Abstract
Linear growth delay is common in children with systemic lupus erythematosus. There have been scattered reports of using recombinant human growth hormone in these children, but safety remains a major concern. We herein report two cases of successful treatment with recombinant human growth hormone in children with systemic lupus erythematosus: a 15-year 10-month-old boy (case 1; disease duration: 4 years; height: 149 cm (<3rd percentile, Z-score <−3); and bone age: 11 years) and a 12-year 8-month-old boy (case 2; disease duration: 5 years; height: 149.5 cm (10th percentile, Z-score = −1); and bone age: 12 years). Both children had been in complete remission for 6 and 30 months, respectively. Insulin-like growth factor-1, insulin-like growth factor-binding protein 3, adrenocorticotropic hormone, and cortisol levels were normal in both cases. Recombinant human growth hormone treatment lasted for 17 and 16 months, respectively. The height increase was 22 and 19 cm during the treatment period, respectively. Systemic lupus erythematosus remained in complete remission on maintenance therapy with hydroxychloroquine/tacrolimus and hydroxychloroquine/mycophenolate mofetil, respectively. Literature search identified two cases. At the time of recombinant human growth hormone treatment initiation, case 1 had proteinuria and elevated serum creatinine level, and case 2 had positive anti-double-stranded DNA antibodies. Relapse occurred immediately after recombinant human growth hormone treatment in both cases. In conclusion, recombinant human growth hormone treatment may be considered for linear growth delay in children with systemic lupus erythematosus if disease activity is sufficiently controlled.
Background
The peak age of childhood-onset systemic lupus erythematosus (cSLE; 12–14 years) coincides with the second growth spurt in children. 1 The use of glucocorticoids during this period has a significant impact on development. Hence, cSLE is often associated with short stature. 2 Recombinant human growth hormone (rhGH) is widely used in the treatment of short stature caused by various conditions, including primary growth hormone (GH) deficiency, Prader–Willi syndrome, Turner syndrome, short stature homeobox-containing gene deficiency, and chronic renal insufficiency. 3 The impact of rhGH on the immune system remains a major concern in children with autoimmune diseases. However, a randomized controlled trial in children with juvenile idiopathic arthritis demonstrated improved final body height with rhGH treatment, with a good safety profile. 4 Herein, we report the cases of two children who underwent rhGH treatment for cSLE-associated short stature. Significant height growth was achieved in both cases; the primary disease remained in remission throughout the treatment period. We also conducted a literature search.
Case presentation
This study was approved by the Ethical Committee of the Children’s Hospital of Hebei Province (approval number: 2024180; date of approval: 2024-9-27). Informed consent for treatment and publication of this report was obtained from the parents in both cases. We have deidentified all patient details. The reporting of this study conforms to the Case Report (CARE) guidelines. 5
Case 1: A diagnosis of lupus nephritis (type III+V) was established in an 11-year 10-month-old boy based on symptoms, massive proteinuria, decreased C3 and C4 levels, positive anti-double-stranded (ds)-DNA and anti-nuclear antibodies, and renal biopsy. He experienced multiple episodes of relapse despite treatment with corticosteroids, cyclosporine A, mycophenolate mofetil, tacrolimus, and hydroxychloroquine sulfate until complete remission for over 6 months at the age of 15 years and 10 months. Proteinuria was negative upon repeated testing. The systemic lupus erythematosus disease activity index 2000 (SLEDAI-2k) score was 0 upon repeated assessment. Prednisone was administered as maintenance therapy at a dose of 5 mg every other day. His height was 149 cm (<3rd percentile [P3], Z-score <−3; 145 cm upon SLE onset, P25, Z-score =−1). The Tanner stage was I. The bone age based on wrist radiographs was 11 years. Insulin-like growth factor-1 (IGF-1), insulin-like growth factor-binding protein 3 (IGF-BP3), adrenocorticotropic hormone (ACTH), and cortisol levels were all within the normal range. Magnetic resonance imaging and tumor-related studies of the pituitary gland revealed no abnormality. Hepatic, renal, and thyroid function tests were unremarkable. rhGH treatment was initiated at a dosage of 0.15 unit/kg/day and lasted for 17 months. The height increase during the treatment period was 22 cm (Figure 1(a)). Upon the completion of rhGH treatment, the bone age was 14 years and the Tanner stage was II. Routine examinations every 3 months (including physical examination; thyroid function test; renal/hepatic function test; and assessment of fasting blood glucose, IGF1, and BP3 levels) were unremarkable. Prednisone was discontinued 1 month after the treatment, and the patient remained in remission with tacrolimus and hydroxychloroquine maintenance therapy. At the last follow-up at 18 years of age, the body height was 175 cm.

Growth charts in case 1 (a) and case 2 (b).
Case 2: A diagnosis of lupus nephritis (type III) was established in a 7-year 10-month-old boy based on symptoms, decreased C3 and C4 levels, positive anti-nuclear and anti-dsDNA antibodies, and renal biopsy. He experienced multiple episodes of relapse until complete remission for 30 months at the age of 12 years and 8 months. His height was 149.5 cm (P10, Z-score = −1; 135 cm upon SLE onset, P90, Z-score > 1). The growth rate over the past 6 months was 3.5 cm/year (<P25). The Tanner stage was I. The bone age based on wrist radiographs was 12 years. IGF-1, IGF-BP3, ACTH, and cortisol levels were within the normal range. Cranial MRI revealed no abnormality. Hepatic, renal, and thyroid function tests were unremarkable. rhGH treatment was initiated at a dosage of 0.12 unit/kg/day and lasted for 16 months, during which the body height increased by 19 cm (P75) (Figure 1(b)). Upon the completion of rhGH treatment, the Tanner stage was II and the bone age was 14 years. Routine follow-up (once every 3 months) revealed normal thyroid function, renal/hepatic function, fasting blood glucose, IGF1, and BP3. Corticosteroids and immunosuppressants were discontinued at the age of 17 years. He remained in remission during and after rhGH treatment until the final follow-up at the age of 17 years, when the body height was 178 cm.
Discussion and conclusions
A literature search of PubMed, Wanfang, and China National Knowledge Infrastructure (CNKI) databases identified two cases in which rhGH was used to treat short stature in children with histologically confirmed type IV lupus nephritis.6,7 Table 1 summarizes the key features of the two cases identified in the literature search versus the two index cases.
Summary of the cases.
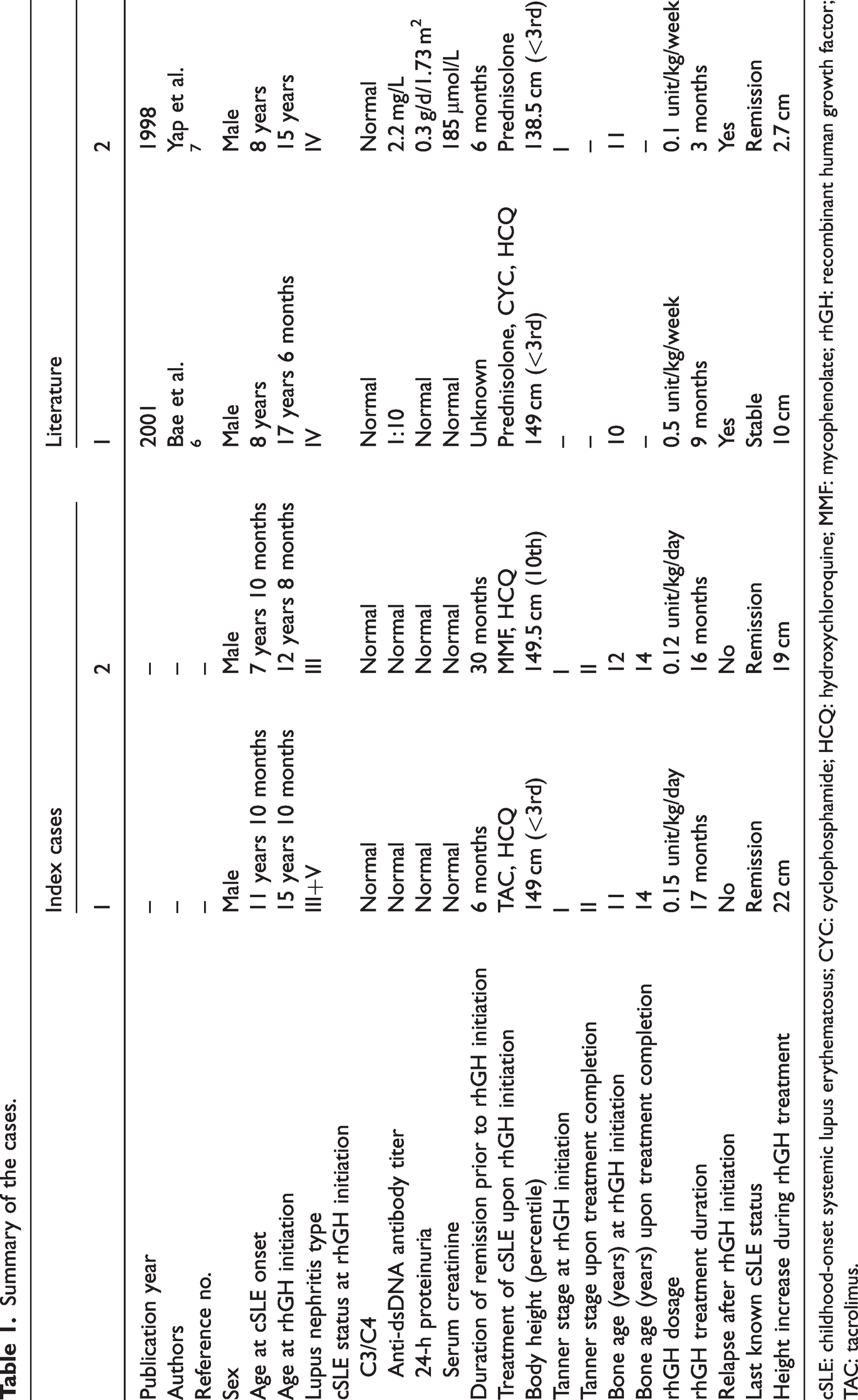
cSLE: childhood-onset systemic lupus erythematosus; CYC: cyclophosphamide; HCQ: hydroxychloroquine; MMF: mycophenolate; rhGH: recombinant human growth factor; TAC: tacrolimus.
In the first case, a Korean boy had had cSLE since the age of 8 years (Table 1). A renal biopsy at the age of 16 years revealed type IV lupus nephritis. At the time of rhGH treatment initiation (aged 17 years and 6 months), proteinuria was negative. Maintenance therapy included prednisolone (7.5 mg/day), hydroxychloroquine (400 mg/day), and cyclophosphamide (every 3 months). C3 and C4 levels were normal. Anti-dsDNA antibody was positive (1:10). The body height was 149 cm (<P3). The bone age was 10 years. rhGH treatment was started at a dosage of 0.5 unit/kg/week but was discontinued 9 months later due to proteinuria (1.5 g/day), decreasing C3/C4 levels, and increasing anti-dsDNA antibody titer (1:320). Prednisolone dosage was increased to 30 mg/day, but proteinuria continued to increase to a maximum level of 4.5 g/day. Prednisolone dosage was then increased to 1 mg/kg/day, and cyclophosphamide treatment (once per month) was initiated. At 10 weeks after rhGH discontinuation, proteinuria decreased to 1.8 g/day. The height increase during the 9-month treatment period was 10 cm.
In the second case, an Indian boy had had cSLE since the age of 8 years. A renal biopsy at the age of 10 years revealed type IV lupus nephritis. At 15 years of age, the body height was 138.5 cm (<P3, Z-score <−3). The bone age was 11 years. He was maintained on prednisolone (10 mg/day). rhGH treatment was initiated at a dosage of 0.1 mg/kg/day despite proteinuria (0.3 g/day/1.73 m2) and elevated serum creatinine (185 μmol/L). C3 and C4 levels were normal, and the anti-dsDNA antibody titer was 2.2 (normal reference range: <5.0) mg/L. Three months after treatment initiation, the erythrocyte sedimentation rate (ESR) increased to 40 mm/h, proteinuria increased to 0.4 g/day/1.73 m2, and the anti-dsDNA antibody titer increased to 7.0 mg/L. rhGH treatment was discontinued, but proteinuria continued to increase to a maximum of 1.1 g/day/1.73 m2. Prednisolone treatment was continued at a dose of 10 mg/day. The height increase during the treatment period was 2.7 cm. Six months after rhGH discontinuation, the ESR declined to 30 mm/h, proteinuria decreased to 0.6 g/day/1.73 m2, and C3/C4 levels returned to near normal.
cSLE is a systemic autoimmune disease that influences multiple organ systems. Compared with adult-onset SLE, cSLE is typically more invasive and thus is associated with higher mortality8,9 Glucocorticoids are the mainstay of cSLE treatment. The prolonged use of glucocorticoids, however, is associated with significant growth inhibition, including short stature and delayed sexual development.10,11 Growth retardation has been reported at very low dosages and even when glucocorticoids are inhaled. 12
The use of rhGH in children with autoimmune diseases is extremely limited due to concerns regarding its impact on the immune system.
In the two index cases, cSLE remained in complete remission for over 6 months. In contrast, the child in the second case in the literature had 0.3 g/day/1.73 m2 proteinuria and elevated serum creatinine upon the initiation of rhGH treatment. 6 In the first case in the literature, proteinuria was negative upon rhGH treatment initiation for an unknown period, but the anti-dsDNA antibody was positive. In our opinion, treatment success with rhGH in the two index cases is related to sustained disease remission prior to rhGH treatment. In contrast, disease activity was not sufficiently controlled prior to rhGH treatment in the two cases reported in the literature. Another consideration is the advancements in cSLE treatment over the past decades. At the time of the two cases reported in the literature (both more than two decades earlier), treatment options for cSLE were limited and included glucocorticosteroids, hydroxychloroquine, and cyclophosphamide. In contrast, cSLE treatment in the two index cases included glucocorticosteroids, hydroxychloroquine, mycophenolate mofetil, and calcineurin inhibitors. The use of immunosuppressants is instrumental in improving disease control and thus creates conditions allowing for rhGH treatment.
In summary, a significant increase in body height was achieved with the use of rhGH without inducing relapse in two patients with controlled disease activity for at least 6 months. Owing to the potential publication bias, we believe that caution must be exercised in case selection for rhGH treatment. Moreover, the two comparison cases were from an era of now-outdated immunosuppressive regimens.
Footnotes
Acknowledgments
We thank the parents for providing informed consent to publish this material.
Authors’ contributions
Data curation: Xiaoying Yuan, Yanjun Yang, Lanlan Ge, Jingxiao Guo, Fujuan Liu; Investigation: Fujuan Liu, Yanan Han, Chunzhen Li; Writing—review & editing: Fujuan Liu, Dongfeng Zhang, Ling Liu.
Consent for publication
Not applicable.
Competing interests
None.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on request.
Ethics approval and consent to participate
This study was approved by the Ethical Committee of the Children’s Hospital of Hebei Province. Informed consents were obtained from the participants’ legal guardian.
Funding
None.
