) other potential sources of bias. Following the guidelines provided in the Cochrane Handbook, the studies were classified into three categories: unclear risk, high risk of bias, and low risk of bias.
Statistical analysis
A random-effects model was employed to perform statistical analysis using the restricted maximum likelihood method. Between-study heterogeneity was assessed using the I2 index and I2 > 50% was considered as high heterogeneity. Effect size was estimated for each variable using the changes in before and after intervention values in both groups. All data were inserted as means ± SD. In studies where data were reported as median, range, and sample size (n), the sample mean (x̄) was estimated using the method proposed by Luo et al. 28 : X− ≈ (4/(4 + n0.75)) × ((a + b)/2) + (n0.75/(4 + n0.75)) × m, where a is the minimum value, b is the maximum value, and m is the median. The SD for this scenario was estimated using the method proposed by Wan et al. 29 : S ≈ (b − a)/(2Φ−1((n − 0.375)/(n + 0.25))), where Φ−1 is the inverse function of the standard normal distribution’s cumulative distribution function. For studies reporting the median and interquartile range (IQR =q3 − q1), the mean was estimated using the corresponding formula from Luo et al., 28 and the SD was estimated as S ≈ (q3 − q1)/(2Φ−1((0.75n − 0.125)/(n + 0.25))), as proposed by Wan et al. 29 In cases where neither SDs nor standard errors (SEs) were reported, the average SDs from other trials included in the meta-analysis were used for imputation. 30 The effect size was expressed as the standardized mean difference (SMD) with a 95% confidence interval (CI). Two approaches were used to identify potential sources of heterogeneity: (a) subgroup analysis and (b) meta-regression analysis. Subgroup analysis was also conducted to report effect sizes across different ranges of BMI, age, and study quality. A leave-one-out sensitivity analysis was performed to assess the robustness of the pooled FEV1 estimate. This approach involves sequentially removing one study at a time and recalculating the overall effect to identify whether any single study disproportionately affected the results. This method follows guidance from the Cochrane Handbook for Systematic Reviews of Interventions (version 6.3). 11 Studies that notably influenced statistical significance or heterogeneity were further examined for methodological limitations or sample characteristics that may explain their impact. Additionally, to assess small-study effects, Begg’s adjusted rank correlation and Egger’s regression asymmetry tests were performed.31,32 Publication bias was assessed through visual inspection of funnel plots for each variable. If publication bias was detected, a trim-and-fill analysis was conducted to simulate a model without publication bias by inserting hypothetical studies, resulting in a new effect size. A P value of <0.05 was considered to indicate statistical significance. All statistical analysis was conducted using Stata 17.0.
Qualitative synthesis of nonquantifiable studies
In addition to the quantitative meta-analysis, a qualitative synthesis was conducted for studies that met the inclusion criteria but were excluded from the meta-analysis due to insufficient statistical data (e.g. missing means, SDs, or sample sizes). These studies were reviewed narratively to summarize their findings and assess their relevance to the overall research question. Key outcomes related to FEV1, FVC, and the FEV1/FVC ratio were extracted and described, focusing on the direction and magnitude of reported effects as well as the context of the intervention. This approach provided supplementary insight into the potential impact of smoking cessation interventions that could not be evaluated through pooled statistical estimates.
Results
Study selection
Through a meticulous search process, we collected 15,942 articles from databases such as PubMed, Scopus, and Web of Science. Of these, 8638 were found to be duplicates and were thus removed. Further scrutiny of the titles and abstracts led to the removal of another 7210 articles. After a detailed review of their full texts, 80 of the remaining 94 articles were excluded for various reasons, including co-intervention (7 articles) and lack of necessary data (73 articles). Finally, our study incorporated 11 articles for meta-analysis and 3 studies for systematic review that satisfied our inclusion criteria. Although our search strategy included terms related to e-cigarette and vape cessation, none of the studies meeting these criteria satisfied our inclusion criteria. Most were observational studies or did not report relevant spirometric outcomes and were therefore excluded. The flow of information through the different phases of this systematic review and meta-analysis is depicted in the study’s PRISMA flowchart (Figure 1).

Flowchart of study selection for the inclusion of trials in the systematic review.
Study characteristics
The breakdown of the articles analyzed, organized by the type of variables investigated, are as follows: 11 articles focused on FEV1, 4 on FVC, and 2 on the FEV1/FVC ratio. This meta-analysis included 1369 participants and covered studies conducted from 2010 to 2020. The number of participants in each study ranged from 32 to 394, with their ages averaging between 31 and 69 years. The duration of interventions varied from 12 to 52 weeks. BMI data were reported for participants in eight studies; apart from two studies by Hashimoto et al. 18 and Pezzuto et al., 23 where participants had normal BMIs, the remaining studies reported that the participants were overweight. The studies were conducted across multiple locations: three in Italy,20,23,25 two in USA,21,33 two in Denmark,22,24 one in Iran, 19 one in France, 17 one in South Korea, 8 and one in Japan. 18 Three studies, conducted by Bohadana et al., 15 Chaudhuri et al., 34 and Willemse et al., 16 were excluded from the analysis because they did not have enough data. They analyzed 32–70 participants each, with ages ranging from 45 to 63 years. The information on all 14 studies is presented in Table 1.
Characteristics of the studies included in the meta-analysis.

BMI: body mass index; CG: control group; COPD: chronic obstructive pulmonary disease; IG: intervention group; FEV1: forced expiratory volume in one second; FVC: forced vital capacity; NRT: nicotine replacement therapy; NR: not reported.
Risk of bias assessment
Table 2 presents the results of the quality assessment of the included studies, which was conducted using the Cochrane Collaboration Risk of Bias tool. The assessment categorized the overall risk of bias as follows: high risk in 3 of the 14 studies,20,24,25 moderate risk in 5 studies,16,18,19,23,34 and low risk in 6 studies.8,15,17,21,22,33 The allocation concealment component achieved the highest high-risk rating. Furthermore, the incomplete outcome data module was most frequently evaluated as low risk.
Risk of bias assessment.

H: high; L: low; M: moderate.
General low risk: fewer than 2 of the 7 components rated as high risk. General moderate risk: exactly 2 of the 7 components rated as high risk. General high risk: more than 2 of the 7 components rated as high risk.
Effect of smoking cessation on FEV1
Based on the findings, the effect of smoking cessation on FEV1 was not significant (SMD = 0.23; 95% CI: −0.00 to 0.47; P = 0.054) (Figure 2(a)). Moreover, a significant between-study heterogeneity was observed (I2 = 70.00%; P < 0.001). Subgroup analyses were conducted to explore potential sources of heterogeneity in FEV1 outcomes based on age, BMI, type of intervention, study quality, and underlying disease. Although effect sizes varied across subgroups, none of the subgroup comparisons reached statistical significance. Notably, greater effect sizes were observed in studies involving younger participants, asthma patients, and those using comprehensive smoking cessation programs. However, these differences should be interpreted with caution due to wide CIs and limited sample sizes in several subgroups. Complete results are presented in Table 3. Furthermore, the results of sensitivity analysis indicated that excluding the studies conducted by Westergaard et al. 22 (SMD = 0.25; 95% CI: 0.001 to 0.50), Le Mao et al. 17 (SMD = 0.26; 95% CI: 0.006 to 0.52), Zarghami et al. 19 (SMD = 0.25; 95% CI: 0.004 to 0.52), and Jang et al. 8 (SMD = 0.11; 95% CI: 0.001 to 0.22) made the overall findings significant. No significant small-study effects were observed in Egger’s and Begg’s tests (P = 0.13 and 0.08). Moreover, a visual inspection of the funnel plot did not show the presence of publication bias (Figure 2(b)).

(a) Forest plot detailing standardized mean difference (SMD) and 95% confidence intervals (CIs) for the effect of smoking cessation on FEV1 and (b) funnel plot for the effect of smoking cessation on FEV1.
Subgroup analyses for the effects of smoking cessation on lung function outcomes.
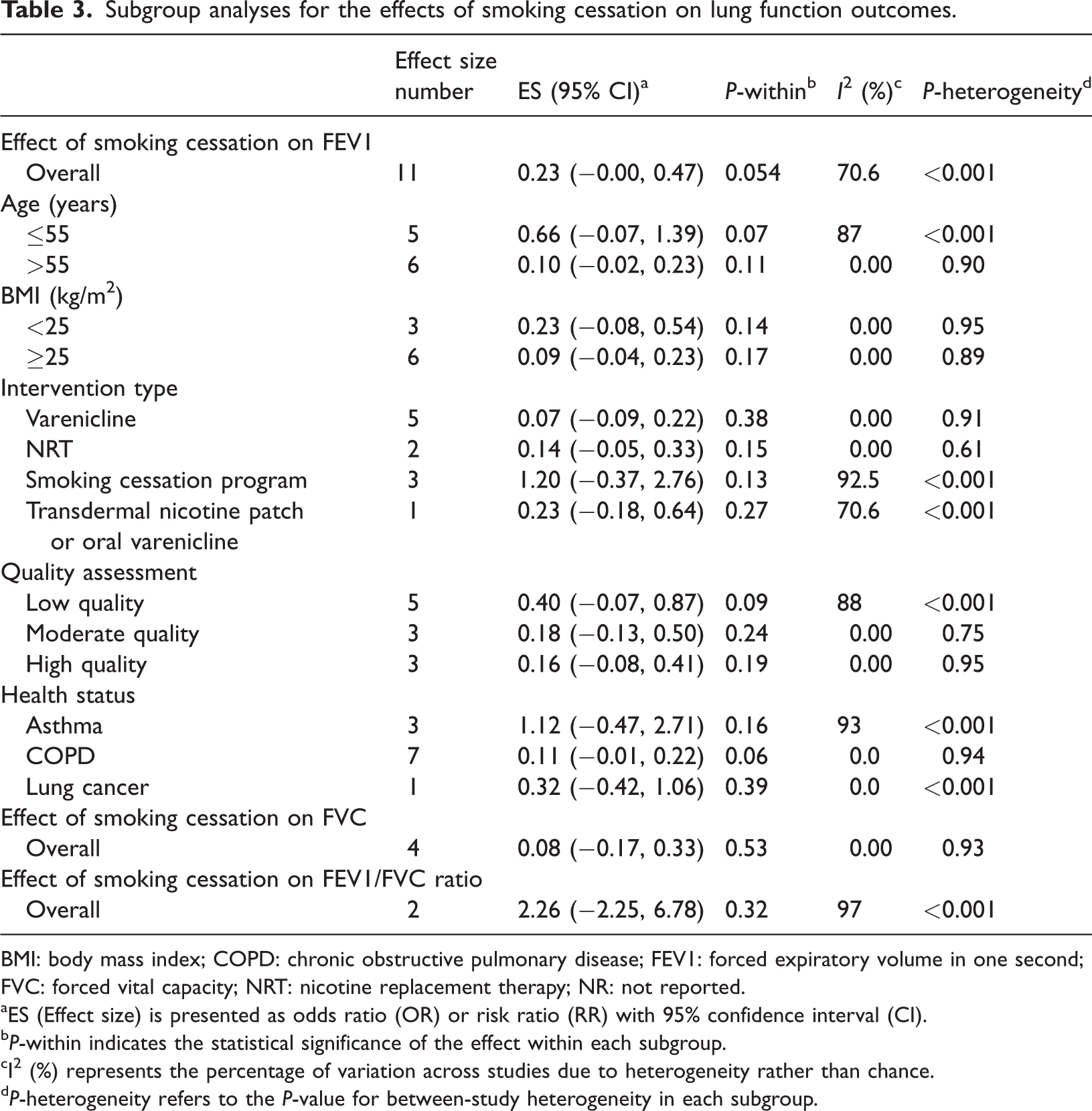
BMI: body mass index; COPD: chronic obstructive pulmonary disease; FEV1: forced expiratory volume in one second; FVC: forced vital capacity; NRT: nicotine replacement therapy; NR: not reported.
ES (Effect size) is presented as odds ratio (OR) or risk ratio (RR) with 95% confidence interval (CI).
P-within indicates the statistical significance of the effect within each subgroup.
I² (%) represents the percentage of variation across studies due to heterogeneity rather than chance.
P-heterogeneity refers to the P-value for between-study heterogeneity in each subgroup.
Effect of smoking cessation on FVC
According to the results of the analysis, smoking cessation did not show a significant effect on FVC (SMD = 0.08; 95% CI: −0.17 to 0.33; P = 0.53) (Figure 3(a)). However, the heterogeneity was not significant (I2 = 0.00%; P = 0.93). There was no significant difference between the results after performing sensitivity analysis.

(a) Forest plot detailing standardized mean difference (SMD) and 95% confidence intervals (CIs) for the effect of smoking cessation on FVC and (b) forest plot detailing SMD and 95% CIs for the effect of smoking cessation on the FEV1/FVC ratio. FEV1: forced expiratory volume in one second; FVC: forced vital capacity.
Effect of smoking cessation on the FEV1/FVC ratio
The results indicated that quitting smoking did not have a significant effect on the FEV1/FVC ratio (SMD = 2.26; 95% CI: −2.25 to 6.78; P = 0.32) (Figure 3(b)). Additionally, heterogeneity was significant (I2 = 97.0%; P < 0.001).
Narrative review of nonquantifiable studies
Bohadana et al. 15 conducted research on patients with COPD, administering varenicline supplements for 6 weeks and assessing the outcomes at week 30. The findings revealed that the overall severity of airway obstruction in the study population did not change from the beginning of the study to week 30. When the data were analyzed by group, the trend remained consistent, showing no significant differences between the groups. Furthermore, a comparison among those who abstained revealed that there were no significant changes in the values of FEV1, FVC, or the FEV1/FVC ratio from the start of the study to week 30. Conversely, Chaudhuri et al. 34 conducted research on asthma patients and revealed that a 6-week smoking cessation program significantly improved FEV1. Specifically, the average change in FEV1 among those who quit smoking was 356 mL (SD = 278) after 1 week, 390 mL (SD = 311) after 3 weeks, and 450 mL (SD = 471) after 6 weeks of quitting, with P values of 0.015, 0.009, and 0.031, respectively. Additionally, when comparing those who quit with the control group of smokers after 6 weeks, the quitters showed an average FEV1 improvement of 407 mL and a 15.2% increase in the predicted percentage of FEV1. Furthermore, a study by Willemse et al. 16 aimed to explore the impact of a year-long smoking cessation program on airway inflammation among 28 smokers with COPD and 25 asymptomatic smokers with normal lung function. The findings revealed that in the 12 patients with COPD who managed to quit smoking, airway inflammation continued to be evident in bronchial biopsies. Additionally, there was a significant increase in sputum neutrophil counts, lymphocyte counts, interleukin-8 levels, and eosinophil cationic protein levels after 12 months. Conversely, among the 16 asymptomatic smokers who quit, inflammation significantly decreased or remained unchanged.
Discussion
This meta-analysis found a small, statistically nonsignificant improvement in FEV1 following smoking cessation. Furthermore, no significant changes were observed in FVC or the FEV1/FVC ratio. These results indicate that the short-term impact of smoking cessation on spirometric parameters is limited, particularly among individuals with advanced or long-standing respiratory disease. Although several individual studies reported clinically relevant improvements—especially in younger patients or those with less severe disease—the overall pooled estimates failed to achieve statistical significance. These findings are consistent with the existing literature, which suggests that smoking cessation is more effective in slowing the progression of lung function decline rather than reversing established airflow obstruction, particularly in the earlier stages of disease.35,36 The subgroup analysis by disease type supports this distinction: patients with asthma—characterized by reversible airway obstruction—demonstrated a greater improvement in FEV1 than those with COPD or lung cancer, where airway obstruction is more likely to be chronic and irreversible. This finding aligns with previous evidence that asthma patients may benefit more rapidly from cessation due to reduced inflammation and improved airway reversibility, whereas structural changes in COPD limit short-term gains.37,38 It should be noted that subgroup analyses by intervention type were limited by the small number of available studies in each category, particularly for behavioral programs, restricting the interpretability of these comparisons. These results should therefore be viewed as exploratory rather than conclusive. Similarly, studies by Tashkin 11 and Webb et al. 39 showed that smoking cessation primarily stabilizes rather than improving FEV1 in patients with COPD, especially over short follow-up durations. These findings contradict those of the research in asthma populations, where reversibility of airway obstruction may yield more pronounced spirometric gains postcessation, as noted in a study by Chaudhuri et al. 34 Therefore, the extent and direction of changes in lung function following smoking cessation appear to be influenced not only by the act of cessation itself but also by factors such as disease type, baseline severity, and the reversibility of underlying pathological changes. The detrimental effects of smoking on pulmonary function are well-documented and include the induction of oxidative stress, persistent inflammation, and structural damage to lung tissue through proteolytic activity, all of which contribute to airflow limitation. 40 Smoking cessation initiates a cascade of physiological recovery processes. It reduces airway inflammation and oxidative stress by limiting the infiltration of inflammatory cells and decreasing the production of proinflammatory cytokines. Concurrently, it lowers oxidative damage by reducing the generation of reactive oxygen species and enhancing endogenous antioxidant defenses. These changes contribute to the restoration of airway epithelial integrity and reduce bronchial hyperresponsiveness. 41 Additionally, smoking cessation helps reestablish the balance between proteolytic enzymes and their inhibitors, thereby mitigating the progressive degradation of lung tissue and potentially slowing the development of emphysema. 42 Finally, smoking cessation facilitates the recovery of ciliary function in the respiratory epithelium, enhancing mucociliary clearance and thereby reducing the risk of respiratory infections. In terms of FEV1 outcomes, the overall effect observed in this meta-analysis was marginal and did not reach statistical significance. 43 Notably, the study by Jang et al. 8 yielded statistically significant improvements in lung function, distinguishing it from other included studies. This may be attributed to a stronger emphasis on patient adherence, facilitated through educational interventions that highlighted the impact of smoking on asthma progression and actively promoted cessation. In contrast, other studies primarily employed pharmacologic approaches such as varenicline or NRT, which may have limited effectiveness in populations with more severe underlying conditions, such as COPD or non–small cell lung cancer. In these cases, the extent of irreversible structural lung damage may diminish the potential for functional improvement, even after successful smoking cessation. 44 Hence, disease condition and commitment to the interventions appear to be crucial factors affecting the outcomes. Moreover, sensitivity analysis indicated that the overall significance increased upon excluding four studies. Notably, the stratification by disease type further clarified the observed heterogeneity. COPD-specific studies showed minimal between-study variance (I2 = 0%), while asthma-specific studies contributed disproportionately to overall heterogeneity (I2 = 93%), reinforcing the necessity of disease-specific interpretations in pooled analyses. For the studies by Zarghami et al. 19 and Westergaard et al., 24 the likely issues were low study quality and poor methodology. Regarding the studies by Westergaard et al. 22 and Le Mao et al., 17 the main concerns were small sample sizes and participants’ low adherence to the interventions. Although our subgroup analysis suggested a particularly large effect size for smoking cessation programs (SMD = 1.20), this finding must be interpreted with significant caution. The reliability of this specific result is compromised by extremely high statistical heterogeneity (I2 = 92.5%), indicating that the studies pooled within this subgroup showed substantially different effects. This inconsistency is likely attributable to two key factors. First, the subgroup is based on a very small number of studies (n = 3), making the pooled estimate statistically weak and highly susceptible to the influence of any single trial. Second, the term “smoking cessation program” is a broad categorization that encompasses various intervention measures and protocols across the individual studies. These inherent differences in program components, intensity, and delivery are a likely source of the observed heterogeneity. Therefore, although this finding is notable, it should be considered exploratory and does not provide reliable evidence that this category of intervention is superior to others. Similarly, the interventions appeared to have a greater impact in younger populations, although this trend also failed to reach significance. These findings underscore the influence of baseline physical health and demographic factors on the effectiveness of cessation strategies. Additionally, smoking cessation did not produce significant improvements in FVC, a result likely influenced by the limited number of available studies and small sample sizes. Age, baseline lung function, and adherence to the intervention protocols emerged as key factors potentially affecting these outcomes. The analysis also showed no significant effect on the FEV1/FVC ratio, a critical marker for airway obstruction, further constrained by the small number of eligible studies and limited participant data, rendering the findings inconclusive. However, the study by Jang et al. 8 reported a significant improvement in the FEV1/FVC ratio, likely attributable to the high level of participant adherence to the intervention protocol, which emphasized education and active engagement. In contrast, the study by Bohadana et al. 15 involving patients with COPD found that varenicline treatment had no significant effect on airway obstruction or spirometric parameters. Similarly, the study by Willemse et al., 16 which included both healthy individuals and patients with COPD, found that smoking cessation did not improve lung function in the COPD group. However, among healthy participants, cessation was associated with a reduction in airway inflammation. These contrasting outcomes may be attributed to the advanced age and the irreversible nature of lung damage in COPD patients, which likely limited their capacity for functional recovery. In contrast, the study by Chaudhuri et al. 34 involving asthma patients demonstrated significant improvements in lung function following cessation. Higher adherence to the intervention and the more reversible nature of airway obstruction in asthma likely contributed to these favorable results. Moreover, these findings from the narrative review—which reported significant improvements in FEV1 among asthma patients—highlight the importance of disease-specific responses to smoking cessation. The underlying pathophysiology and reversibility of airway obstruction in asthma differ from those in COPD and lung cancer, potentially leading to more pronounced spirometric improvements in certain populations.45–48 Therefore, pooling heterogeneous respiratory conditions may have obscured differential effects that are only detectable in disease-specific subgroup analyses. Additionally, the nonsignificant effects observed for FVC (n = 4) and the FEV1/FVC ratio (n = 2) may primarily reflect insufficient statistical power due to the limited number of studies reporting these outcomes. As a result, the pooled estimates for these parameters should be interpreted with caution. The small sample base limits the precision of effect size estimates and increases the risk of type II error, whereby true effects may remain undetected despite their clinical relevance. 49
Importantly, although this meta-analysis focused on short-term changes in spirometric indices, it is essential to emphasize that smoking cessation is strongly associated with long-term clinical benefits. These include reduced rates of exacerbation, slower disease progression, improved health-related quality of life, and lower mortality among individuals with chronic respiratory diseases. Therefore, our findings should not be interpreted as diminishing the overall value of cessation but rather as highlighting the modest and potentially delayed improvements in spirometric parameters, particularly in advanced disease stages. Furthermore, subgroup analysis suggested that younger patients may experience greater improvements in FEV1, possibly due to the greater reversibility of airway obstruction and lower cumulative structural lung damage. These findings imply that early smoking cessation—before the onset of severe or irreversible pathology—may provide more substantial functional recovery. As such, preventive cessation strategies targeting younger individuals or those with less advanced disease are especially important to maximize the pulmonary benefits of quitting.
Strength and limitations
As far as existing literature suggests, this is the first meta-analysis to systematically evaluate the effects of different smoking cessation interventions on lung function outcomes in individuals with chronic respiratory diseases. By rigorously selecting eligible clinical trials and performing comprehensive subgroup and sensitivity analyses, we sought to generate findings that are both robust and clinically meaningful. Nonetheless, several limitations should be acknowledged. A key limitation of this review is the limited number of studies on FVC and FEV1/FVC outcomes, potentially reducing statistical power. Additionally, the inclusion of heterogeneous populations (e.g. those with COPD, asthma, or lung cancer) may have introduced variability. Future research should explore disease-specific effects using stratified analyses with sufficient sample sizes. These challenges highlight the complexity of assessing smoking cessation’s effects in diverse populations. The variability in findings suggests a need for personalized approaches considering factors such as age and disease severity. Future high-quality, long-term studies are essential to clarify the benefits and predictors of lung function recovery. It is also important to note that three studies included in the meta-analysis had a high overall risk of bias, primarily due to issues with allocation concealment (Table 2). These studies may have influenced the pooled estimates, and caution is warranted when interpreting the findings. We acknowledge that the study was not prospectively registered, which is a methodological limitation and may have introduced a risk of bias related to the review process. A further limitation is that all included trials reported outcomes over a short-to-moderate duration (≤52 weeks), which may be insufficient to capture meaningful or sustained changes in lung function, particularly in chronic conditions with slow progression such as COPD. Long-term studies are needed to assess whether initial improvements persist or evolve over time following cessation interventions. 11
Conclusion
In summary, this meta-analysis found that smoking cessation was associated with a small and statistically nonsignificant improvement in FEV1, with no significant effects observed on FVC or the FEV1/FVC ratio. Although a trend toward improved lung function was observed in some subgroups, particularly younger individuals and those with asthma, these findings did not reach statistical significance and were limited by high heterogeneity and small subgroup sizes. Therefore, the short-term impact of smoking cessation on spirometric parameters appears limited, particularly in individuals with established or irreversible respiratory disease. Future research with longer follow-up durations and larger, stratified trials is needed to better assess the long-term pulmonary benefits of different cessation strategies.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251374626 - Supplemental material for Lung function improvement following smoking cessation in chronic respiratory conditions: A meta-analytic approach
Supplemental material, sj-pdf-1-imr-10.1177_03000605251374626 for Lung function improvement following smoking cessation in chronic respiratory conditions: A meta-analytic approach by Junhua Wang, Tao Zhong, Fang Guo, Chengcheng Sun, Parsa Jamilian and Yanrui Jia in Journal of International Medical Research
Footnotes
Acknowledgements
None.
Author contributions
Availability of data and materials
All analyses are available within an institutional repository and can be provided upon request.
Consent for publication
All authors consent to the submission and possible publication of this manuscript.
Declaration of conflicting interests
No potential conflicts of interest relevant to this article were reported.
Ethics approval and consent to participate
Not applicable.
Funding
None.
Human ethics
Not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
