Abstract
Objective
The cause of most congenital heart defect cases remains unknown. A previous study showed a link between maternal iron deficiency and congenital heart defects in mice; however, this association is not yet clear in humans. This study aimed to explore the potential relationship between maternal iron status and congenital heart defects in infants.
Methods
We conducted a case–control study, including 70 mothers whose infants had congenital heart defects and their infants (case group) and 30 mothers whose infants did not have any congenital heart defects and their infants (control group). We interviewed eligible mothers and collected peripheral blood samples to analyze iron-related biomarkers. We used binomial logistic regression to estimate the odds ratio (95% confidence intervals) for the association of maternal iron status with congenital heart defects.
Results
Serum ferritin, serum iron, and transferrin saturation levels were lower, while total iron binding capacity was higher among mothers of the case group than among those of the control group (p < 0.05). Additionally, mothers in the case group were at higher risk of having iron deficiency (odds ratio = 2.91, 95% confidence interval: 1.143–7.418) than those in the control group.
Conclusions
Maternal iron status was lower among mothers of the case group than among those of the control group. Furthermore, mothers in the case group had a greater likelihood of experiencing iron deficiency than those in the control group.
Introduction
Congenital heart defects (CHD) are structural abnormalities of the heart or great vessels of the heart. CHD develop during early fetal development and present at birth. CHD are considered the most common birth defect, accounting for approximately one-third of all congenital anomalies. 1 An estimated 8 to 12 per 1000 live births worldwide have CHD. 2 CHD is a major health problem associated with lifelong illness and complication in infants with birth defects and is the leading cause of death in children with congenital defects.1,3 The etiology of CHD remains elusive, with approximately 20% of CHD cases being attributed to well-known chromosomal or genetic disorders. Thus, a majority of CHD cases are believed to be multifactorial, and both genetic and environmental factors have contributed to the pathogenesis of the disease.4,5
These environmental risk factors include maternal diseases, teratogen exposure, and nutrient deficiencies.5,6 Iron deficiency (ID) is the most prevalent nutritional deficiency worldwide and is the most common contributor to anemia. ID anemia (IDA) affects approximately 50% of pregnant women globally. 7 Pregnancy is associated with increased iron demand due to enhanced blood volume in pregnant women and the growth of the fetus and placenta. Thus, pregnant women are at high risk of developing ID. 8 Animal studies have shown that ID during pregnancy affects the growth and development of fetal organs such as the heart. 9 An animal study on mice demonstrated that maternal ID disrupts embryonic heart development. 10 However, clinical studies on the relationships between maternal iron status and CHD are limited. A clinical study suggested a relationship between low iron intake during pregnancy and the risk of CHD. 11 Another study found that iron levels were high among mothers of CHD infants. 12
This study aimed to evaluate iron status in the mothers of CHD infants and their infants. Moreover, this study aimed to verify the relationship between maternal iron status and the risk of CHD.
Methods
Experimental design and type of participants
We performed a case–control study in two hospitals in Damascus City, Syria, a Pediatric University Hospital and the Heart Surgery University Hospital from November 2022 to November 2023. This research was reviewed and obtained both academic and ethical approval from the corresponding committees of the Damascus University. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 13
Cases and control were selected randomly among mothers during the period after delivery, (median = 16 weeks, interquartile range (IQR): 4–44 weeks). The case group included 70 mothers whose infants had an isolated CHD and their infants (n = 70), whereas the control group included 30 mothers whose infants had no congenital abnormalities and their infants (n = 30).
The diagnosis of CHD in infants was made based on echocardiography performed by specialist pediatric cardiologists, and all infants in the study were being breastfed.
Mothers in the two groups were excluded if they had any of the following: (a) diabetes mellitus; (b) blood hypertension; (c) hyperthyroidism; (d) CHD; (e) hemolytic anemia; (f) blood transfusion within the past 6 months; (g) infections or inflammatory conditions; (h) active smoking during pregnancy; (i) ongoing birth control tablet intake; (j) iron supplementation during pregnancy and after delivery; or (k) severe bleeding during pregnancy and after delivery.
Mothers in both the groups were excluded if their infants had any of the following: (a) genetic disorders or chromosomal abnormalities; (b) siblings or first-degree relatives diagnosed with CHD; (c) recent infections or inflammation; (d) hemolytic anemia; (e) blood transfusion; (f) ongoing intake of iron supplements; and (g) ongoing intake of iron-fortified formula.
Finally, 70 mothers were included as cases and 30 as controls as per the inclusion criteria.
However, it should be noted that we intended to include mothers in both groups from a similar socioeconomic background to minimize the effect of dietary habits on iron levels.
Sample size assessment
The estimated ID prevalence was 32.7%, as reported in the World Health Organization (WHO) global anemia estimate. 14
Using the online Epitools Calculators for a matched case–control study, we set the assumed odds ratio (OR) at 3, power at 80%, and confidence interval (CI) at 95%; the minimum required sample size for each group was 39. However, based on our achieved control-to-case ratio, which was equal to 0.4, the required sample sizes for cases and controls were 97 and 39, respectively. Considering the strict inclusion criteria and timeline of the study, we could enroll 70 mothers as cases and 30 as controls.
Ethics approval
This study was approved by the Biomedical Research Ethical Committee at Damascus University, and the reference number is PH-290122-30. The study was conducted according to the guidelines of the Declaration of Helsinki of 1975 as revised in 2024. All mothers who were enrolled provided written informed consent.
The informed consent included a statement confirming that the identity of any participant would be concealed. The information of the participants would be included anonymously and would not be used anywhere other than the current study.
Covariable assessment
We interviewed the mothers using a standardized questionnaire to verify their alignment with the study criteria and collect important information. The study information consisted of sociodemographic characteristics (maternal age, gestational age, type of delivery, residence, education level, and income) and maternal health factors during pregnancy (active/passive smoking, bleeding, illnesses, medication use, and supplements use).
We obtained permission to access the medical files of the enrolled infants and obtained health-related information, including infant age, birth weight of the infant, CHD type, bleeding, other illnesses, medication use, and supplements use.
Biomarker measurement
We collected peripheral venous blood from each mother and infant into two tubes, an ethylenediaminetetraacetic acid tube for the assessment of complete blood count (CBC) and a dry tube to obtain serum for the assessment of iron-related biomarkers. The iron-related biomarkers included serum ferritin (SF), serum iron (Fe), unsaturated iron binding capacity (UIBC), total iron binding capacity (TIBC), and transferrin saturation (TSAT). We analyzed CBC using an automatic analyzer. We used a sandwich chemiluminescent immunoassay (Mindray, China) for measuring SF levels. We used a colorimetric assay (Biomajesty, Germany) for measuring Fe levels and UIBC. Then, we calculated TIBC by summing Fe level and UIBC and calculated TSAT using the following formula: (TSAT = (Fe/TIBC) × 100).
According to the WHO criteria, we diagnosed ID among the mothers at a ferritin concentration <15 ng/mL. We used a hemoglobin cutoff value of 12 g/dL to diagnose anemia in nonpregnant women. Therefore, we detected IDA among the mothers at a ferritin concentration <15 ng/mL and hemoglobin level <12 g/dL and absolute ID at a ferritin concentration <15 ng/mL and hemoglobin level ≥12 g/dL.15,16
We identified absolute ID rather than functional ID, adhering to the definitions of both the conditions. Functional ID occurs when the body cannot use the available iron due to inflammation or tumors, while absolute ID results from a true lack of iron, often due to low intake. 17 The mothers in our study were healthy, had no medical history related to functional ID, and were not taking iron supplements, with most reporting low-iron diets due to their low income status. The ferritin cutoff level used to detect ID in these mothers was 15 ng/mL, as recommended by the WHO for absolute ID, while women with functional ID typically show ferritin levels of 50–500 ng/mL according to various guidelines. 18
Statistical analysis
Statistical data analysis was performed using Statistical Package for Social Sciences version 24. The normality distribution of data was detected using the Komogorov–Smirnov test. We applied the chi-square (x2) test to compare categorical variables between the two groups. Continuous variables with normal and non-normal distributions were compared between the two groups using the Student’s t-test and Mann–Whitney U test, respectively. We performed a binomial logistic regression model to estimate the OR with corresponding 95% CIs for total CHD associated with maternal iron status. The sociodemographic characteristics (maternal age, gestational age, type of delivery, education level, income, and residence) were chosen as covariables in the model to minimize confounding. P-value <0.05 was considered to indicate statistical significance.
Results
General characteristics of the study sample
The general demographic characteristics of the study participants are shown in Table 1. There were no significant differences in the maternal age, gestational age, neonatal gender, type of delivery, passive smoking status, residence, education level, and income between the two groups.
General characteristics of the study participants. a

SD: standard deviation.
Categorical variables were compared between the case and control groups using chi-square test. Continuous variables were compared between the two groups using independent t-test and were expressed as mean and SD values.
Infants in the case group (those with CHD) had lower birth weights than the control infants (2.54 ± 0.61 vs. 2.97 ± 0.51 kg).
The CHD types among the case groups are shown in Table 2. Approximately two-thirds of the CHD infants had cyanotic CHD (64.28%) with multiple lesion CHD being the most common type (31.42%).
CHD types among infants in the case group. a
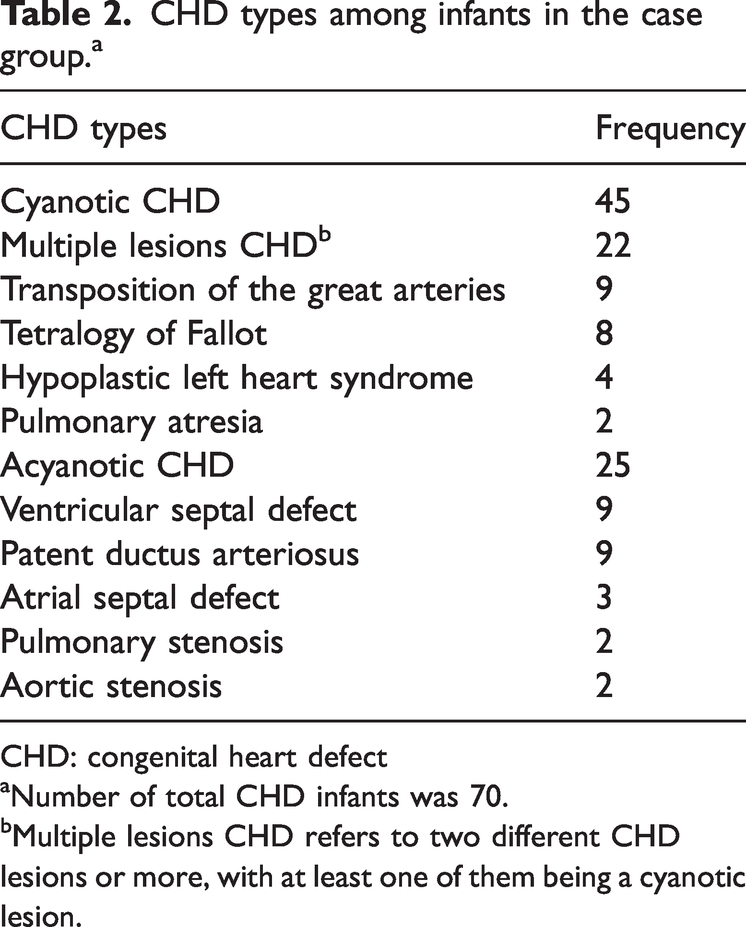
CHD: congenital heart defect
Number of total CHD infants was 70.
Multiple lesions CHD refers to two different CHD lesions or more, with at least one of them being a cyanotic lesion.
Blood biomarkers in mothers
Table 3 shows the blood biomarkers of mothers and infants in the two groups. Compared with the mothers in the control group, those in the case group had lower SF (13.69 vs. 21.2 ng/mL), Fe (51 vs. 72 µg/dL), and TSAT (10.77% vs. 17.45%) levels. Maternal TIBC was higher (446 vs. 406 µg/dL) among mothers in the case group than among those in the control group.
Iron-related biomarkers of the mothers and infants enrolled in the study.a

TIBC: total iron binding capacity; TSAT: transferrin saturation; Hb: hemoglobin; MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin; MCHC; mean corpuscular hemoglobin concentration; RDW: red cell distribution width.
We used the independent t-test for comparing continuous variables with normal distribution, and Mann–Whitney U test for comparing continuous variables with non-normal distribution.
The continuous variables are expressed as median and IQR (interquartile range) values (25th percentile–75th percentile).
We observed no significant differences in hemoglobin levels, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration (MCHC), and red cell distribution width (RDW) between the mothers in the two groups.
Blood biomarkers in infants
The infants in the case group had higher average hemoglobin level (11.85 vs. 11.2 g/dL) and RDW (16.35% vs. 13.3%) and lower MCHC (32.1 vs. 34.65 g/dL) than those in the control group. No significant differences were found in SF, Fe, TSAT, and TIBC between the two groups. We further categorized the infants in the case group into two groups as follows: cyanotic group (n = 45) and acyanotic group (n = 25). We found that infants in the cyanotic group had lower SF (10.8 (1.45–165.9) ng/mL) than those in the acyanotic group (24.5 (6.97–288.7) ng/mL; P = 0.010) and control group (34.39 (2.44–245.5) ng/mL; P = 0.021).
Therefore, the infants in the cyanotic group were at higher risk of developing ID than those in the acyanotic group (OR = 3.5, 95% CI: 1.25–9.85; P = 0.015) and control group (OR = 2.84, 95% CI: 1.09–7.40; P = 0.030).
We also categorized the infants based on their age as follows: infants aged <6 months and infants aged >6 months. In the CHD group, the infants aged <6 months had an average SF level of 11.04 (2.54–201.37) ng/mL, while those aged >6 months had an average SF level of 21.4 (1.45–288.7) ng/mL. In the control group, the infants aged <6 months had an average SF level of 55.45 (3.6–245.5) ng/mL, while those aged >6 months had an average SF level of 34.39 (2.4–81.3) ng/mL.
Maternal ID and CHD
ID (SF <15 ng/mL) was detected in 34 of the 70 mothers in the case group, compared with 8 of the 30 mothers in the control group.
We found that the mothers of CHD infants (OR = 2.91, 95% CI: 1.143–7.418) were more likely to have ID than those of non-CHD infants.
We have classified the mothers of each group into three groups based on their SF and hemoglobin levels:
Mothers with ID but no anemia whose SF levels were <15 ng/mL and hemoglobin levels were ≥12 g/dL. Mothers with IDA who had ferritin levels <15 ng/mL and hemoglobin levels <12 g/dL. Mothers with adequate iron levels and ferritin levels >15 ng/mL.
Table 4 presents the classification of the mothers in the case and control groups based on their SF and hemoglobin levels.
Classification of the mothers in the case and control groups based on their ferritin and hemoglobin levels.

SF: serum ferritin; Hb: hemoglobin
We classified the mothers of the two groups based on their serum ferritin and hemoglobin levels.
Table 5 shows that the mothers of CHD infants (OR = 4.01, 95% CI: 1.08–15.5) were more likely to have absolute ID than the control mothers.
Maternal iron deficiency and congenital heart defects. a

CHD: congenital heart defects; OR: odds ratio; CI: confidence interval.
The correlation was performed using binomial logistic regression models.
The reference group was the control group (mothers of infants without CHD).
Adjusted for sociodemographic characteristics (maternal age, gestational age, type of delivery, education level, income, and residence).
Discussion
Recent studies suggest a relationship between environmental risk factors and CHD, including maternal nutrition deficiency during the first trimester of pregnancy. 6 A recent animal study revealed a potential association between maternal ID and the development of CHD in the fetus. 10 However, the clinical studies exploring this relationship are limited and controversial.
Our study revealed that the mothers whose infants have CHD had lower levels of SF, Fe, and TSAT and higher TIBC than mothers of infants without CHD. The results also suggested that the mothers of infants with CHD are more likely to have ID, which was similar to the result observed in a previous case–control study by Yang et al. 11 In a study by Chou et al., 19 it was suggested that maternal anemia increases the risk of CHD in the fetus. The previous study by Yang et al. also suggested that the mothers of infants with CHD are less likely to have an iron-rich diet and take iron supplements during pregnancy compared with controls. 11 Another study on maternal diets and CHD showed that an iron-rich diet during pregnancy might have a protective effect against CHD. 20
However, a study demonstrated a correlation between high maternal iron status and the risk of CHD in the infant. 12
Low SF levels without compensation from iron supplements may indicate significant ID. Therefore, ID in the mothers of infants with CHD after delivery is indicative of ID during pregnancy. 21
During the postpartum period, women are typically regarded as being at lower risk of ID. This belief stems from the assumption that iron stores are replenished following delivery because of a decrease in blood cell mass and a delayed return to menstruation. However, contrary to conventional knowledge, several studies have shown that iron stores, as measured by SF levels, can remain deficient for up to 6 months postpartum in women who did not receive iron supplementation during their pregnancy. 22
We found that cyanotic CHD infants were more likely to have ID than acyanotic CHD and control infants. This could be attributed to persistent hypoxia, which induces secondary erythrocytosis and subsequently increases the consumption of iron stores.23,24
Infants with CHD who were aged <6 months exhibited a lower average SF level (11.04 ng/mL) than control infants of the same age (55.45 ng/mL). However, this difference was not statistically significant (P = 0.219).
Furthermore, we found a positive correlation between the SF levels of infants aged <6 months and their mothers in the case group (P < 0.001, r = 0.565) and control group (P = 0.043, r = 0.547). This suggests that ID in infants may be a result of maternal ID during pregnancy. At this age, infants rely on iron stores accumulated during pregnancy; moreover, if the mother has ID, the infant is at increased risk of developing ID.25,26
Iron is crucial in embryogenesis, and ID during early pregnancy may affect heart development through multiple possible mechanisms.
First, the myocardium is a highly metabolic tissue, and cardiogenesis involves controlled, energy-dependent processes, including the proliferation, differentiation, and maturation of cardiomyocytes.27,28 The energy needed for cardiogenesis is primarily obtained from mitochondrial oxidative phosphorylation, which relies on iron-dependent enzyme complexes.27,29
Iron is a crucial component in mitochondria, as Fe-protoporphyrin, Fe-S clusters, and Rieske center are involved in the mitochondrial complexes that mediate electron transportation and energy generation. 29
Therefore, ID can reduce energy production and negatively affect cardiomyocyte differentiation and maturation, leading to CHD.30,31 ID seems to be (at least partly) the missing part that links the metabolic abnormalities reported in previous studies.
Second, ID is associated with disruption of mitochondrial biogenesis, changes in mitochondrial structure, increased release of cytochrome c, and induction of apoptosis. Therefore, ID may play a role in mitochondria dysfunction. 32 This could explain the results of previous studies suggesting that mitochondrial dysfunction and metabolic disturbance contribute to CHD development.33,34
Third, cardiogenesis processes are regulated by various transcription factors, genes, and signaling pathways, all of which are influenced by genetic and environmental factors. 4 ID may reduce the activity of the iron-dependent enzyme CYP26, which could result in increased retinoic acid signaling in the second heart field. This could, in turn, activate the cardiac transcription factor GATA4 and cause premature differentiation of a group of cardiac progenitor cells, leading to CHD. 10
ID can also lead to reduced activity of another iron-dependent enzyme, endothelial nitric oxide synthase (eNOS). 35
The inhibition of eNOS activity may negatively affect the expression of cardiac-specific genes, impeding the differentiation of cardiac progenitor cells and thus the development of CHD. 36 In addition, iron can interact with other nutrients, such as folic acid, through intricate mechanisms. A study on animals demonstrated that low iron levels may impact the body's utilization of folate, even with sufficient folate intake. 37 A meta-analysis revealed that the use of folic acid supplements during pregnancy reduces the risk of CHD in the offspring. 38 Furthermore, a case report noted a synergistic interaction between folic acid and iron intake during pregnancy, leading to a reduced risk of CHD in the offspring. 39
This study provides evidence of a possible correlation between maternal ID and the risk of congenital heart disease. However, our study has certain limitations that need to be acknowledged. After reviewing the data of CHD infants attending the two hospitals, we observed that no cases were diagnosed during pregnancy. Due to the delay in diagnosis until after birth, we collected maternal blood post-delivery. This timing of sample collection could impact the assessment of iron levels in our study. The iron-related biomarkers collected during the post-partum period may not accurately reflect the parameters in early pregnancy, during which the heart is formed. We also did not achieve the required initial number of cases and controls, which may have compromised the accuracy of the statistical analysis. Additionally, it is important to note that we cannot rule out the possibility of residual confounding.
Considering the external validity, we randomly selected a representative sample from the study population. To increase the generalizability of the study results, it is recommended to conduct the study with a larger sample size.
Conclusion
Our study indicates that the mothers of infants with CHD have lower iron status than those with infants without CHD. Additionally, mothers of infants with CHD are more likely to have ID. It is essential to conduct prospective clinical studies with a substantial number of pregnant women to confirm our results and gain a deeper understanding of the underlying mechanism.
Footnotes
Acknowledgment
I would like to thank the medical and nursing staff at the Pediatrics’ University Hospital for their assistance during sample collection as well as the staff at the Pediatrics’ University Hospital Laboratory for facilitating sample analysis.
Authors’ contributions
RQ collected and analyzed the data and wrote the manuscript. RA designed the study and supervised data analysis. RQ and RA interpreted the results, revised the manuscript, and approved the paper.
Availability of data and materials
All materials and data generated during this study are included in this article.
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors declare that they have no conflicts of interest.
Ethics approval and consent to participate
This study was approved by the Biomedical Research Ethical Committee (BMREC) at Damascus University, and the reference number is PH-290122-30. The study was conducted according to the guidelines of the Declaration of Helsinki of 1975 as revised in 2024, and all mothers provided written informed consent.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
