Abstract
Ovarian hemangioma is an extremely rare benign tumor in clinical practice. We report the case of a postmenopausal woman presenting with vaginal bleeding, in whom imaging revealed a right adnexal mass. Surgical excision of the adnexa was performed, and pathological examination confirmed a right ovarian hemangioma. Concurrent findings included endometrial polyps and cervical polyps.
Case presentation
A postmenopausal woman in her early 50s presented with vaginal bleeding to The Affiliated Dongguan Songshan Lake Central Hospital, Guangdong Medical University, China, in August 2024. Transvaginal ultrasound revealed a hyperechoic endometrial lesion (11 × 6 mm), suggestive of an endometrial polyp, and a right adnexal mixed-echogenicity mass (51 × 38 mm) (Figures 1 to 3). Gynecological examination identified a 0.2 × 0.2-cm polypoid lesion at the cervical os. She had no significant medical history and no family history of malignancy.

Hyperechoic endometrial lesion.

Right adnexal mixed-echogenicity mass.
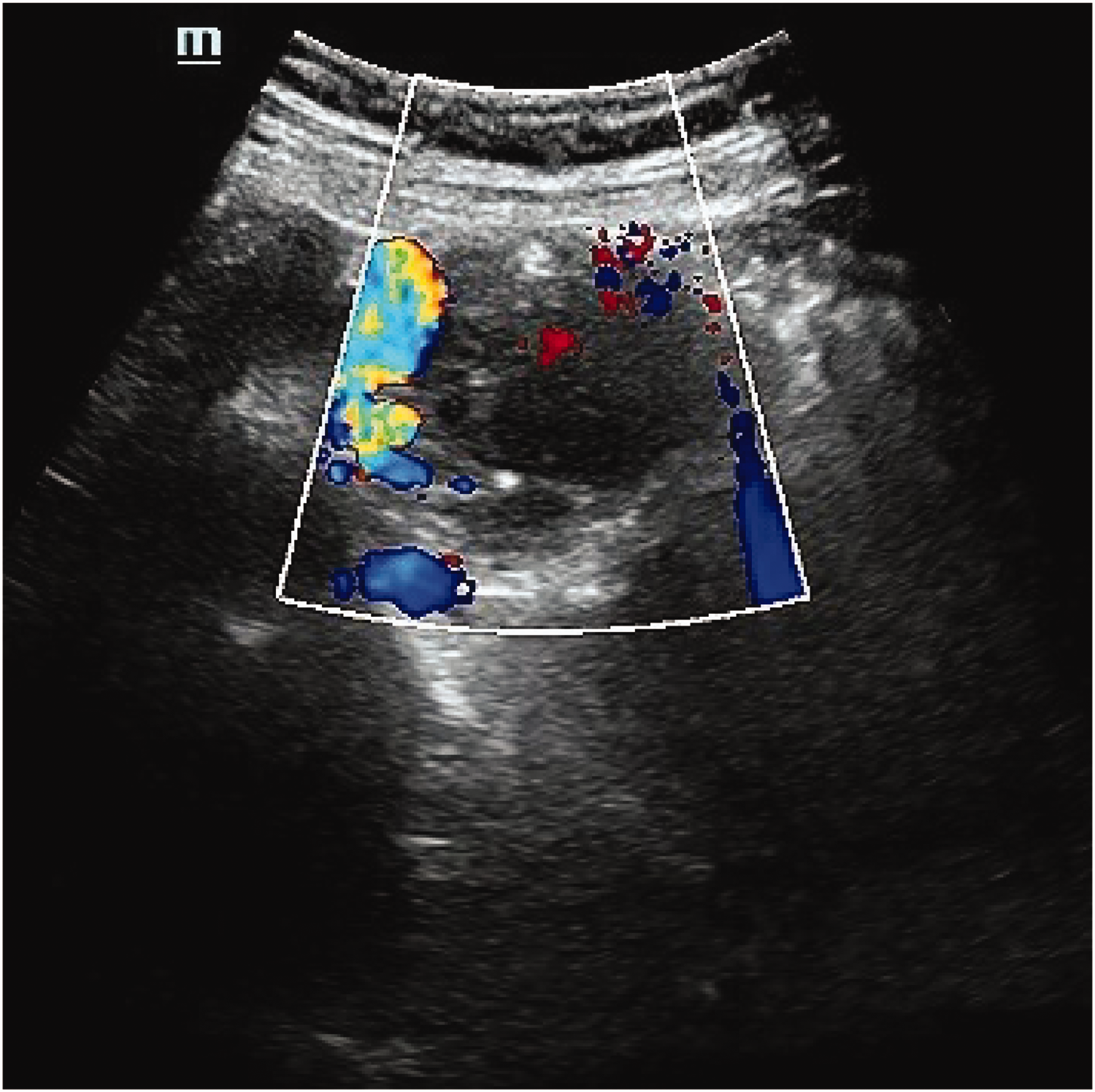
CDFI showing spot-shaped blood flow signals in the mixed echo area. CDFI: color Doppler flow imaging.
Pelvic magnetic resonance imaging (MRI) showed that the mass demonstrated low signal intensity on T1-weighted imaging (Figure 4) and slightly high signal intensity on T2-weighted imaging (T2WI) (Figure 5); this high signal intensity remained prominent on fat-suppressed T2WI (Figure 6). Noncontrast computed tomography (CT) revealed an irregular marginated mass with soft-tissue density and scattered calcifications (Figure 7). Post-contrast CT showed centripetal enhancement of vessels from the periphery toward the center. Persistent vascular enhancement was observed in the delayed phase, consistent with the characteristic “rapid wash-in and slow wash-out” pattern (Figures 8 to 10).

The mass demonstrating low signal intensity on T1WI. T1WI: T1-weighted imaging.

The mass demonstrating slightly high signal intensity on T2WI. T2WI: T2-weighted imaging.

The mass demonstrating prominently high signal intensity on fat-suppressed T2WI. T2WI: T2-weighted imaging.

Noncontrast CT revealing an irregular mass with soft-tissue density and scattered calcifications. CT: computed tomography.

The CT arterial phase showing contrast around the periphery of the tumor. CT: computed tomography.
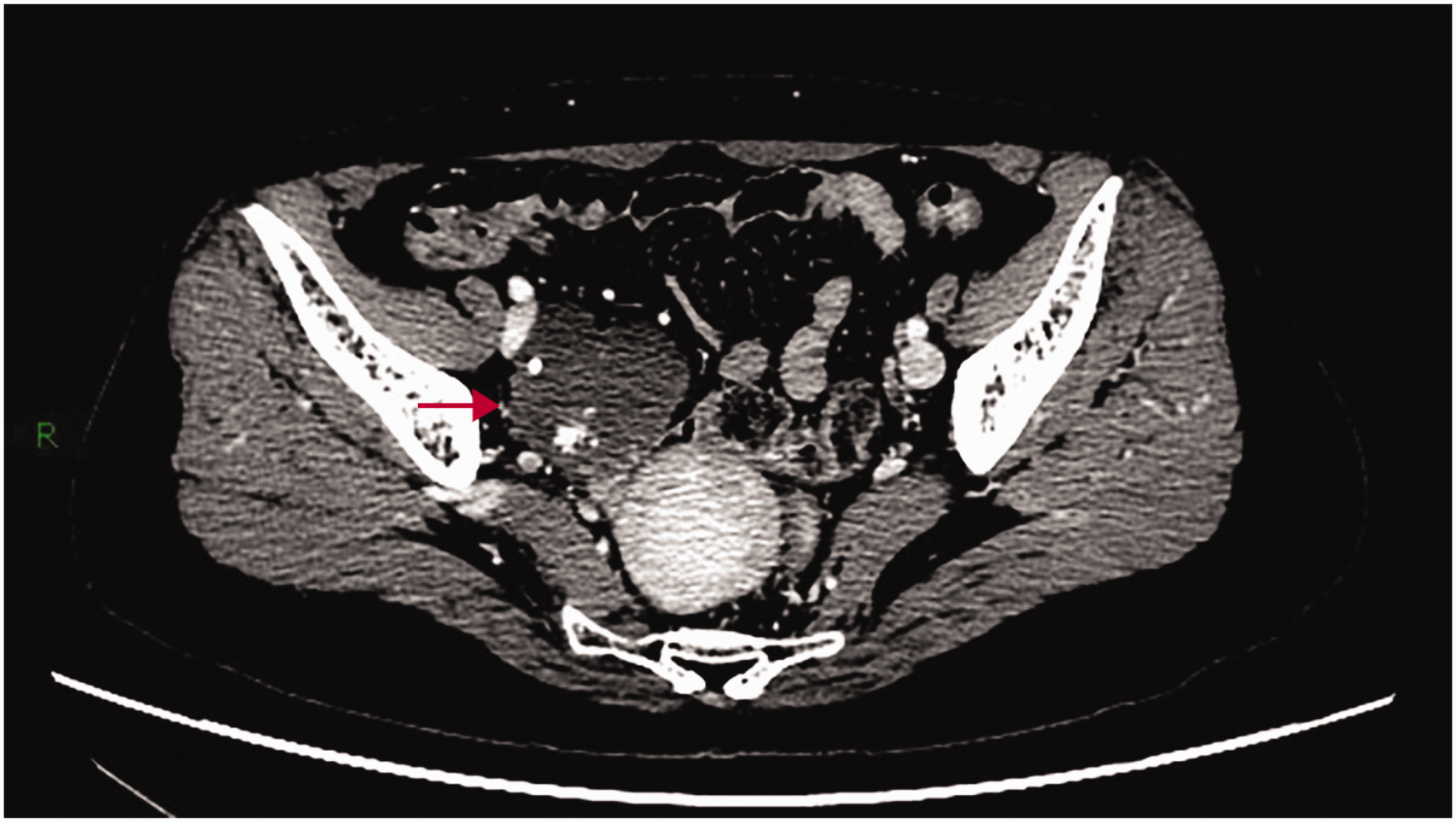
The CT venous phase showing contrast filling the central portion of the tumor.

The CT delayed phase showing increased contrast retention within the tumor. CT: computed tomography.
Laboratory investigations showed that tumor markers were within normal limits. Hormonal profile analysis revealed the following results: estradiol, 34.1 pg/mL; follicle-stimulating hormone, 23.0 mIU/mL; and luteinizing hormone, 10.4 mIU/mL. Her progesterone, testosterone, and prolactin levels were within the normal ranges.
Hysteroscopy was initially performed, revealing both cervical and endometrial polyps, which were subsequently confirmed via histopathological examination. Laparoscopic exploration demonstrated a vascular-rich, clustered tissue on the surface of the right ovary, while the uterus, bilateral fallopian tubes, and left ovary appeared normal (Figure 11). A laparoscopic bilateral salpingo oophorectomy was performed. Intraoperative frozen section analysis suggested an ovarian vascular tumor, likely hemangioma. Postoperative paraffin-embedded section pathology confirmed proliferative vascular channels consistent with hemangioma in the right adnexa. Immunohistochemistry results were as follows: (F8)(+), CD31(+); D2-40(+); calretinin(−) (Figures 12 to 14). Since the operation, the patient has remained in good health and is being monitored regularly.

A vascular-rich, clustered tissue on the surface of the right ovary based on laparoscopic exploration.
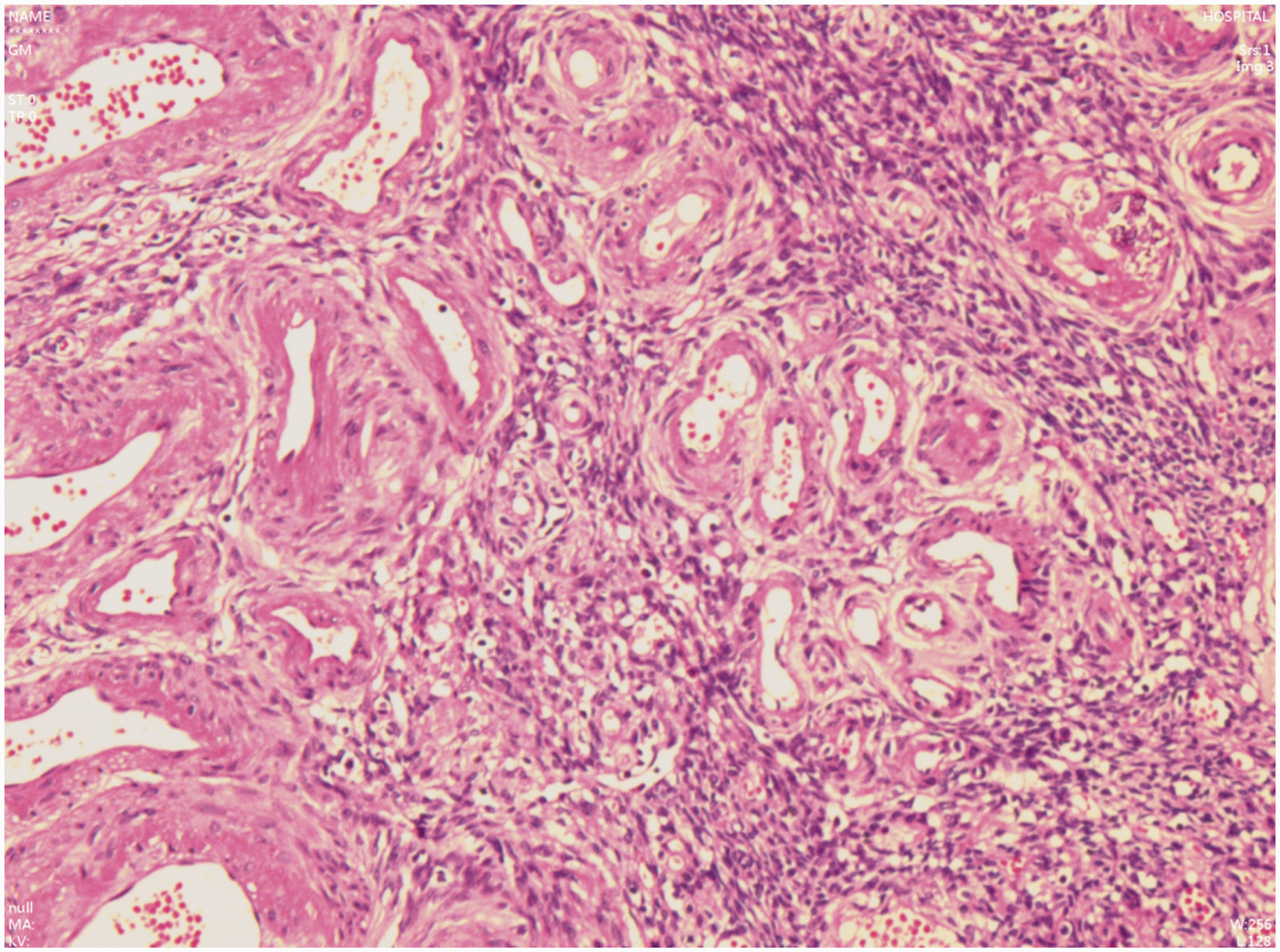
HE staining showing vascular proliferation within the tumor, with varying diameters of vessels, consistent with mixed hemangioma. HE: hematoxylin and eosin.

HE staining showing a thin and venous blood vessel wall inside the tumor. HE: hematoxylin and eosin.

Immunohistochemistry (IHC) showing positive staining for F8.
We have de-identified the patient’s details and obtained approval from the Ethics Committee of The Affiliated Dongguan Songshan Lake Central Hospital, Guangdong Medical University (2023A1515220219) for publication of this report. The reporting of this study conforms to the Case Report (CARE) guidelines. 1 We obtained informed consent from the patients for treatment and publication.
Discussion
Hemangiomas are benign tumors arising from the abnormal proliferation of vascular endothelial cells, histologically classified into three subtypes: cavernous, capillary, and mixed types. Ovarian hemangiomas are exceptionally rare, with fewer than 60 cases reported in the literature (published in English) to date. The age at diagnosis varies widely, ranging from 2 to 81 years.2–4 Clinical presentations are nonspecific; patients typically present with abdominal distension, pain, incidental findings on imaging, abnormal uterine bleeding, or postmenopausal bleeding, as exemplified in this case.
On ultrasonography, ovarian hemangiomas typically present as well-circumscribed, hyperechoic adnexal masses with irregular hypoechoic areas and moderate-to-marked vascularity on Doppler imaging. 5 CT and MRI features closely resemble those of hepatic hemangiomas, demonstrating centripetal enhancement and delayed contrast enhancement on multiphase imaging. 6 Preoperative diagnosis is often challenging due to nonspecific imaging findings that overlap with those of borderline/malignant tumors and the absence of elevated tumor markers (e.g. cancer antigen 125 (CA-125) and human epididymis protein 4 (HE4)). Definitive diagnosis ultimately requires histopathological confirmation.
Ovarian hemangiomas are frequently associated with concurrent endometrial polyps, endometrial hyperplasia, and even endometrial carcinoma.7–10 This association could be explained by ovarian stromal luteinization. A study reported stromal luteinization in 78.6% of all ovarian hemangiomas. 11 These luteinized stromal cells produce steroid hormones, including androgens and estrogens, which can lead to clinical manifestations such as hirsutism, endometrial polyps, endometrial hyperplasia, and endometrial carcinoma in patients.
However, this mechanism cannot account for all cases. As illustrated in the present case, bilateral ovarian pathology revealed no evidence of stromal luteinization; however, endometrial polyps and cervical polyps were present. Similar findings have been reported in other cases; 12 furthermore, immunohistochemistry has demonstrated the absence of estrogen receptor (ER) and progesterone receptor (PR) expression in the endothelial cells of ovarian hemangiomas. This suggests that the pathogenesis of ovarian hemangiomas may be independent of estrogen and progesterone stimulation. Unfortunately, ER or PR testing was not performed in the current case. In cases of ovarian hemangioma without interstitial luteinization, the mechanisms underlying the concurrent occurrence of endometrial polyps or other endometrial lesions require further investigation.
Notably, ovarian hemangiomas must be differentiated from estrogen-secreting tumors (e.g. granulosa cell tumors and thecomas) and may mimic malignancy when accompanied with pseudo-Meigs syndrome (ascites and pleural effusion),13,14 which can result in unnecessary extensive surgery.
So far, definitive management involves surgical excision of the adnexa with intraoperative frozen section analysis to confirm diagnosis. The prognosis is favorable, and long-term clinical surveillance is generally sufficient. However, additional case studies are warranted to elucidate the pathogenesis, refine the diagnostic criteria, and optimize the management of ovarian hemangiomas.
Footnotes
Acknowledgements
We thank the participant and her family for their cooperation during this study.
Author contributions
Sai-Tian Zeng performed the clinical diagnosis and operation. Min Guo, Xu-Ni Wu, and Yin Wang wrote the manuscript. Cui-Fen Li provided guidance for diagnosis, treatment, and manuscript writing.
Availability of data and materials
Data sharing is not applicable to this article because no datasets were generated or analyzed during the current study.
Consent for publication
We obtained written informed consent from the patient before preparing the case report.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the medical ethical committee of The Affiliated Dongguan Songshan Lake Central Hospital, Guangdong Medical University (2023A1515220219).
Funding
No funds, grants, or other support was received.
