Abstract
Objective
To investigate the analgesic effects of acetaminophen–mannitol injections after endoscopic lumbar discectomy.
Methods
This is a prospective case–control study involving 60 patients who were randomly selected from those who underwent endoscopic lumbar discectomy for lumbar disc herniation or lumbar spinal stenosis at Sanshui District People’s Hospital of Foshan between April and September 2024. Two groups were formed by randomly assigning patients, with each group containing 30 individuals. The observation group underwent treatment with acetaminophen–mannitol for pain relief, whereas the control group was administered oral etoricoxib and intravenous mannitol. A comparison was made between the analgesic effects of the two groups.
Results
Compared with the control group, the observation group showed significantly lower visual analog scale scores on the first, third, and seventh days after operation (P < 0.05). The observation group had a similar time to start walking and length of hospital stay as the control group. The total incidence rate of adverse reactions was zero.
Conclusion
Acetaminophen–mannitol injection has a good analgesic effect after endoscopic lumbar discectomy, which is beneficial for accelerating the postoperative recovery of patients.
Keywords
Introduction
Endoscopic lumbar discectomy’s surgical methods and its perioperative management have been gradually enhanced.1–5 However, there are still some difficulties and complications that are difficult to surmount. Among the complications following endoscopic lumbar discectomy, postoperative nerve root swelling and root pain are very common and cause significant discomfort to patients. In particular, this type of complication decreases the patients’ overall assessment during the surgical operation, impacts the postoperative functional restoration, and extends the length of hospitalization. As enhanced recovery after surgery is vigorously promoted, the application of multimodal analgesia in the perioperative period of spinal surgery has been the subject of increasing research, as seen in studies.6–8 Although acetaminophen (APAP) is a commonly used antipyretic and analgesic drug in clinics, its mechanism of action is still unclear. To the best of our knowledge, as a new compound injection, there is no relevant study on the analgesic effect of APAP–mannitol injection after lumbar endoscopic surgery. From April to September 2024, 60 patients who were hospitalized in the Foshan Sanshui District People’s Hospital after undergoing endoscopic lumbar discectomy were randomly selected as research subjects for this study. All of them were patients with lumbar disc herniation or lumbar spinal stenosis. The aim of the study was to determine the effectiveness of APAP–mannitol injections in relieving pain after endoscopic lumbar discectomy. The findings are presented in the following sections.
Literature and methods
General information
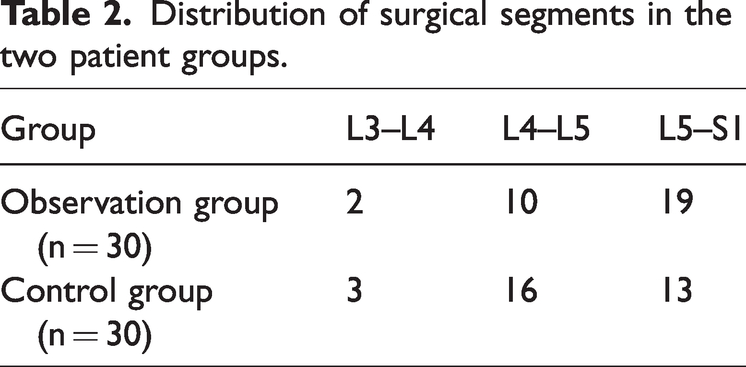
In our hospital, 60 patients who had lumbar disc herniation or lumbar spinal stenosis and were scheduled to undergo endoscopic lumbar discectomy were randomly selected as research subjects. These patients were randomly split into two groups using a computer. Each group comprised 30 patients. There were 17 men and 13 women in the observation group. Specifically, among them, 29 patients had undergone single-level surgery and 1 had undergone double-level surgery. There were 2 cases at L3–4 level, 10 cases at L4–5 level, and 19 cases at L5–S1 level, with an average patient age of 54.33 ±16.386 (range: 17–86) years. Specifically, the control group included 13 females and 17 males. Single-level surgery was performed on 28 patients, and 2 patients underwent double-level surgery. There were 3 cases at L3–4 level, 16 cases at L4–5 level, and 13 cases at L5–S1 level. The average age was 52.63 ± 14.845 years (range, 26–84 years).There was no significant difference in age between the two groups (P > 0.05), as shown in Tables 1 and 2. This research was approved by the Medical Ethics Committee of Sanshui District People’s Hospital in Foshan (SRY - KY - 2023052).
Comparison of the general data between the two patient groups (

Distribution of surgical segments in the two patient groups.
Inclusion and exclusion criteria
The inclusion criteria were as follows: (a) presence of radiographic lumbar spinal stenosis and/or foraminal stenosis, whether with or without disc herniation; (b) presence of certain radicular symptoms, such as radiating pain, numbness, and weakness, consistent with the imaging findings; (c) received an ineffective conservative treatment that included physical therapy and selective nerve root block for a minimum of 6 weeks; (d) an ability to understand pain rating scales and willingness to participate in this research and provide a signed consent form.
The exclusion criteria included the following: (a) patients with diseases such as lumbar degenerative spondylolisthesis or instability, congenital spinal stenosis, prior lumbar surgeries, fractures, tumors, and infections; (b) pregnant women as well as patients with mental disorders; (c) patients who had contraindications to the drugs that were used in the study; (d) patients with serious underlying diseases; (e) patients with a history of chronic pain or opioid abuse; (f) patients who regularly used sedatives and antipruritic and antiemetic drugs; and (g) patients who were judged as unqualified to participate in the study by the investigators.
Methods
The study is a prospective case–control study. Before the surgery, all patients fasted for 8 h. During the operation, a single surgeon performed either single-level or double-level percutaneous endoscopic lumbar discectomy via a posterolateral approach, and all patients were given epidural anesthesia.
The specific steps are as follows: 1. Under fluoroscopy, the needle was placed at the lateral edge of the pedicle shadow on the affected side of the target intervertebral space in the anteroposterior position and at the lower edge of the intervertebral foramen in the lateral position. 2. A guide wire was inserted, the puncture needle was removed, a skin incision was made at the guide wire entry point, and the working channel was inserted along the guide wire. 3. Under the channel, the bone of the ventral upper part of the superior articular process was removed by trephination, and the posterior inferior foramen was expanded and formed. 4. Under fluoroscopy, the channel had entered the spinal canal, and the spinal endoscope was inserted through the working channel. 5. Under the endoscope, the spinal canal was decompressed; parts of the ligamentum flavum, posterior longitudinal ligament, and nucleus pulposus tissue of the intervertebral disc were removed; the dura and nerve root were fully decompressed; and the nerve root and dural sac were pulsated well.
The postoperative analgesic regimen was as follows: 1. In the control group, etoricoxib tablets (Chinese medicine approval number H20193273; specification: 60 mg), with a dosage of 60 mg were consumed orally once a day, along with 20% mannitol injection (approval number in Chinese medicine: H32022586; specification: 250 mL: 50 g), 125 mL administered intravenously every 12 h. 2. In the observation group, 50 mL of APAP–mannitol injection (Chinese medicine approval word H20223028; specification: 50 mL: 500 mg) was administered intravenously every 12 h.
This study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. All patient details were de-identified in this study.
Indicators of observation
The assessment of the visual analog scale (VAS) scores for lower back and lower limb pain was performed on the first, third, and seventh days following the surgery. The 10-point VAS score was positively correlated with the level of pain. The time to start walking and the duration of the postoperative hospital stay were noted. Gastrointestinal reactions, allergic reactions, and other adverse drug reactions were also recorded.
Statistical methods
Statistical software (SPSS 25.0) was used for data analysis. Count data (the incidence of adverse reactions) were presented as the number of cases (n) and the rate (%), and comparisons among groups were conducted through the chi-square test. The measurement data (VAS scores) were normally distributed. A t-test was used for comparisons between groups, and the outcomes were expressed as (
Results
Comparison between the two groups regarding the time until postoperative ambulation and hospital stay
Prior to surgery, there was no significant difference in the VAS scores between the two groups (P > 0.05). The VAS scores of the waist and lower limbs in the observation group were markedly lower than those in the control group on the first, third, and seventh days after surgery (all P < 0.05), as shown in Table 3.
Comparison of the VAS scores between the two patient groups (

CI: confidence interval.
Comparison of the time to postoperative walking and the length of hospital stay between the two groups
The time until postoperative ambulation and the length of hospital stay in the observation group were comparable to those in the control group (P > 0.05) (Table 4).
Comparison of the time until postoperative ambulation and hospital stay between the two groups (

CI: confidence interval.
Comparison of the incidence of adverse reactions between the two patient groups
In both groups, the overall incidence of adverse reactions was zero.
Discussion
In patients with lumbar disc herniation or lumbar spinal stenosis, long-term nerve root problems lead to lumbar and lower limb pain. Moreover, during endoscopic lumbar discectomy, the traction and stimulation of nerve roots often result in obvious nerve root edema. Consequently, these patients are likely to experience lumbar wound pain and pain radiating to the lower limbs following the surgery. 9 Typically, such symptoms tend to disappear on their own. However, during the recovery period, patients may suffer from pain, which could lead to an unpleasant experience and might even make them question the surgical outcome.
There are multiple factors that lead to nerve root edema. Chiefly, in terms of anatomical structure, the spinal nerve roots do not have an epineurium or perineurium. Consequently, they do not have the nerve barrier function and can be easily influenced by changes in the surrounding environment. Thus, compared with peripheral nerves, they are more vulnerable to compression and traction forces. 10 Research has indicated that nerve root compression can result in a decrease in the blood flow of the relevant nerve root as a result of increased vascular permeability within the nerve, which in turn gives rise to nerve root edema.11,12 Furthermore, because of the nerve root’s anatomical structure and the nucleus pulposus inflammation in intervertebral disc herniation, compression of the nerve root by the nucleus pulposus will ultimately result in nerve root edema and nerve root damage. This can lead to pain and a decrease in nerve function.
Consequently, the treatment for reducing swelling and relieving pain after the operation is of great significance. 13 Furthermore, for patients with a moderate or high risk of venous thromboembolism following endoscopic lumbar discectomy, effective postoperative pain relief is required. This can decrease the incidence of deep vein thrombosis, enhance sleep quality and immunity, facilitate postoperative recuperation, shorten the hospital stay, and boost patient satisfaction. For postoperative analgesia following endoscopic lumbar discectomy, APAP and non–steroidal anti-inflammatory drugs typically form the main elements in most multimodal pain management plans,14–16 and mannitol is a frequently used drug for relieving nerve root edema after operation. 17 Consequently, compared with the traditional “non–steroidal anti-inflammatory analgesic drugs +mannitol” plan, the use of APAP–mannitol injection in the analgesia plan after endoscopic lumbar discectomy has the following benefits. First, it can decrease the use of opioids during or after the operation and opioid–related adverse reactions. 18 Second, in contrast to the “nonsteroidal anti-inflammatory analgesics + mannitol” plan, using APAP–mannitol injection solely for postoperative pain relief can reduce adverse reactions to common drugs.
In this study, there was no significant difference in the VAS scores between the two groups before surgery (P > 0.05). Specifically, VAS scores of the waist and lower extremities in the observation group on the first, third, and seventh days following the surgery were 3.53 ± 0.776, 1.93 ±0.868, and 1.20 ± 0.407, respectively, which were lower than those in the control group (all P < 0.05). It was indicated that the analgesia of APAP–mannitol could relieve the pain in patients. There was no difference in the time to ambulation and the length of hospital stay between the two groups. This was considered to be related to the minimally invasive operation and the accelerated postoperative recovery. As stated by Manley and Taddio, 19 gastrointestinal reactions (such as nausea and vomiting), fever, and headache are common adverse reactions to APAP. In this study, no adverse drug reactions were observed in either group, although insomnia was noted as a possible reaction. This was because of the small sample size.
APAP is among the most frequently used drugs for postoperative pain relief; however, its mode of action is still not fully understood. There are some indications that APAP exerts its analgesic effects by inhibiting the cyclo-oxygenase pathway and reducing prostaglandin generation. The 5-hydroxytryptamine–mediated descending inhibitory pain pathway can be stimulated by APAP.20–22 Ohashi et al. 23 and Mallet et al. 24 reported that the main mechanism of APAP–mannitol is that its metabolites can act on the TRPV1 receptor in the brain, and AM404, another metabolite of APAP–mannitol, can directly induce analgesia through the TRPV1 receptor with fewer adverse reactions.
Regarding the mechanism of drug overdose and toxicity, studies have shown that APAP overdose can lead to severe liver toxicity, and the mechanism involves the binding of APAP oxidation products with glutathione to form APAP–glutathione conjugates, which are further metabolized to APAP–cysteine and APAP-N-acetylcysteine conjugates. 25 Excessive APAP leads to glutathione depletion, which in turn causes hepatocyte necrosis 7. 26 Another study demonstrated that APAP and structurally similar compounds can bind to quinone reductase 2 (NQO2), which may act as a novel nontarget to regulate APAP levels. NQO2-mediated superoxide anion production may serve as a new mechanism that enhances the toxicity of APAP. 27
In terms of pharmacokinetics and metabolism, a study explored the pharmacokinetics and metabolism of APAP as well as potential hepatotoxic damage to evaluate its possibility as a toxicity model in humans. 28 At an APAP dose of 900 mg/kg/day, no adverse effects were observed during 14 consecutive days of administration. In another study, metabolite analysis of APAP and its metabolites in urine determined via (1)H NMR spectroscopy revealed APAP and its glucuronide and sulfate metabolites, while glutathione-derived metabolites, such as cysteine conjugates, were found in very low amounts and sulfhydrylurate was not detected. 28
In terms of drug safety and dosage, although APAP is well tolerated at therapeutic doses, it may cause hepatotoxicity in the case of overdose, and hepatotoxicity has been reported even at therapeutic doses. 29 A study noted that adolescents may have insufficient awareness of the toxicity and potential lethality of APAP overdose, suggesting the need to increase public awareness of the safe use of APAP. 30
In terms of other effects of drugs, some studies have shown that APAP may exert antiproliferative effects and induce apoptosis in certain cancer cells, preventing cell growth. 31 Another study proposed that APAP may be a hormone disrupter, interfering with sex and thyroid hormone functions that are essential for normal brain development and thus may not be considered a safe drug during pregnancy. 32
Conclusion
To summarize, APAP–mannitol injection has a favorable analgesic effect following endoscopic lumbar discectomy and induces few adverse reactions, which make it advantageous for promoting the postoperative recovery of patients. Nevertheless, because of the unique appearance and dosage form of APAP–mannitol injection, the use of a placebo in clinical trials may cause patients’ aversion and other factors, barriers to the use of blinding in the study. In fact, diabetes mellitus and smoking pose significant challenges to postoperative pain management. These factors were not taken into account during the initial design of the study, which is one of the shortcomings of this research. The study’s small sample size and the selection of a particular surgical method, namely, minimally invasive spinal endoscopic surgery, with its merits of minimal trauma and rapid recovery, might restrict its reliability and generalizability. To verify and expand on the current research results, future research should include large sample sizes, explore different surgical procedures, and enable blinding.
Footnotes
Acknowledgments
This study was conducted according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 33
Author contributions
Jie-Bin Huang wrote the main manuscript text; Yin-Feng Guo and Cheng-Zhao Li collected the data; Wei-Xiong Chen and Lei Pan contributed to the data analysis; Bai-Yong Zhu, Yu Lei, Wei-Dong Jiao, and Li-Bin Zheng collected the references; Hou-Jun Xue conceived and designed the study; and all the authors read and approved the final manuscript.
CONSORT 2010 checklist statement
We have read the CONSORT 2010 checklist, and the manuscript was prepared and revised according to the CONSORT 2010 checklist.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper.
Data access statement
All the data generated or analyzed during this study are included in this published article (and its supplementary information files).
Ethics approval statement
This study was reviewed by the Medical Ethics Committee of Sanshui District People’s Hospital of Foshan (date: 29-08-2023, relevant reference number: SRY-KY-2023052).
Funding
This study was supported by a grant from the Red Cross Foundation of China from the Foundation (Item Number: CRCF-YXFN-202302168) of Basic Research.
Informed consent statement
Written informed consent was obtained from the patients for publication of this study and data.
