Abstract
Systemic lupus erythematosus is a chronic autoimmune disease often presenting with cutaneous symptoms such as malar rash. While hydroxychloroquine is standard therapy, some cases remain treatment-resistant. Molecular hydrogen possesses anti-inflammatory and immunomodulatory effects in autoimmune disorders. This case report examines the use of molecular hydrogen as adjunctive therapy in a 27-year-old woman with systemic lupus erythematosus and refractory malar rash. Despite hydroxychloroquine treatment, her rash persisted and significantly impacted the quality of life. After initiating oral molecular hydrogen therapy, the patient showed sustained clinical improvement, including reduced malar rash, decreased fatigue, and enhanced well-being over several months. These outcomes were accompanied by notable immunological changes: increased killer cell lectin-like receptor G1 positive effector T cells, decreased plasmablast activity, stabilization of B cell exhaustion markers, and reductions in and a decline in antinuclear antibody and anti-double-stranded DNA antibody levels. This case suggests that molecular hydrogen may be a safe and promising adjunctive treatment for cutaneous manifestations and systemic immune modulation in systemic lupus erythematosus.
Introduction
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease of global prevalence, capable of affecting virtually any organ or tissue in the human body. 1 The clinical course of SLE is often unpredictable, with alternating periods of remission and acute disease flares. 2 This complexity, combined with the diverse spectrum of organ involvement, presents significant challenges in both diagnosis and long-term management. 3 Cutaneous manifestations are a frequent and clinically significant aspect of SLE, affecting up to 80% of patients at some point during the disease course. 4 In ~25% of cases, skin involvement may serve as the initial presenting symptom. 5 The diagnostic importance of cutaneous findings is underscored by the inclusion of four skin-related criteria, malar rash, discoid rash, photosensitivity, and oral ulcers, among the eleven classification criteria established in the 1997 American College of Rheumatology criteria for SLE diagnosis. 6 Given the high prevalence and diagnostic relevance of cutaneous involvement, further research in this area is essential to improve disease understanding and management.
Serologically, antinuclear antibodies (ANAs) are a hallmark of SLE, present in the vast majority of patients and serving as a fundamental diagnostic tool. 7 Anti-double-stranded DNA (anti-dsDNA) antibodies, which exhibit greater specificity for SLE than ANAs, are strongly associated with disease activity, particularly lupus nephritis. 8 Hydroxychloroquine remains the first-line systemic therapy for SLE, including its cutaneous manifestations such as malar rash. This antimalarial agent has been shown to enhance treatment response, reduce disease flares, and improve overall survival in patients with SLE. 9 Recent studies have suggested that molecular hydrogen (H2) may have therapeutic potential in autoimmune and inflammatory diseases, including SLE, due to its antioxidant and immunomodulatory properties. 10 Multiple studies have demonstrated the therapeutic potential of H2 in various dermatological conditions, notably through its capacity to attenuate oxidative stress, modulate inflammatory pathways, and facilitate tissue repair and skin regeneration.11,12
Here, we present the case of a 27-year-old female with a well-established diagnosis of SLE who developed a persistent, cosmetically significant malar rash refractory to hydroxychloroquine treatment. Given the immunomodulatory potential of H2, we introduced it as an adjunctive therapy alongside hydroxychloroquine. The patient exhibited notable clinical improvement, accompanied by immunological changes, including increased killer cell lectin-like receptor G1 positive (KLRG1)+ effector T cells (Tc), reduced plasmablast activity, and stabilization of B cell exhaustion markers. These findings suggest a potential mechanism underlying the observed therapeutic response. This report details the clinical history of the patient, therapeutic interventions, and outcomes, providing insights into the potential role of H2 as an adjunct in refractory cutaneous SLE.
Case report
A 27-year-old female with SLE was first diagnosed at the age of 14 years, presenting with a malar rash and a positive ANA titer of 1:1280. Over the years, her disease remained relatively stable without immunosuppressive therapy. However, in August 2022, her ANA titer was 1:640, with normal complement C3 and C4 levels and low anti-dsDNA levels. Despite the absence of significant organ involvement, she continued to experience recurrent malar rash, significantly affecting her quality of life.
Between December 2022 and June 2023, her ANA titer remained at 1:640, while C3 levels fluctuated between 141.9 and 167.1 mg/dl. Complement C4 levels showed moderate elevations, peaking at 68.8 mg/dl in March 2023. Flow cytometry analysis revealed an increase in effector Th KLRG1+ and effector memory (EM) Tc KLRG1+ cells, indicating immune activation. Due to persistent malar rash, hydroxychloroquine therapy was initiated in March 2023 as the initial immunomodulatory approach. However, only partial improvement was observed, prompting the addition of H2 therapy in August 2024. By February 2024, laboratory findings showed a reduction in ANA titer to 1:160 and a decline in anti-dsDNA levels from 17 IU/ml (2023) to 7.3 IU/ml. Complement levels remained stable (C3: 127.0 mg/dl; C4: 54.2 mg/dl). Tc subset analysis demonstrated a low CD4/CD8 ratio (0.65), attributed to an increased CD8+ suppressor Tc population (42.7%). Following hydroxychloroquine treatment, the patient reported gradual improvement in rash severity and overall symptoms.
On August 7, 2024, hydrogen capsule therapy was introduced as an adjunctive treatment, with the patient receiving one capsule per day. Over the following months, significant immunological changes were observed. Effector Th KLRG1+ cells, which had previously declined, began to increase, peaking at 20% in February 2025 from a nadir of <5% in early 2024 (Figure 1). Similarly, EM Tc KLRG1+ cells, which had declined to 25% in February 2024, gradually increased to 60% by February 2025 (Figure 2), suggesting improved immune modulation. Natural regulatory T cells (Tregs), initially at 8% in August 2023, dropped to <2% in February 2024, but later stabilized at 3% in February 2025 (Figure 3). A notable reduction in plasmablasts was also observed, decreasing from a peak of 75% in August 2023 to 55% by February 2025 (Figure 4), aligning with reduced autoimmune activity.

Timeline of the patient’s progression of immune-related diseases and associated medication treatments.

KLRG1+ T cell expression dynamics reflect therapeutic immune modulation. (a and b) Both T cell subsets exhibited a significant decline in KLRG1+ expression by early 2024, corresponding with reduced disease activity. After the initiation of hydroxychloroquine in March 2024 and hydrogen capsule therapy in August 2024, a gradual increase in KLRG1+ expression was observed in both cell populations by February 2025, suggesting potential restoration of effector T cell function and immune modulation.

Modulation of Treg-associated markers in response to molecular hydrogen therapy. (a and b) Both subsets showed a transient decline in early 2024, coinciding with reduced disease activity. After the initiation of hydroxychloroquine and hydrogen capsule therapy, a modest rebound in expression was observed by February 2025, suggesting partial restoration of Treg-mediated immune regulation.

Longitudinal reduction in plasmablast frequency following molecular hydrogen therapy. The longitudinal dynamics of plasmablasts, identified as CD27+CD38high B cells, were assessed from HC to the posttreatment follow-up period. A peak in plasmablast frequency was observed in August 2023, followed by a progressive reduction after the initiation of hydroxychloroquine and hydrogen capsule therapy. This trend suggests decreased B cell hyperactivation and improved immunological regulation.
By February 2025, laboratory results showed an ANA titer of 1:160, anti-dsDNA levels of 7.3 IU/ml (Figure 5), C3 at 127.0 mg/dl, and C4 at 54.2 mg/dl. Additional findings included the normal thyroid-stimulating hormone at 0.15 μU/ml, free T4 at 1.56 ng/dl, a CD4 percentage of 27.92%, a CD8 percentage of 42.70%, and a CD4/CD8 ratio of 0.65. B cell subset analysis revealed a decline in naïve B cell Fas+ expression from 85% in August 2023 to 25% in February 2025, while SM B cell Fas+ expression decreased from 90% to 40%, indicating reduced B cell hyperactivity. Additionally, naïve B cell programmed death-1+ (PD-1+) expression, which peaked at 45% in early 2024, declined to 15% by February 2025 (Figure 6), suggesting improved immune tolerance.

The blue line represents anti-dsDNA antibody titers, expressed as dilution ratios (left Y-axis), while the red line shows ANA levels, measured in IU/ml (right Y-axis). Both antibody markers remained elevated until late 2023, followed by a progressive decline beginning with the initiation of hydroxychloroquine in March 2024 and hydrogen capsule therapy in August 2024. Linear regression lines indicate an overall downward trend in autoantibody burden, supporting the observed clinical improvement and immunological stabilization.

(a and b) A progressive reduction in both Fas+ and PD-1+ SM B cell subsets was observed from August 2023 to February 2025, indicating decreased B cell hyperactivation and improved immune homeostasis following immunomodulatory therapy.
The patient continued hydroxychloroquine therapy, which remained essential for controlling her malar rash. However, the introduction of hydrogen capsules appeared to enhance immune regulation, as evidenced by increased KLRG1+ effector Tc, reduced plasmablast activity, and stabilization of B cell exhaustion markers. These findings suggest a potential immunomodulatory effect of H2 in SLE, warranting further investigation.
At her most recent follow-up, the patient reported sustained improvement in her malar rash (Figure 7), reduced fatigue, and overall enhanced well-being. The absence of new lupus flares over the past 6 months suggests a possible synergistic effect between hydroxychloroquine and hydrogen therapy in modulating disease activity. Continued monitoring is essential to assess long-term disease stability and guide potential treatment adjustments. This case highlights the importance of personalized treatment strategies in SLE, integrating hydroxychloroquine as a primary therapy with hydrogen therapy as a supportive intervention for immune modulation and symptom relief. The CARE checklist is available as Supplemental Material.
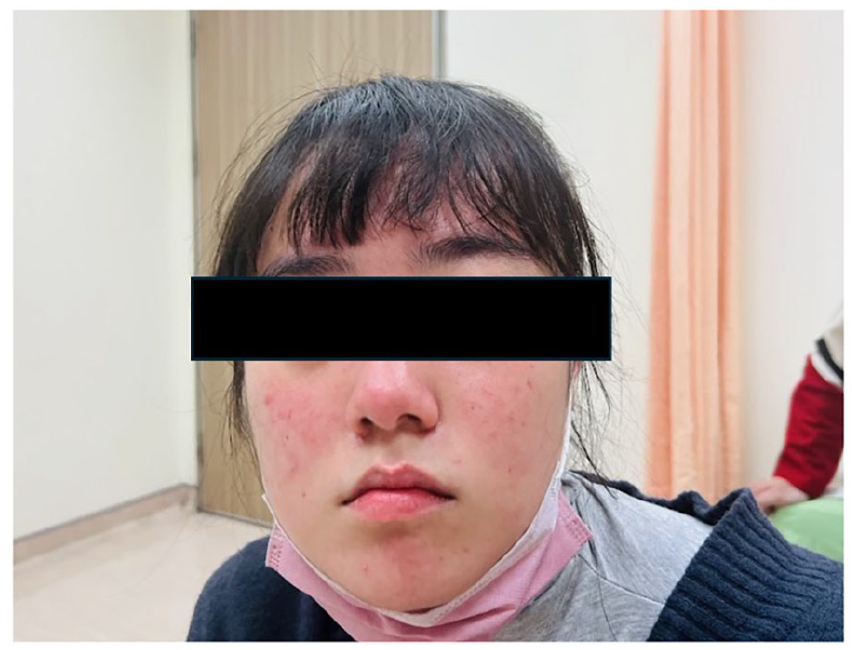
Malar rash following hydroxychloroquine and hydrogen therapy. Compared to earlier disease flares, the malar rash shows a significant reduction in intensity and distribution, with fading erythema and resolution of facial edema.
Discussion
This case report details the clinical course of a 27-year-old female with a well-established diagnosis of SLE, who presented with a persistent and cosmetically significant malar rash that had proven refractory to standard treatment with hydroxychloroquine. The introduction of adjunctive therapy with H2 capsules was followed by sustained improvement in her malar rash, a reduction in fatigue, and an overall enhanced sense of well-being. Notably, these clinical improvements coincided with significant changes in key immunological markers, including an increase in effector Th KLRG1+ and EM Tc KLRG1+ cells, stabilization of natural Tregs, a marked decrease in plasmablasts, a decline in the expression of Fas on both naïve and switched memory B cells, a reduction in PD-1 expression on naïve B cells, and a decrease in both ANA titer and anti-dsDNA levels. The temporal association between the initiation of H2 therapy and these observed clinical and immunological changes suggests a potential link that warrants further exploration into the underlying mechanisms.
H2 has garnered increasing attention for its potential therapeutic benefits in various autoimmune and inflammatory conditions, including SLE, primarily due to its established antioxidant and anti-inflammatory properties. 13 Research indicates that H2 selectively neutralizes cytotoxic reactive oxygen species, such as hydroxyl radicals and peroxynitrite, mitigating oxidative damage at the cellular level. 14 Furthermore, it has been shown to modulate inflammatory pathways, including the nuclear factor kappa-light-chain-enhancer of activated B cells pathway, and reduce the secretion of pro-inflammatory cytokines, contributing to an overall reduction in inflammation. 15 In the context of SLE, a disease characterized by heightened oxidative stress and chronic inflammation, the introduction of H2 may have contributed to immune regulation by helping restore redox balance and dampen the inflammatory milieu, potentially influencing the function of various immune cell populations and the production of autoantibodies.
The observed changes in specific immune cell populations following hydrogen therapy are particularly noteworthy. KLRG1 is an inhibitory receptor expressed on natural killer cells and Tc subsets, including Tregs and effector Tc. While KLRG1 expression on Tregs has been associated with suppressive function, on effector Tc, it is often linked to reduced proliferative capacity and cytokine production, indicating a state of exhaustion or senescence. 16 The increase in both effector Th KLRG1+ and EM Tc KLRG1+ cells, alongside clinical improvement, is an intriguing finding. 17 Another case report noted enhanced KLRG1 expression on Tc cells with hydrogen therapy in the context of subdural hematoma, suggesting a potential link between hydrogen and the modulation of KLRG1+ Tc. Further investigation is needed to fully understand the functional implications of this increase in KLRG1+ effector Tc in the context of SLE and hydrogen therapy. 13
Cutaneous manifestations, such as malar rash, are a frequent and significant aspect of SLE. The pathogenesis of malar rash involves photosensitivity and the deposition of immune complexes in the skin, leading to local inflammation and tissue damage. 18 The sustained improvement in the patient’s refractory malar rash following the introduction of H2 could be attributed to its antioxidant and anti-inflammatory effects on the skin. Several studies have highlighted the beneficial effects of H2 in skin conditions, including the reduction of oxidative damage, regulation of inflammatory responses, and promotion of skin repair. In a mouse model of atopic dermatitis, hydrogen water was shown to ameliorate skin lesions by decreasing inflammation. 19 By reducing local oxidative stress and inflammation in the skin, H2 may have acted synergistically with hydroxychloroquine to alleviate the persistent malar rash in this patient.
The reduction observed in both ANA titer and anti-dsDNA levels following hydrogen therapy is clinically significant. ANA is a highly sensitive but less specific marker for SLE, while anti-dsDNA antibodies exhibit higher specificity and are associated with disease activity, particularly lupus nephritis. The decrease in these key autoantibodies suggests a potential systemic immunomodulatory effect of H2, influencing the fundamental autoimmune processes underlying SLE. This finding aligns with other case reports where H2 therapy was associated with a reduction in anti-dsDNA antibody levels. One case even reported the conversion of anti-dsDNA from positive to negative with hydrogen therapy in a patient with subarachnoid hemorrhage and newly diagnosed SLE. 13 These observations collectively suggest that H2 may play a role in modulating autoantibody production in SLE.
The findings of this case report align with a growing body of evidence suggesting a potential role for H2 in the management of SLE and related autoimmune conditions. Several case reports have documented potential benefits such as immune modulation, reduction in autoantibodies, and improvement in fatigue in patients with SLE treated with H2. One case reported improved renal function and reduced fatigue with hydrogen capsules in an elderly patient with SLE and comorbidities.10,20 Additionally, another study explored the potential of hydrogen therapy in SLE-associated pulmonary arterial hypertension, demonstrating modulation of immune markers. 21 While these findings are encouraging, it is important to acknowledge that the current literature primarily consists of case reports, emphasizing the need for more rigorous, controlled studies to definitively establish the efficacy and role of H2 in the treatment of SLE.
This case report possesses several strengths. It provides a detailed longitudinal account of a patient with refractory malar rash who experienced sustained clinical and immunological improvements following the introduction of adjunctive H2 therapy. Comprehensive monitoring of both clinical symptoms and a detailed panel of immunological markers offers valuable insights into the potential mechanisms of action of H2 in SLE. Furthermore, the use of H2 as an adjunct therapy for refractory malar rash represents a novel approach, and the detailed reporting of specific immune cell subset changes contributes to our understanding of the potential immunomodulatory effects of hydrogen in this complex autoimmune disease. However, a key limitation of this report is the concurrent use of hydroxychloroquine alongside H2 therapy, which complicates the attribution of clinical improvement solely to hydrogen treatment. The overlapping therapeutic effects of these interventions may confound the interpretation of outcomes. Therefore, further investigations under controlled conditions are necessary to delineate the individual contributions of each therapy.
Conclusion
This case report describes a 27-year-old female with SLE and refractory malar rash who experienced sustained clinical improvement and notable immunological changes following adjunctive H2 capsule therapy. These changes included increased KLRG1+ effector Tc, reduced plasmablast activity, stabilization of B cell exhaustion markers, and decreased ANA and anti-dsDNA levels. These findings suggest that H2 may serve as a safe and potentially beneficial adjunctive therapy for managing refractory cutaneous manifestations and modulating systemic autoimmunity in SLE.
The potential clinical relevance of these preliminary observations warrants further investigation. H2 therapy may serve as a complementary approach to conventional SLE treatments, particularly for patients with persistent or refractory symptoms. However, in the absence of controlled clinical data, no clinical recommendations can be made at this stage. Rigorous, well-designed clinical trials involving larger patient cohorts are essential to determine the efficacy and safety of H2 in SLE. Future research should focus on (a) Randomized, placebo-controlled trials to assess the impact of H2 on SLE disease activity and outcomes; (b) optimization of dosage, delivery methods, and treatment duration for clinical application; (c) mechanistic studies to elucidate the immunological and molecular effects of hydrogen therapy; (d) identification of biomarkers predictive for treatment response.
While this case provides encouraging early evidence, controlled trials are critical to validate these findings and to establish the therapeutic role of H2 in the clinical management of SLE.
Supplemental Material
sj-pdf-1-sco-10.1177_2050313X251372755 – Supplemental material for Adjunctive molecular hydrogen therapy for refractory malar rash in systemic lupus erythematosus: A case report with immunological insights
Supplemental material, sj-pdf-1-sco-10.1177_2050313X251372755 for Adjunctive molecular hydrogen therapy for refractory malar rash in systemic lupus erythematosus: A case report with immunological insights by Nai-Sung Hsu, Jeng-Wei Lu, Hsiao-Chen Liu, Yi-Jung Ho, Kuang-Yih Wang and Feng-Cheng Liu in SAGE Open Medical Case Reports
Footnotes
Acknowledgements
The authors would like to thank the patient for providing informed consent and participating in this case report. We also acknowledge the clinical and laboratory staff at Tri-Service General Hospital for their support in patient care and data collection.
Ethical Considerations
This study was approved by the Institutional Review Board (IRB; C202405129, approval date: July 31, 2024) of Tri-Service General Hospital, National Defense Medical Center, Taipei, Taiwan, and was conducted in accordance with applicable ethical guidelines.
Consent for Publication
Written informed consent (V2.0-20240704) was obtained from the patient for publication of the case report, including relevant clinical information and images. All procedures were performed in accordance with institutional ethical standards and the principles outlined in the Declaration of Helsinki (1964) and its later revisions.
Author Contributions
N.-S.H.: conceptualization, methodology, writing original draft, writing review and editing. J.-W.L.: conceptualization, methodology, writing original draft, writing review and editing. H.-C.L.: conceptualization, methodology, writing review and editing. Y.-J.H.: conceptualization, methodology, writing, review and editing. K.-Y.W.: conceptualization, methodology, writing, review, and editing. F.-C.L.: conceptualization, investigation, supervision, writing, review, and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Science and Technology Council, Taiwan (grants NSTC 112-2314-B-016-033, NSTC 113-2314-B-016-052 and NSTC 114-2314-B-016-052-MY3) and Tri-Service General Hospital, Taiwan (grants TSGH-E-112218 and TSGH-E-113238).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Artificial Intelligence Disclosure
During the preparation of this manuscript, a large language model (ChatGPT, by OpenAI) was used solely for language editing and stylistic improvements in select paragraphs. No sections involving the generation, analysis, or interpretation of research data were produced by generative AI. All scientific content was created and verified by the authors. Furthermore, no figures or visual data were generated or modified using generative AI or machine learning-based image enhancement tools.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
