Abstract
Objective
This study evaluated the therapeutic efficacy of Lactobacillus in women with cervical human papillomavirus infection. Specifically, it aimed to assess the impact of Lactobacillus therapy on human papillomavirus clearance and cervical lesion resolution in clinical settings.
Methods
A literature search was conducted in October 2024 using PubMed, EBSCO, Google Scholar, and ScienceDirect. Eligible studies included randomized controlled trials and quasi-randomized controlled trials investigating Lactobacillus therapy for human papillomavirus infection. This review followed the Preferred Reporting Items for Systematic Review and Meta-Analysis guidelines. Risk of bias was assessed using the risk of bias 2.0 and ROBINS-I (Risk Of Bias In Non-randomized Studies – of Interventions) tools. Data were analyzed using a random-effects model with 95% confidence intervals, and heterogeneity was evaluated using the I2 statistic. A subgroup analysis was performed based on the method of administration (oral vs. vaginal).
Results
Five studies were included in the meta-analysis. Among 326 participants assessed for lesion resolution, Lactobacillus therapy showed a statistically significant improvement (relative risk = 1.93, 95% confidence interval: 1.47–2.53, p < 0.00001; I2 = 0%). Similarly, among the 262 participants assessed for viral clearance, the treatment showed a statistically significant effect (relative risk = 1.39, 95% confidence interval: 1.00–1.94, p = 0.05; I2 = 33%).
Conclusion
In patients with low-grade squamous intraepithelial lesions/atypical squamous cells of undetermined significance cervical lesions, Lactobacillus therapy administered over a minimum duration of 6 months may serve as an adjuvant treatment for human papillomavirus infection.
Keywords
Introduction
Human papillomavirus (HPV), belonging to the family Papillomaviridae, is a non-enveloped circular double-stranded DNA virus. 1 More than 230 types of HPV have been fully described, and new types continue to be identified. 1 HPV is categorized into two groups based on its cancer-causing potential: high-risk (HR) HPV and low-risk (LR) HPV. The International Agency for Research on Cancer has classified HPV types 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, and 59 as HR-HPV (group 1). 2 Additionally, HPV type 68 is classified as probably carcinogenic (group 2A), while HPV types 26, 53, 66, 67, 69, 70, 73, and 82 are considered possibly carcinogenic (group 2B). 2 Strong evidence indicates that LR-HPV types (group 4: HPV types 6, 11, 40, 42, 43, 44, 55, 61, 81, and 83) do not cause cancer. 2 LR-HPV types cause warts on cutaneous and genital areas, while HR-HPV types are associated with oropharyngeal and anogenital cancers, including anal, vaginal, vulvar, cervical, and penile cancers. 3 After lung cancer, colorectal cancer, and breast cancer, cervical cancer ranks fourth among cancers that affect women globally.4,5 In 2022, the World Health Organization reported approximately 350,000 deaths from cervical cancer and 660,000 incident cases worldwide. 6 Approximately 10% of sexually active women with normal cytology may currently have genital HPV infection. 7 By the age of 45 years, up to 80% of sexually active women are estimated to have been exposed to HPV. 7 Although most infected individuals are asymptomatic and clear the infection spontaneously while maintaining normal cytology, some may develop precancerous or cancerous lesions. 7
Recently, a possible association has been described between vaginal dysbiosis and pathobiosis in promoting cervical HPV infection. Healthy women have a complex vaginal microflora characterized by a balanced population of anaerobic and aerobic microorganisms—a state known as eubiosis. 8 Lactobacillus species dominate the vaginal and cervical microbiomes in healthy women. Under certain conditions, this balance may be disrupted (dysbiosis), allowing other microorganisms to proliferate. 8 The loss of this microbial stability may contribute to the incidence of sexually transmitted diseases. 8 Some studies have suggested that certain Lactobacillus strains, such as Lactobacillus reuteri, L.cripatus, and L. rhamnosus, exert inhibitory effects on HPV infection.9,10 Lactobacillus exerts anticancer effects by preventing pathogenic bacterial adhesion, producing bioactive metabolites that suppress tumors, and modulating immune responses. 11
Several noninvasive strategies for treating HPV infection have been evaluated in both preclinical and clinical studies.9,12 One such strategy is the administration of probiotics, such as Lactobacillus. Although some clinical studies have reported the effectiveness of Lactobacillus therapy in promoting HPV clearance and lesion resolution, the findings remain inconsistent. Notably, Lactobacillus can be administered either orally or intravaginally, each exhibiting a distinct mode of action. Oral probiotics may influence HPV infection by modulating the gut microbiota, which, in turn, affects systemic immunity and interacts with the vaginal microbiota through the gut–vaginal axis. In contrast, vaginal administration allows for direct colonization of the cervical and vaginal mucosa.12,13
To address this knowledge gap, the present study conducted a systematic review and meta-analysis to evaluate current evidence on the effectiveness of Lactobacillus therapy in enhancing HPV clearance and cervical lesion resolution. In particular, a subgroup analysis was performed to compare clinical outcomes between oral and vaginal administration. This analysis is essential for formulating optimal therapeutic strategies and informing future research directions.
Materials and methods
Method
This systematic review and meta-analysis were conducted according to the Preferred Reporting Items for Systematic Review and Meta-Analysis protocols. This study is filed on PROSPERO under the registration number CRD42024610320.
Search strategy
This study included literature published until 31 October 2024. The literature search was completed in October 2024 from the following four electronic databases: PubMed, EBSCO, Google Scholar, and ScienceDirect. We formulated the research question using the PICO (Population, Intervention, Comparison, and Outcome) framework to ensure clarity. The population of interest was women with cervical HPV infection (negative for intraepitelial malignancy). The intervention was Lactobacillus therapy, administered either orally or vaginally. This intervention was compared with either placebo, observation only, or other treatment. The outcome was clearance of HPV infection (based on polymerase chain reaction (PCR)-negative result or Papanicolaou (PAP) test-negative result). The search terms were as follows: ((((human papilloma virus) OR (HPV)) OR (papilloma*)) AND ((((cervical) OR (cervix)) OR (genital)) OR (vagina*))) AND ((probiotic*) OR (lactobacil*)). This study also conducted a manual search technique to prevent the inclusion of unqualified papers. Two authors (LAC and YWW) independently assessed the titles and abstracts for duplication and eligibility. The relevant articles were subjected to full-text screening. When discrepancies occurred, decisions were reconciled by consulting with a third author (DAS) to determine the suitability of the full-text article.
Inclusion and exclusion criteria
The inclusion criteria were as follows: (a) studies involving women of all ages who were diagnosed with cervical or vaginal HPV infection, irrespective of subtypes; (b) studies involving cervical smear test results that were negative for high-grade intraepithelial lesion or malignancy; (c) experimental studies with noninvasive treatment (oral and topical administration of Lactobacillus); (d) full-text articles written in English; and (e) experimental study designs (randomized controlled trials (RCTs) or quasi-experimental studies) were prioritized, with observational cohort studies considered alternatives for reviewing therapeutic studies.
The exclusion criteria were as follows: (a) studies that did not report the cervical or vaginal HPV infection or only reported cervical cancer; (b) studies involving nonhuman subjects; and (c) studies that did not report Lactobacillus as the treatment option for HPV infections.
Quality assessment and data extraction
Two authors (SRP and YWW) independently assessed the risk of bias in each included study. The Cochrane risk of bias tool was used to assess RCTs, and the ROBINS-I (Risk Of Bias In Non-randomized Studies – of Interventions) tool was employed to assess quasi-RCTs. 14 When discrepancies occurred, decisions were reconciled by consulting with a third author (DAS) to determine the article’s suitability.
Data collected included authors, year of publication, study design, country, number of participants, HPV testing, cervical lesion type, cervical lesion diagnosis, Lactobacillus species, dose of Lactobacillus, route and duration of administration, duration of follow-up, other medication involvement, and HPV clearance outcome. All data were summarized independently as descriptive data by two authors (SRP and YWW). The other two authors (DAS and LAC) verified the extraction findings.
The meta-analyses were conducted independently using Review Manager software by two authors (DAS and LAC). The Mantel–Haenszel’s method with random-effect meta-regression was used to calculate the risk ratio and 95% confidence interval (CI). Higgins’ I2 was used as a quantitative parameter for determining heterogeneity in this study. The results were presented as a forest plot. Additionally, the publication bias was evaluated using funnel plots. The Egger’s test was performed when the funnel plot asymmetry was used to measure the bias, and the result was considered significant when the p-value was <0.05.
Results
The literature search was completed on 30 October 2024. A total of 514 articles were identified through database searches: PubMed (286 articles), EBSCO (82 articles), ScienceDirect (130 articles), and Google Scholar (16 articles). After removing duplicates, 403 articles remained for title and abstract screening, resulting in 22 articles selected for full-text examination. Ultimately, seven studies8–10,15–18 met the inclusion and exclusion criteria and were included in the systematic review analysis, with five of them eligible for meta-analysis.8–10,16,18 A flowchart of the procedure is shown in Figure 1.
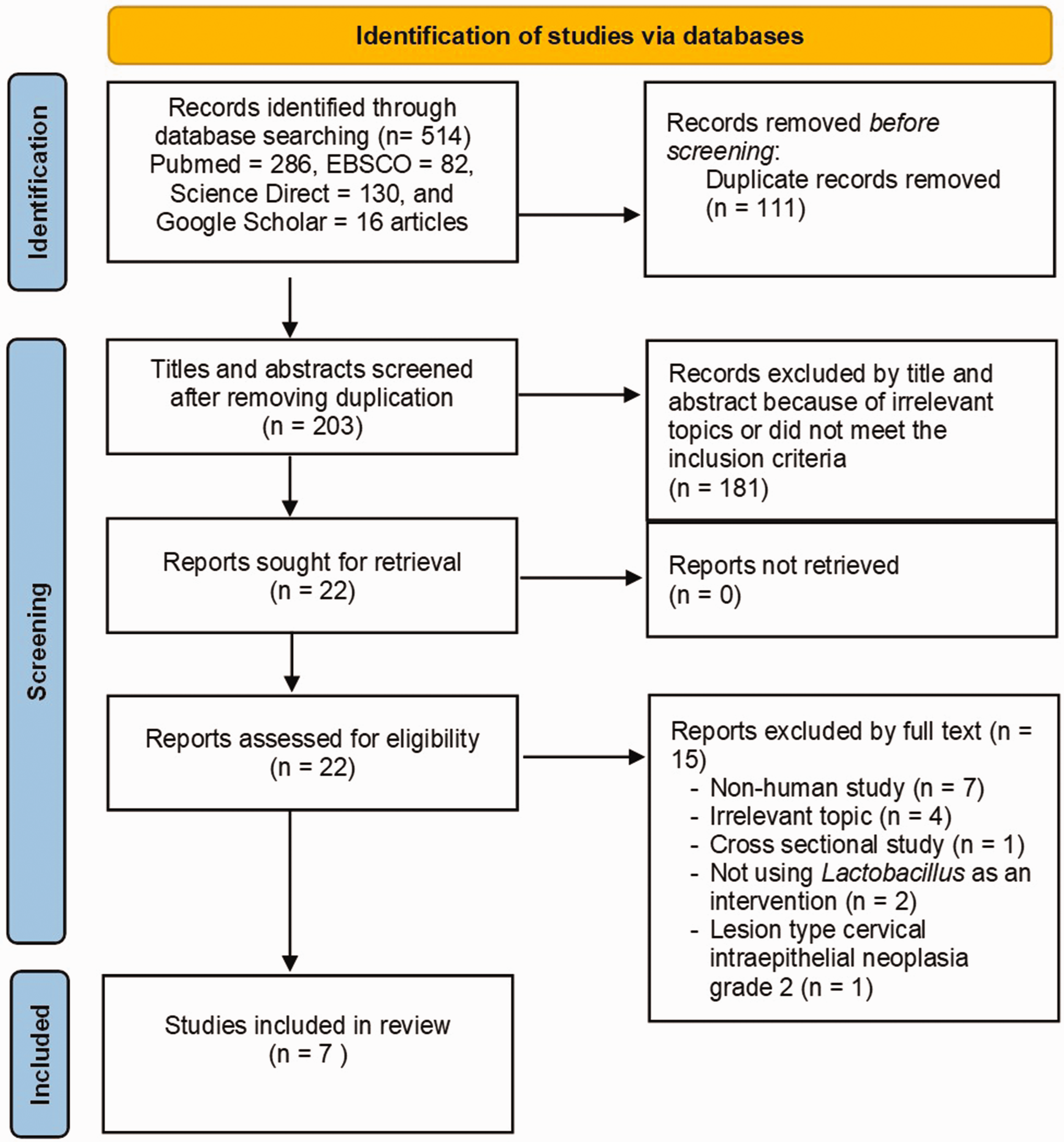
Flowchart of the PRISMA diagram. PRISMA: Preferred Reporting Items for Systematic Review and Meta-Analysis.
Baseline characteristics of the included studies
This systematic review summarized the outcomes of 7 studies (Table 1), comprising 712 participants with cervical HPV infection with atypical squamous cells of undetermined significance/low-grade squamous intraepithelial lesions (ASCUS/LSIL) lesion types who received Lactobacillus treatment.8–10,15–18 Two studies were RCTs,8,9 four studies were quasi-RCTs,10,15,16,18 and one study was a prospective uncontrolled pilot study. 17 However, only five studies were included in the meta-analysis.8–10,16,18 The baseline characteristics extracted from each study included the authorship, year of publication, study location, study design, sample size, mean participant age, HPV testing method, lesion type, treatment duration, Lactobacillus strain and dosage, route of administration, follow-up duration, and primary outcomes. These data are summarized in Table 1.
Characteristics of included studies.

HPV: human papillomavirus; ASCUS: atypical squamous cells of undetermined significance; LSIL: low-grade squamous intraepithelial lesion, RCT: randomized controlled trial; PCR: polymerase chain reaction; mo: months; CFU/sach: colony-forming units per sachet; CFU/tab: colony-forming units per tablet.
Risk of bias assessment
Some studies described their design as randomized, but their classification as randomized or nonrandomized remained uncertain. Therefore, we categorized them as non-RCTs.10,16,18 In this study, we assessed the quality of three quasi-RCTs10,16,18 using the ROBINS-I tool 19 and two RCTs8,9 using the ROB 2.0 tool. 19 Based on these assessments, two quasi-RCTs16,18 were found to have a low risk of bias, one quasi-RCT 10 had a moderate risk of bias, one RCT 8 had some concerns regarding the risk of bias, and one RCT 9 had a low risk of bias. The risk of bias assessed using ROBINS-I is presented in Table 2, and that assessed using ROB 2.0 is shown in Table 3.
Risk of bias assessment in quasi-RCTs using ROBINS-I tool. 19

Domains: D1: Bias due to confounding; D2; Bias due to selection of participants; D3: Bias in classification of interventions; D4: Bias due to deviations from intended interventions; D5: Bias due to missing data; D6: Bias in measurement of outcomes; D7; Bias in selection of the reported result.
Judgment: −: moderate; +: Low.
RCT: randomized controlled trial; ROBINS-I: Risk Of Bias In Non-randomized Studies - of Interventions.
Risk of bias assessment in RCTs using the ROB 2.0 tool. 19
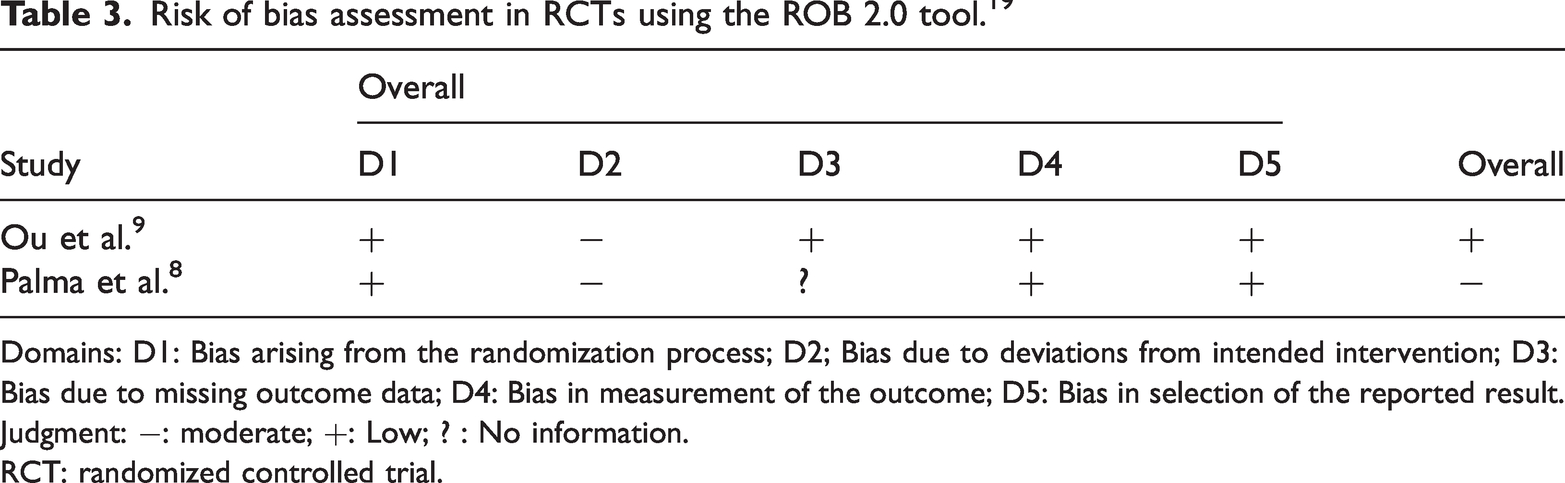
Domains: D1: Bias arising from the randomization process; D2; Bias due to deviations from intended intervention; D3: Bias due to missing outcome data; D4: Bias in measurement of the outcome; D5: Bias in selection of the reported result.
Judgment: −: moderate; +: Low; ? : No information.
RCT: randomized controlled trial.
Oral Lactobacillus
The oral Lactobacillus intervention was examined in 4 studies involving 367 patients.9,16–18 In these studies, various Lactobacillus species were administered orally, including L. casei Shirota (Yakult), L. crispatus M247, and a combination of 50% L. rhamnosus GR-1 and 50% L. reuteri RC-14.
In 2 studies,16,17 L. crispatus M247 was administered orally to a total of 160 participants with ASCUS/LSIL-type cervical lesions, using a dose of 20 billion colony-forming units (CFU) per day for 12 months. Cytological clearance evaluated at 6 months showed no significant difference between the treatment and control groups. However, by month 12, the difference became statistically significant (61.5% vs. 41.3%), although viral clearance remained nonsignificant. 16 In a preliminary noncontrolled study involving 35 participants who received the same oral dose of L. crispatus M247 for 3 months, 71% of the participants exhibited HPV clearance at the 3-month evaluation. 17
For oral administration of L. casei Shirota (8 × 109 CFU/day) over a 6-month period, viral clearance at months 3 and 6 showed no significant difference; however, cytological clearance at month 6 revealed a statistically significant improvement in the treatment group compared with that in the untreated group (60% vs. 30%). 18 Similar findings were reported in a study involving 121 participants who received a combination of 50% L. rhamnosus GR-1 and 50% L. reuteri RC-14 (180 mg; 5.4 billion CFU/tablet). Viral clearance was assessed at months 3, 6, 9, and 12. During the 12-month follow-up no significant differences in viral clearance were observed between the intervention and control groups across all time points. Interestingly, although lesion resolution at month 3 showed no significant differences between the two groups, a significant improvement was observed in the intervention group at month 6; however, this effect was not sustained at months 9 and 12. 9
Vaginal Lactobacillus
In three clinical studies, Lactobacillus was administered vaginally.8,10,15 Vaginal administration of L. crispatus Chen-01 was provided to 100 participants with ASCUS/LSIL-type cervical lesions for 5 months, and the results were evaluated in the sixth month. The outcomes were similar to those observed with oral administration of L. casei Shirota and L. crispatus M247, showing no notable difference in HPV clearance but demonstrating a significant difference in lesion resolution. 10 Although no significant difference was observed in viral clearance, the study assessed viral load in both intervention and control groups before and after treatment. The results indicated a notable difference in viral load between the two groups (L. crispatus Chen-01) at the 6-month evaluation. 10
A total of 117 participants with confirmed bacterial or yeast infections and concurrent HPV infection—identified through PCR analysis or abnormal Pap smears—received targeted treatment based on their conditions. They were also supplemented with vaginal L. rhamnosus BMX54 at a dosage of 104 CFU/tablet for either short (3 months) or long (6 months) durations. The evaluation was conducted in the ninth month. The study found a significant difference in cytological and viral clearance between participants treated for 3 months and those treated for 6 months. 8
Additionally, a study involving 128 HR-HPV participants treated with a combination of Lactobacillus vaginal capsules (H10980293) and interferon alpha-2B gel for 3 months demonstrated significant HR-HPV clearance compared with that in a group receiving only recombinant interferon alpha-2B gel. A notable difference in viral load reduction was also observed between the two groups. This evaluation was conducted at 6 months. 15
Meta-analysis
Five studies were included in this meta-analysis. Of these, two were RCTs8,9 and three were quasi-RCTs.10,16,18 The meta-analysis assessed the effectiveness of oral and vaginal Lactobacillus therapy on HPV clearance and cervical lesion resolution. The evaluation period for all included studies was at least 6 months. The characteristics of the included studies are presented in Table 1.
Cervical lesion resolution
The assessment of cervical lesion resolution was based on five studies, all of which included a minimum follow-up period of 6 months. Specifically, four studies reported evaluations at 6 months,9,10,16,18 and one study reported evaluation at 9 months. 8 A total of 326 patients were included in these studies (163 each in the intervention and control groups). The random-effects model revealed a statistically significant improvement in lesion resolution in the intervention group (relative risk (RR) = 1.93, 95% CI: 1.47–2.53, p < 0.00001; I2 = 0%). These results are illustrated in Figure 2.

Forest plot for Lactobacillus therapy with lesion resolution outcome.
A subgroup analysis was also conducted to examine the effect of the route of administration on lesion resolution. In the oral administration group (three studies9,16,18 that conducted evaluation at 6 months; 193 participants: 96 in the intervention group and 97 in the control group), there was no significant difference between the two groups (RR = 1.51, 95% CI: 0.96–2.38, p = 0.07; I2 = 0%). In contrast, the vaginal administration group, which consisted of 133 patients who were evaluated at 6 10 and 9 months 8 (67 in the experimental group and 66 in the control group), showed a significant difference in lesion resolution after the 6-month evaluation (RR = 2.21, 95% CI: 1.57–3.11, p < 0.00001; I2 = 0%), as illustrated in Figure 2.
A funnel plot was also used to assess publication bias. Overall, the funnel plot showed symmetry between the left and right sections. However, a subgroup analysis revealed asymmetry, with the vaginal studies clustered to the right and the oral studies to the left of the line of effect. There were no studies with small sample sizes, as only a limited number of original articles were included in this analysis. The relatively small number of currently published studies contributes to this limitation. Nonetheless, the funnel plot indicated no evidence of publication bias. These findings are presented in Figure 3. This visual assessment was supported by Egger’s test, which yielded a p-value of 0.452 (>0.05), further indicating no significant publication bias (Figure 4).

Funnel plot for Lactobacillus therapy with lesion resolution outcome.

Egger’s test for lesion resolution outcome.
HPV clearance
A comprehensive analysis of five studies (two RCTs and three quasi-RCTs) was conducted to assess viral clearance.8,10,15,16,18 Due to the limited number of available studies, the analysis of viral clearance in this review focused on those with a follow-up period of at least 6 months. Specifically, two studies reported evaluations at 6 months,10,18 one study at 9 months, 8 and two studies at 12 months.9,16 A total of 262 patients were included in the experimental group and 267 in the control group. The experimental group consisted of patients treated with Lactobacillus species, whereas the control group included participants from two observational-only, two placebo, and one active control study who received Lactobacillus therapy for a short duration (3 months). The pooled data showed a statistically significant difference in HPV clearance between the experimental and control groups using a random-effects model (RR = 1.39, 95% CI: 1.00–1.94, p = 0.05; I2 = 33%). The findings are illustrated in Figure 5.

Forest plot for Lactobacillus therapy with HPV clearance outcome. HPV: human papillomavirus.
An additional subgroup analysis was performed based on the administration of Lactobacillus in relation to viral clearance. A study 16 conducted evaluation at 6 months, and two studies11,14 conducted evaluation at 12 months, involving 332 patients (166 each in the experimental and control groups) who were included in the oral Lactobacillus subgroup. The analysis indicated no statistically significant difference in HPV clearance between the oral Lactobacillus and control groups using a random-effects model (RR = 1.16, 95% CI: 0.86–1.54, p = 0.31; I2 = 0%). In the vaginal Lactobacillus group, which involved two trials (197 participants) that conducted evaluation at 6 months 10 and 9 months 8 , also demonstrated no significant difference between the experimental and control groups (RR = 1.76, 95% CI: 0.81–3.84, p = 0.15; I2 = 65%). The results are presented in Figure 5.
Although only a few studies were included in the viral clearance analysis, heterogeneity was assessed. The funnel plot was symmetrical, with an equal number of studies on both sides of the line of effect and an even distribution across large and small samples. This visual symmetry indicated an absence of publication bias, as shown in Figure 6. This visual assessment was supported by Egger’s test, which yielded a p-value of 0.332 (>0.05, further indicating no significant publication bias), as shown in Figure 7.
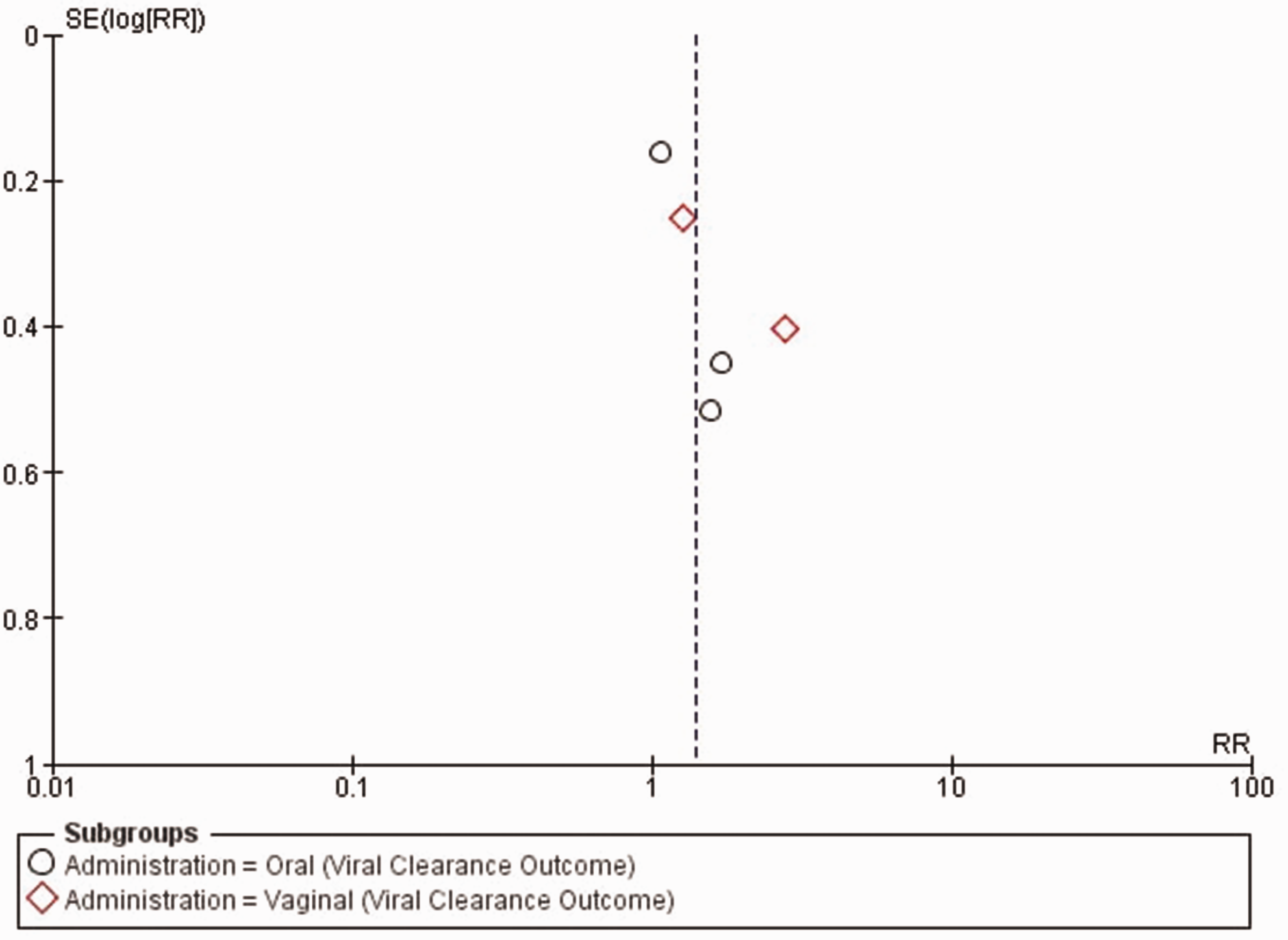
Funnel plot for Lactobacillus therapy with HPV clearance outcome. HPV: human papillomavirus.

Egger’s test for HPV clearance outcome.
Discussion
This study reviewed the effectiveness of Lactobacillus therapy for cervical HPV infection in patients with LSIL/ASCUS-type lesions, compared with placebo or observation only. Based on the risk of bias analysis, all studies included in the meta-analysis were of high quality. The funnel plot analysis indicated no evidence of publication bias in the included studies.
HPV progression and lesion types
This systematic review assessed the characteristics of women with cervical HPV infection, including both HR-HPV and LR-HPV types, as determined by PCR or DNA testing. Four studies8–10,15 focused on HR-HPV infections, whereas three studies16–18 included participants regardless of whether they were infected with HR-HPV or LR-HPV. Genital HPV types are classified as either HR (oncogenic) or LR (nononcogenic) based on their association with cervical cancer and its precursor lesions. 20 Although the majority of HPV infections are transient and typically resolve within 24 months, persistent infections may lead to the development of precancerous conditions. 21 Persistent HR-HPV infection significantly increases the risk of cervical intraepithelial neoplasia (CIN) and cervical cancer, ranging from CIN1 to CIN3. 20
A Pap smear is an essential diagnostic and screening tool to monitor the development of lesions. Abnormal Pap smear results include ASCUS, AGC (atypical glandular cells), LSIL, ASC-H (atypical squamous cells—cannot exclude high-grade squamous intraepithelial lesions (HSIL)), HSIL, AIS (adenocarcinoma in situ), and invasive squamous cell carcinoma or adenocarcinoma. 22 CIN is graded based on the extent of abnormal cell growth in the cervical tissue. CIN is histologically categorized into three grades: (a) CIN 1, indicating mild dysplasia; (b) CIN 2, moderate dysplasia; and (c) CIN 3, severe dysplasia or carcinoma in situ. 23 Lesions categorized as LSIL on a Pap smear generally correspond to CIN 1, whereas HSIL may reflect CIN 2 or CIN 3. 23 Although CIN 1 often resolves spontaneously, a cohort study showed that 1.5% of women with LSIL progressed to CIN 3 after 4 years of follow-up.9,24
Clinical efficacy of vaginal versus oral administration of Lactobacillus
In this study, given the variation in follow-up duration, Lactobacillus species, and clinical protocols across the included studies, a random-effects model was employed for the analysis. A total of 326 women diagnosed with ASCUS/LSIL-type lesions were included. The pooled data indicated that Lactobacillus treatment, when evaluated over a minimum duration of 6 months, was associated with significantly increased lesion resolution, with an RR of 1.93 (95% CI: 1.47–2.53, p < 0.00001; I2 = 0%), suggesting nearly twice the likelihood of lesion resolution compared with that in the control group. Although it remains unclear whether the restoration of normal vaginal flora can directly improve cervical smear cellularity, the influence of probiotics on smear quality is a promising area of investigation. Across three studies, HPV-infected participants with ASCUS/LSIL lesions demonstrated significant lesion resolution compared with controls at the 6-month evaluation.9,10,18 In addition, Perisić et al. 25 reported that L. rhamnosus GR-1 and L. reuteri RC-14 reduced the incidence of unsatisfactory and borderline cytological findings, thereby contributing to more reliable diagnostic outcomes. Although a definitive causal relationship has not been established, the findings suggest that Lactobacillus administration serves as a beneficial adjunct therapy in promoting lesion resolution, particularly for mild-to-moderate lesions such as ASCUS and LSIL.
This study is the first to assess the effectiveness of oral and vaginal Lactobacillus administration in patients with HPV infection presenting with ASCUS/LSIL lesions. The findings indicated that vaginal administration was significantly associated with HPV lesion resolution (RR = 2.21, 95% CI: 1.57–3.11, p < 0.00001; I2 = 0%), whereas oral administration showed a positive trend that did not reach statistical significance (RR = 1.51, 95% CI: 0.96–2.38, p = 0.07; I2 = 0%). This difference may be attributed to the distinct ways in which each administration route affects the local vaginal microbiota. Vaginal administration enables higher local concentrations of probiotics. 26 In contrast, orally administered probiotics are subject to digestive processes, such as exposure to acidic gastric fluids, which can destroy a substantial portion of the bacterial population in the upper gastrointestinal tract, potentially diminishing their effectiveness. In a study involving participants with bacterial vaginosis, intravaginal administration of Lactobacillus produced therapeutic effects within 2–3 days, compared with 7 days required for orally administered Lactobacillus to reach the vaginal area.27,28 Nonetheless, the gut–vaginal axis is a recognized pathway, and emerging evidence suggests that dysbiosis in the vaginal microbiota originates from the gut.29,30 Two studies revealed that patients with cervical cancer exhibited higher gut microbiota diversity than healthy women; although initial studies with small samples did not yield statistically significant results, larger sample studies demonstrated significant associations.16,30 Therefore, given the limited number of studies included in this review, along with variations in bacterial strains, dosages, and study populations, conclusions regarding the comparative efficacy of oral versus vaginal Lactobacillus administration on lesion resolution should be interpreted with caution. Further targeted research with larger, well-defined cohorts is necessary to draw more definitive conclusions.
The overall meta-analysis of viral clearance demonstrated that Lactobacillus may have the potential to enhance viral clearance, with an RR of 1.39 (95% CI: 1.00–1.94; p = 0.05; I2 = 33%). Although the CI includes the null value, this finding suggests a possible clinical benefit. Subgroup analysis by route of administration revealed that neither the oral nor the vaginal Lactobacillus groups showed statistically significant differences in viral clearance compared with that in the control group. However, the effect estimate in the vaginal subgroup (RR = 1.76) was higher than that in the oral subgroup (RR = 1.16). This interpretation should be approached with caution, as the effect size in the oral group, although modest, was consistent across studies (I2 = 0%), whereas the vaginal subgroup exhibited greater heterogeneity (I2 = 65%). These findings require further validation through studies with stronger methodological designs, larger sample sizes, and more standardized evaluation durations. Despite the limited number of studies, publication bias testing showed no evidence of bias (p > 0.05), and the funnel plot appeared symmetrical around the line of effect. Based on three studies, Lactobacillus administration demonstrated potential as an adjunctive therapy in promoting HPV clearance. Specifically, intravaginal administration 10 of L. crispatus CHEN-0110, intravaginal Lactobacillus capsules combined with topical interferon α-2B, 15 and oral administration of L. rhamnosus GR-1 combined with L. reuteri RC-14 9 were all associated with a significant reduction in viral load and an increased proportion of patients converting to HPV-negative status compared with controls.
Strain specificity, dosing, and treatment duration
Women’s reproductive health is influenced by the complex microbial community known as the cervical microbiota. 31 Lactobacillus predominates in the cervical microbiota of healthy women and those with transient HPV infections. In contrast, individuals with chronic HPV infection show markedly increased microbial diversity and a higher proportion of anaerobic bacteria. Hydrogen peroxide, lactate, and bacteriocins produced by Lactobacillus may inhibit the spread of pathogens throughout the genitourinary tract, which is essential for maintaining women’s reproductive health.32,33 These findings are supported by preclinical studies conducted by Borgdorff et al., 34 which demonstrated that certain Lactobacillus species act as protective factors against HPV infection by preserving the “cervical epithelial barrier,” thereby preventing HPV from penetrating basal keratinocytes.
Based on existing clinical studies, various Lactobacillus species were administered orally, including L. casei Shirota, 18 L. crispatus M247,16,17 and a combination of 50% L. rhamnosus GR-1 with 50% L. reuteri RC-14. 9 Oral administration of Lactobacillus varied in both dosage and duration. Two studies utilized L. crispatus M247 at the same dose of 20 billion CFU/day, but with different treatment durations—3 and 6 months, respectively.16,17 In the study by Verhoeven et al., 18 L. casei Shirota was administered orally at a dose of 8 × 109 CFU/day for 6 months. In the study by Ou et al., a combination of L. rhamnosus GR-1 and L. reuteri RC-14 (180 mg; 5.4 billion CFU/tablet) was given orally until the participants tested negative for HPV. 9 Vaginal administration was also investigated in several studies. Liu et al. 10 administered L. crispatus CHEN-01 vaginally at a dose of 1 × 109 CFU per capsule, with one capsule inserted daily for 14 consecutive days during the first 3 months. In the fourth month, it was administered once every 3 days, and in the fifth month, once every 6 days. In the study by Palma et al., 8 women with bacterial vaginosis or candidiasis coinfected with HPV and presenting with ASCUS/LSIL lesions received standard treatment, followed by vaginal administration of L. rhamnosus BMX 54 (104 CFU/tablet) for either 3 or 6 months. The dosing schedule was as follows: days 1–10, one tablet daily; days 11–30, one tablet every 3 days; months 2–3, one tablet every 5 days; and months 4–6, one tablet weekly. Additionally, in the study by Kong et al., 15 Lactobacillus vaginal capsules (H10980293, 0.25 g/capsule) were administered in combination with interferon alpha-2B gel. Lactobacillus was given at a dose of two capsules per administration, once every 2 days, across four cycles (one cycle consisting of seven administrations).
Two studies included in this review used L. rhamnosus as part of the treatment regimen: one employed a combination of 50% L. rhamnosus GR-1 and 50% L. reuteri RC-14, 9 whereas another used L. rhamnosus BMX 54. 8 L. rhamnosus is among the most extensively studied strains owing to its beneficial effects on the vaginal microbiota. Strains such as L. rhamnosus GR-1 and CA15 (DSM 33960) have demonstrated mechanisms of action that include the production of antimicrobial compounds—such as lactic acid, bacteriocins, and hydrogen peroxide—which effectively inhibit pathogenic growth.35,36 These strains also exhibit immunomodulatory effects by downregulating proinflammatory cytokines such as tumor necrosis factor-alpha (TNF-α) and interleukin (IL)-6 while enhancing anti-inflammatory responses through IL-10, thereby supporting local immune homeostasis.35,36 Among the included studies, two utilized L. crispatus administered orally, and one study used it via vaginal administration. L. crispatus produces both L- and D-isomers of lactic acid, which contribute to lowering vaginal pH and suppressing pathogenic organisms. Notably, D-lactate has been shown to be more effective in maintaining vaginal homeostasis. 37 This effect is partly attributed to its ability to inhibit matrix metalloproteinase-8 expression, thereby preventing HPV from accessing basal keratinocytes. It also modulates local innate immunity by inhibiting histone deacetylase activity and stimulating immune molecule expression. 10 In addition, L. crispatus produces bacteriocins and hydrogen peroxide (H2O2) and promotes an anti-inflammatory environment through T-cell activation and cytokine regulation. 37
As not all Lactobacillus strains exhibit the same effectiveness against HPV infection, selecting the appropriate strain based on clinical evidence is essential to achieve optimal outcomes. For example, oral administration of L. casei Shirota and the combination of 50% L. rhamnosus GR-1 with 50% L. reuteri RC-14 resulted in significantly improved cytological clearance compared with controls at the 6-month evaluation. In contrast, oral administration of L. crispatus did not yield statistically significant results over the same period.9,16,18 The duration of Lactobacillus administration also appears to be a critical factor influencing its effectiveness in HPV management. For instance, in the study by Palma et al., 8 intravaginal administration of L. rhamnosus BMX 54 for 6 months led to a higher HPV clearance rate than a 3-month course, as assessed at the 9-month evaluation. Similarly, in a study involving oral administration of L. crispatus for 12 months, no significant difference in cytological clearance was observed at 6 months; however, a significant improvement was noted at the 12-month evaluation. 16
Based on existing clinical studies, the dosage of Lactobacillus administered as an adjunct therapy for HPV infection varies widely, ranging from 104 to 1010 CFU per day. This variation depends on the specific bacterial strain used and the route of administration (oral vs. vaginal). Although standardized doses were not applied across studies, Gutiérrez Salmeán et al. 38 recommended the following for HPV-related outcomes: for prevention or clearance of HPV infection, vaginal administration of L. casei and L. rhamnosus at 104 CFUs or oral administration of L. crispatus at 108 to 1010 CFUs; for CIN regression and cervical cancer prevention, L. rhamnosus and L. crispatus at 108 to 1010 CFUs; and for antitumoral effects, oral doses between 108 and 1010 CFUs and vaginal doses of at least 104 CFUs of L. reuteri, L. crispatus, L. rhamnosus, L. plantarum, L. acidophilus, and L. gasseri. Reid et al. 39 and Andreu 40 also demonstrated that probiotic treatment with doses ranging from 109 to 1011 CFUs—administered orally or vaginally—can effectively restore the vaginal flora. Although both routes have proven effective for treating urogenital infections, the optimal route of administration remains undetermined. 41 Another study indicated that in certain cases of vaginal dysbiosis, the administration of a combination of L. crispatus (1 × 109 CFU), L. rhamnosus (1 × 109 CFU), L. jensenii (0.2 × 109 CFU), and L. gasseri (0.3 × 109 CFU) per dose—whether in the form of yogurt or capsules—may improve the vaginal microbial profile, as measured by the Nugent score. 42
Clinical implications and future directions
The results of this study are clinically relevant because ASCUS and LSIL lesions represent early stages that may regress spontaneously but can also progress to more severe lesions or cervical cancer if not managed properly. 10 Appropriate treatment may help inhibit the progression of HPV infection. This study showed that Lactobacillus can serve as an adjuvant therapy that increases the potential for lesion resolution and viral clearance. Longer durations of administration appear to yield better outcomes, but factors such as patient compliance, product availability, and preference for oral over vaginal routes should be considered. Further studies are needed to evaluate optimal treatment duration and address comfort, effectiveness, and compliance. This review is preliminary and should be expanded using more uniform protocols, strain types, dosages, and treatment durations. More RCTs and clinical studies are needed to confirm causal relationships and assess the long-term effects of Lactobacillus therapy.
Limitations
This review combined data from studies with follow-up durations of 6, 9, and 12 months, and differences in evaluation timing may have influenced the outcomes. The meta-analysis included a few RCTs, and several studies had small sample sizes. There was heterogeneity in probiotic strains, dosages, and intervention durations, which may have affected the consistency of the findings. In addition, although some proposed mechanisms suggest that Lactobacillus exhibits benefits against HPV, many are based on preclinical data and require further validation in human clinical studies. These findings should therefore be considered preliminary and require confirmation through larger, well-designed studies.
Conclusion
Based on evaluations with a minimum follow-up of 6 months, both oral and vaginal Lactobacillus administration may be used as adjuvant treatment for HPV infection in patients with LSIL or ASCUS lesions.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251363006 - Supplemental material for Effectiveness of Lactobacillus therapy in women with cervical human papillomavirus infection: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605251363006 for Effectiveness of Lactobacillus therapy in women with cervical human papillomavirus infection: A systematic review and meta-analysis by Devi Artami Susetiati, Satiti Retno Pudjiati, Yohanes Widodo Wirohadidjojo and Lukman Ade Chandra in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251363006 - Supplemental material for Effectiveness of Lactobacillus therapy in women with cervical human papillomavirus infection: A systematic review and meta-analysis
Supplemental material, sj-pdf-2-imr-10.1177_03000605251363006 for Effectiveness of Lactobacillus therapy in women with cervical human papillomavirus infection: A systematic review and meta-analysis by Devi Artami Susetiati, Satiti Retno Pudjiati, Yohanes Widodo Wirohadidjojo and Lukman Ade Chandra in Journal of International Medical Research
Footnotes
Acknowledgements
We would like to thank Universitas Gadjah Mada, Yogyakarta, Indonesia.
Authors contributions
DAS was responsible for the study concept, design, definition of intellectual content, literature search, data acquisition, data analysis, manuscript preparation, manuscript editing, manuscript review, and guarantor. SRP was responsible for the definition of intellectual content, data analysis, manuscript preparation, manuscript editing, manuscript review. YWW was responsible for the literature search, data acquisition, manuscript preparation, manuscript editing, and manuscript review. LAC was responsible for the literature search, data acquisition, data analysis, manuscript preparation, manuscript editing, and manuscript review.
Data availability statement
Data will be made available on request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
