Abstract
Objective
This study aimed to determine the effectiveness and safety of using intranasal ketamine, midazolam, or their combination for sedation during pediatric computed tomography.
Method
In this randomized, double-blind study, 60 children aged ≤2.5 years who were scheduled to undergo computed tomography were allocated into the following three groups: Group K (ketamine 10 mg/kg), Group M (midazolam 0.2 mg/kg), and Group C (ketamine 7.5 mg/kg + midazolam 0.1 mg/kg). In all cases, the drugs were administered intranasally. Sedation parameters were assessed.
Result
Of the 60 randomized participants, 10 were excluded due to failed sedation. Data from 16 (Group M), 14 (Group K), and 20 (Group C) participants were analyzed. The mean sedation scores were similar: Groups K (2.86 ± 0.38), M (2.63 ± 0.74), and C (2.70 ± 0.68) (p = 0.579). Time to adequate sedation was shortest in Group C (22 ± 9 min; p = 0.022). Recovery was fastest in Group M (63 ± 25 min; p = 0.007). Discharge times differed significantly (p = 0.036) among the groups, with Group M exhibiting a discharge time of 92 ± 29 min. Group C had superior parental separation scores compared with the other groups (p = 0.035).
Conclusion
Intranasal administration of ketamine, midazolam, or their combination is safe and effective for inducing sedation during computed tomography in pediatric patients with minimal side effects.
Introduction
Sedation is being more commonly used for pediatric patients undergoing radiological imaging procedures because it decreases patient anxiety and parental emotional discomfort while facilitating procedure completion.1,2 However, sedation can be unsafe for children and therefore requires special expertise. 3 A desirable sedating agent should have a rapid onset, short duration of action, good patient and parental acceptance, predictable results, rapid recovery, and no serious side effects. 4
In modern computed tomography (CT) machines, multi-slice scanners allow fast image acquisition. 5 Nonetheless, imaging results are adversely affected by patient motion.5,6 In addition, CT procedures in certain circumstances may require intravenous (IV) cannula insertion for contrast administration, which is a painful procedure. Therefore, there is a need for pediatric procedural sedation to provide comfort for the child. 7 These procedural sedations are performed in the radiological suites outside the operating rooms. Sedation outside the operating room can be challenging as the environment may be unusual, especially when performed by a less skilled professional. 8 IV and intramuscular routes of sedation, although effective, are not well received by children due to their natural fear of syringes, needles, and pain. 9 The intranasal route as a possible alternative route for procedural sedative medications facilitates easy administration because it is associated with less distress, decreased risk of needle-stick injuries, and less staff and skills needed for its administration.2,10
Owing to its noninvasiveness, rapid onset of action, and simplicity of administration, the intranasal route has gained increasing popularity as a means of inducing sedation in pediatric patients during CT, especially for uncooperative pediatric population. 2 Despite its benefits, significant knowledge and implementation gaps persist, particularly in environments with limited resources. Intranasal sedatives including midazolam, dexmedetomidine, and ketamine have been well-researched and used in clinical guidelines for procedural sedation in children undergoing CT and other diagnostic imaging procedures in developed countries.11,12 Compared with conventional IV methods, these agents are more effective in achieving appropriate sedative levels while minimizing respiratory and cardiovascular adverse effects.11,12 However, in low-resource settings, various constraints prevent broad adoption and optimization of intranasal sedation procedures. There are few context-specific clinical trials or observational studies that evaluate the safety, effectiveness, and appropriate dose regimens of intranasal sedatives in pediatric populations. The majority of available data are derived from research performed in high-income nations, which may fail to account for changes in population health status, pharmacogenomics, or healthcare infrastructure. 13 Secondly, there are logistical and infrastructural challenges that are unique to low-resource settings, such as limited availability of atomization devices or mucosal atomizers, insufficient training of healthcare personnel in performing intranasal drug administration techniques, and sporadic supply chains for essential sedative medication. 13 Many hospitals lack procedural sedation guidelines or rely largely on parenteral administration, which necessitates more intense monitoring and personnel experience, both of which may be in short supply. No national or institutional guidelines have been established for achieving intranasal sedation during imaging procedures. Without established methods, the selection of sedative drug, dose, and monitoring vary greatly, thus jeopardizing patient safety and procedural success. Furthermore, caregiver concerns and cultural views of sedative procedures may restrict acceptance and adherence to new intranasal approaches.12,13
To overcome these gaps, pragmatic research trials and implementation studies in low-resource settings are required to assess the feasibility, safety, and effects of intranasal sedation procedures for improving pediatric care quality and equity in imaging facilities.
This study primarily aimed to assess the mean sedation score using the Pediatric Sedation State Scale (PSSS) among children who received intranasal ketamine alone, midazolam alone, or a combination of the two. The study’s secondary goals were to compare times to achieve adequate sedation, recovery, and discharge as well as changes in hemodynamic parameters, respiratory rate (RR), oxygen saturation (SpO2), parental separation scores, IV cannulation acceptance, and adverse effects in children receiving intranasal ketamine alone, midazolam alone, or a combination of ketamine and midazolam.
Materials and methods
Study design
This was a randomized, double-blind clinical study (trial registration number: https://pactr.samrc.ac.za/PACTR202211882566561, Date: 18 November 2022). This study was conducted between December 2022 and December 2023.
Study site
The study was conducted at the Radiology Department of Korle-Bu Teaching Hospital (KBTH), a tertiary referral center in Ghana. The department has one CT machine managed by consultants, resident radiologists, and radiographers. It caters for a wide range of patient categories from neonates to geriatric patients.
Study population
The study population included all children scheduled for CT under sedation.
Inclusion criteria
Children with ASA I and II physical status who weighed ≤10 kg and whose parents/guardians provided informed consent for participation in the study were enrolled.
Exclusion criteria
Children with known allergies to midazolam or ketamine, anomalies of the cardiovascular system, otorhinolaryngeal disease, and active respiratory tract infection were excluded from the study.
Sample size
The sample size was first estimated using the Hajian–Tilaki formula, 14 which was based on sedation score data obtained by Gharde et al. 15 To achieve 80% power at a 5% level of significance and 95% confidence interval (CI), each group needed at least nine participants. To account for any attrition or procedure violations, a total of 60 individuals were enrolled and randomized evenly into three groups. During the trial, 10 participants were removed from the final analysis due to unsuccessful sedation, including 6 in Group K (ketamine alone), 4 in Group M (midazolam alone), and none in Group C (combination treatment). The total sample size was 50 (Group C: 20; Group M: 16; and Group K: 14). These exclusions were essential owing to the difficulty in providing a meaningful sedation score following protocol deviation and the requirement for rescue sedation. A follow-up power analysis utilizing the retained group sizes and initial effect size estimations found that the study had power >80%, exceeding the originally anticipated 80%. This supports the study’s capacity to uncover statistically significant variations in sedative effectiveness despite the exclusions.
The omission of failed sedation instances was justified to maintain the integrity and comparability of the primary result. However, the pattern of treatment failure was clinically relevant, and the data are presented descriptively to illustrate potential differences in efficacy.
Although sedation scores are fundamentally ordinal, the scale utilized in this study had a wide range with thorough consistency and accuracy, allowing for interval-level evaluation. This enabled the use of parametric approaches and justified the use of a sample size formula intended for continuous variables.
Procedures
This study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024, and with approval from the ethics committee. After ethical approval for the study was obtained from the Institution Review Board of the Korle-Bu Teaching Hospital in Ghana (Protocol Number: KBTH-IRB/000120/2021, Date: 22/06/2022), children who were scheduled to undergo CT under sedation were assessed for fitness. Written informed consent was obtained from the parents/guardians after they were educated on fasting and the study protocol. Children who met the eligibility criteria were consecutively recruited and randomized into three groups (Groups K, M, and C). Randomization was achieved by simple balloting without replacement by an independent investigator not involved in the study. In total, 60 ballot papers with 3 sets of 20 papers, labeled either as A, B, or C, were created by the independent investigator. All the ballot papers were folded and mixed in a sealed opaque envelope. The parent/guardian of each recruited child was asked to pick a ballot from the sealed opaque envelope to determine which group the child had been randomized into. The ballot paper once picked was discarded. The same procedure was followed for all subsequent qualifying patients until all ballot papers were exhausted from the envelope. Upon study completion, the identities of the codes A, B, and C were revealed as intranasal ketamine intervention (Group K), intranasal midazolam intervention (Group M), and a combination of intranasal ketamine and midazolam intervention (Group C), respectively.
This was a double-blind study. The study medications were prepared using midazolam (1 mg/mL) and ketamine (50 mg/mL) preparations. To maintain the integrity of the blinding process, several measures were implemented to ensure that the drug solutions were visually and physically indistinguishable. All study medications were prepared using colorless, transparent solutions and drawn into identical 5-mL syringes without labels or identifying marks. Each syringe contained an equal volume of 2.5 mL, regardless of group allocation, eliminating volume-based cues. The syringes were sealed and handled uniformly to prevent any external differentiation. Additionally, the preparation was conducted in a separate location by an independent investigator who was not involved in participant care, medication administration, or outcome assessment. These steps ensured that neither the administering anesthesia provider nor the participants or their guardians could discern the contents of the syringes, thereby maintaining double-blind conditions throughout the study. The contents of the syringe received by the participants in each group was revealed to the principal investigator at the end of the study.
Children in Group K received 10 mg/kg intranasal ketamine, those in Group M received 0.2 mg/kg intranasal midazolam, and those in Group C received a combination of 7.5 mg/kg ketamine and 0.1 mg/kg midazolam intranasally. Study drugs were prepared to a total volume of 2.5 mL, with half of the volume placed in each nostril via a mucosal atomization device. On arrival at the CT scan suite, the study protocol was again explained to the parents/guardians, and children were kept warm in the arms of the parents and covered using a cloth. The basal heart rate (HR), blood pressure (BP), RR, and SpO2 were recorded. The parents were encouraged to keep interacting with the children playfully in their arms until they were calm (not crying). The parents then assisted in administering the study drugs to the children in a semi-recumbent position (child in the mother’s arm) under supervision approximately 45 min before the planned procedure.
The HR, BP, RR, and SpO2 of the children were monitored and recorded prior to administration of the intranasal solution and then at 5-min intervals thereafter until imaging was performed. Continuous monitoring was conducted after the imaging procedure in a recovery area until discharge.
Patients’ age, sex, and body mass index (BMI) were recorded. The level of sedation in the patients after drug administration was assessed using the Pediatric Sedation State Scale (PSSS) 16 every 5 min for 40 min. PSSS <3 was considered an adequate sedation depth for CT. Children who did not show satisfactory response to the sedative drugs within 40 min of sedative administration were excluded and recorded as failed sedation. The interval between administration of sedative drugs and achieving PSSS <3 was recorded as the time to achieve adequate sedation. Once adequate sedation was achieved, the patients were transferred to the scanner room, and imaging was performed. Children who failed to achieve sedation were administered with halothane inhalational anesthesia to complete the imaging procedure as mandated by local guidelines.
On transfer of patients into the scanner, the ease of separation from their parent was assessed according to a 4-point separation score scale (1: poor (crying, clinging); 2: fair (crying but not clinging); 3: good (whimpers, easily reassured); and 4: awake excellent (easy separation)); 15 a score of ≥3 was considered satisfactory. IV cannulation was performed before the scan procedure. A 4-point scoring system was used to assess the acceptance of IV cannulation (1: poor (terrified, crying); 2: fair (fear of the needle, not reassured); 3: good (slight fear of the needle, easily reassured); and 4: excellent (unafraid, accepts IV cannula readily)); 15 a score of ≥3 was considered satisfactory.
Adverse events such as bradycardia (20% reduction in pre-sedation HR), tachycardia (20% increment in pre-sedation HR), and excessive salivation (secretion of saliva requiring cleaning with dry gauze more than once or requiring suctioning) were recorded.
The time to discharge from the unit was defined as the interval between the start of sedative administration and return to baseline alertness. The discharge criteria were based on a Modified Aldrete score ≥8. The recovery time was defined as the time from the end of the procedure to the time of discharge.
The reporting of this study conforms to the Consolidated Standards of Reporting Trials (CONSORT) 2025 statements. 17 There was no public or patient involvement in the design of the study.
Statistical analysis
The data were analyzed using Statistical Package for Social Sciences (IBM SPSS® version 25). Continuous data were summarized as mean (±SD), and categorical variables were presented as frequencies and proportions. The chi-squared test was used to analyze sex-based differences. The participants’ age, BMI, baseline hemodynamic parameters, SpO2, and RR were compared across the three groups using one-way analysis of variance (ANOVA). The primary outcome (sedation scores) was compared across the three groups using one-way ANOVA and a pairwise comparison with Tukey’s test. Fisher’s exact test was performed to compare the sedation failure rates between the three groups.
Secondary outcomes (times to adequate sedation, recovery, and discharge) were compared across the three groups using one-way ANOVA and a pairwise comparison with Tukey’s test. Other secondary outcomes such as changes in hemodynamic parameters, RR, and SpO2 were analyzed between the three groups using a three-way repeated measures ANOVA. Bonferonni test was used to analyze the intergroup differences in the mean hemodynamic parameters, RR, and SpO2 at each time point.
Other outcomes such as separation scores and IV cannulation acceptance were compared between the three groups using Fisher’s exact test. Adverse effects were compared between the three groups using the chi-squared test. A p-value <0.05 was considered to indicate statistical significance.
Results
In total, 60 patients were enrolled in the study. Among them, 10 did not complete the study as shown in the CONSORT diagram (Figure 1). Four participants in Group M and six in Group K did not complete the study on account of failed sedation. All the participants in Group C completed the study. The difference in failed sedation between the three groups was statistically significant (p = 0.003).

CONSORT flow diagram. CONSORT: Consolidated Standards of Reporting Trials.
There was no statistically significant difference in the demographic characteristics and pre-sedation vital sign measurements between the three groups (Table 1).
Demographic and pre-sedation vital sign measurements of the participants.

Mean (±SD): mean (±standard deviation), bn (%): number in each variable category (percentage).
Group K comprised patients administered with ketamine, Group M comprised patients administered with midazolam, Group C comprised patients administered with ketamine and midazolam.
BMI: body mass index; HR: heart rate; SBP: systolic blood pressure; DBP: diastolic blood pressure; RR: respiratory rate, SpO2: oxygen saturation.
There was no statistically significant difference in the demographic characteristics and pre-sedation vital sign measurements between patients with successful and failed sedation (Table 2).
Comparison of demographic characteristics and pre-sedation vital sign measurements in patients with successful and failed sedation.
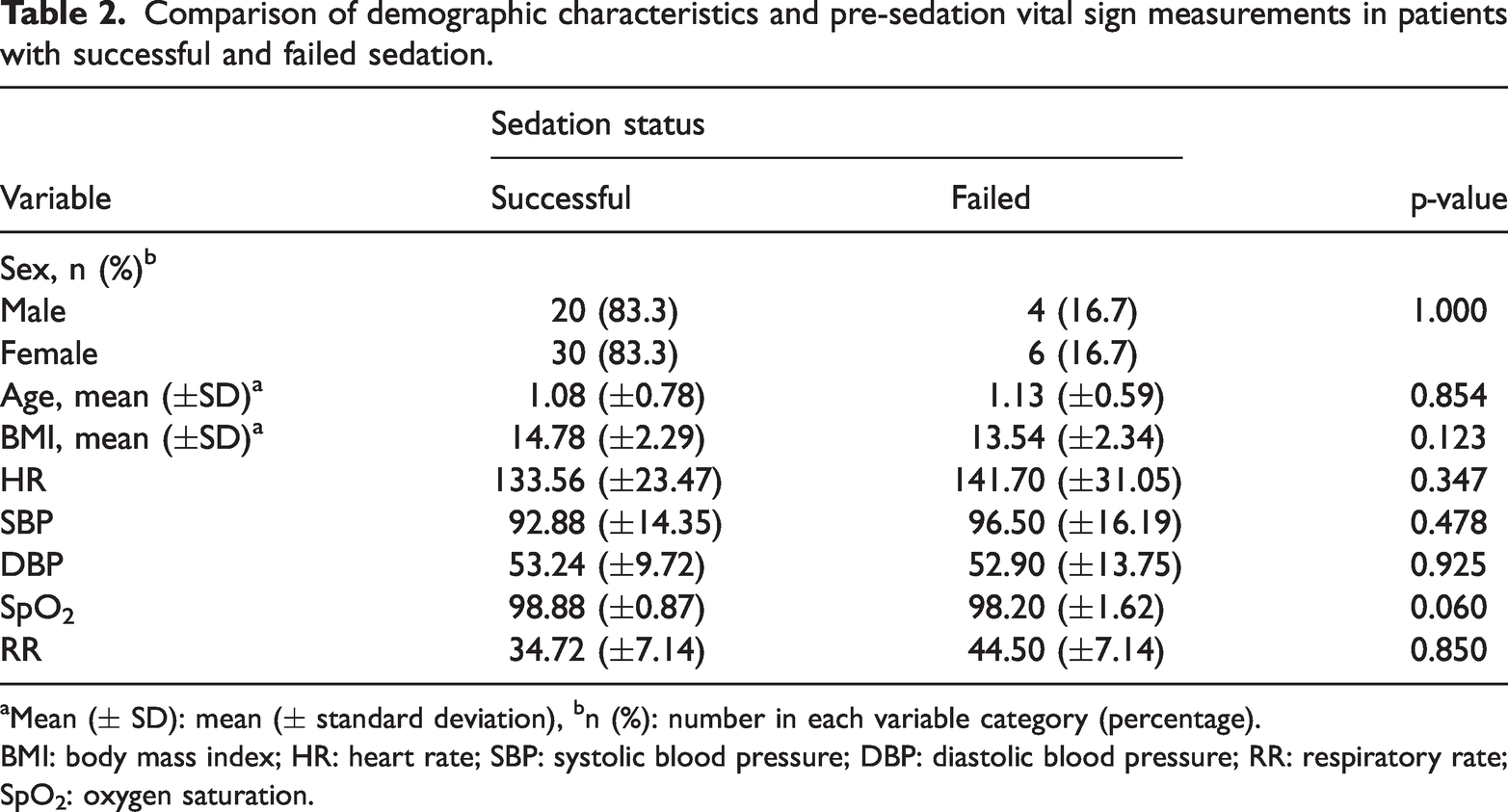
Mean (± SD): mean (± standard deviation), bn (%): number in each variable category (percentage).
BMI: body mass index; HR: heart rate; SBP: systolic blood pressure; DBP: diastolic blood pressure; RR: respiratory rate; SpO2: oxygen saturation.
A three-way repeated measures ANOVA showed no statistically significant difference in the mean HRs between the three groups (p = 0.412). Participants in Group M recorded higher mean HRs than those in Groups K and C. Intergroup analysis showed a significant difference in the mean HR at 30 min post-sedation between Groups M and C (p = 0.007) (Figure 2).

Changes in heart rate after the administration of intranasal medications. Group K comprised patients administered with ketamine, Group M comprised patients administered with midazolam, Group C comprised patients administered with ketamine and midazolam.
A three-way repeated measures ANOVA showed that there was no statistically significant difference in the mean systolic BPs among the three groups over the study period (p = 0.056). However, higher mean systolic BPs were recorded in Group K than in Groups M and C (Figure 3).

Changes in systolic (left) and diastolic (right) blood pressure after the administration of intranasal medications. Group K comprised patients administered with ketamine, Group M comprised patients administered with midazolam, Group C comprised patients administered with ketamine and midazolam.
A three-way repeated measures ANOVA showed no statistically significant difference in the mean diastolic BPs among the three groups over the study period (p = 0.498). However, lower mean diastolic BPs were recorded in Group M than in Groups K and C (Figure 3).
Intergroup analysis showed a statistically significant difference in systolic BPs, but not in diastolic BPs, between Groups K and M at 30 (p = 0.039) and 40 (p = 0.050) min post-sedation (Figure 3).
A three-way repeated measures ANOVA showed no statistically significant difference in the mean SpO2 (p = 0.457) and mean RR (p = 0.138) between the three groups over the study period. Higher mean RRs were observed in Group C than in Groups K and M.
Intergroup analysis showed a significant difference in SpO2 between Groups C and M at 30 (p = 0.023), 35 (p = 0.023), and 40 (p = 0.044) min post-sedation. Intergroup analysis showed a statistically significant difference in RR between Groups K and C at 10 (p = 0.038), 15 (p = 0.049), and 25 (p = 0.008) min post-sedation (Figure 4).

Changes in the mean SpO2 (left) and respiratory rate (right) after the administration of intranasal medications. Group K comprised patients administered with ketamine, Group M comprised patients administered with midazolam, Group C comprised patients administered with ketamine and midazolam. SpO2: oxygen saturation.
There was a statistically significant difference between the groups with respect to time to adequate sedation, time to recovery, time to discharge, and separation score (Table 3).
Time to adequate sedation, recovery and discharge, PSSS, separation score, acceptance of IV cannulation, and adverse events.

p-value (statistically significant).
Group K comprised patients administered with ketamine, Group M comprised patients administered with midazolam, Group C comprised patients administered with ketamine and midazolam.
aMean (±SD): mean (±standard deviation), b n (%): number in each variable category (percentage).
PSSS: Pediatric Sedation State Scale; IV: intravenous.
“Adequate sedation: PSSS <3”; “satisfactory separation score ≥3: good (whimpers, easily reassured)”; “satisfactory acceptance of IV cannulation ≥3: good (slight fear of needle, easily reassured)”; “bradycardia: 20% reduction in pre-sedation heart rate”; “tachycardia: 20% increment in pre-sedation heart rate”; “excessive salivation: secretion of saliva requiring cleaning with dry gauze more than once or requiring suctioning.”
There was no statistically significant difference in adverse effects recorded between the three groups. The adverse effects were transient and needed no intervention (Table 3).
A pairwise comparison test showed a statistically significant difference in time to adequate sedation between Groups C and M (95% CI = −13.8 to −0.9; p = 0.021) and a statistically significant difference in time to recovery between Groups K and M (95% CI = 4.0–50.8; p = 0.018) and Groups C and M (95% CI = 4.3–47.3; p = 0.015). There was a statistically significant difference in time to discharge between Groups K and M (95% CI = 1.1–49.3; p = 0.039) (Table 4).
Pairwise comparison test for time to adequate sedation, recovery, and discharge.
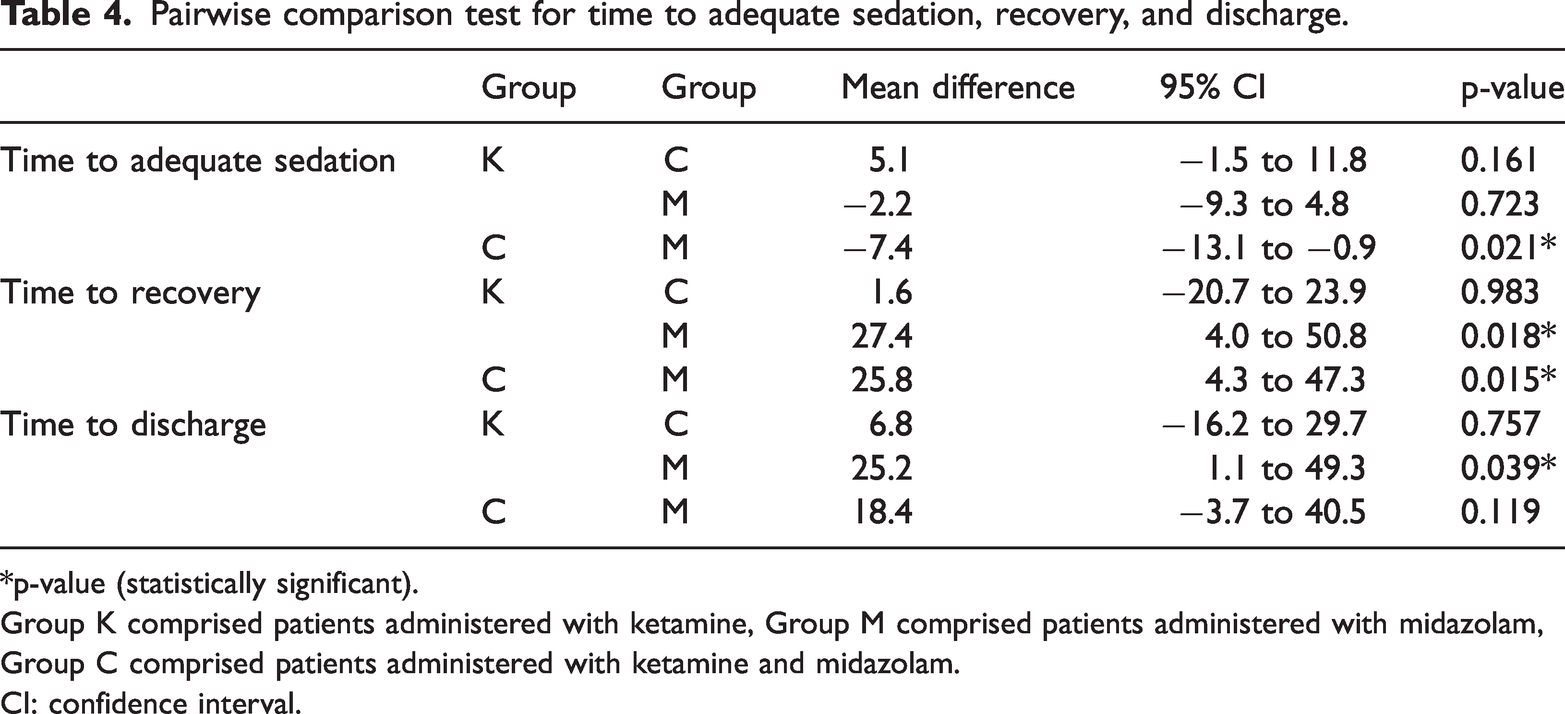
*p-value (statistically significant).
Group K comprised patients administered with ketamine, Group M comprised patients administered with midazolam, Group C comprised patients administered with ketamine and midazolam.
CI: confidence interval.
Discussion
Intranasal drug administration has gained popularity in pediatric sedation because it is a less intrusive alternative to IV or oral routes, which can be distressing or poorly tolerated in children. Although the IV route is still chosen by many anesthesia practitioners because of its titratability and predictable onset of action, its usage is frequently limited in reluctant young patients. Consequently, intranasal drugs such as midazolam and ketamine have been intensively studied as potential premedication choices in this group.18–20
Participants in this research varied in age from 1 week to 2.5 years, the age range where separation anxiety is common. 2 Group K showed the highest sedation failure rate (30%), followed by Group M (20%); no failures were observed in Group C. This demonstrates the diversity in sedation efficacy between drugs, emphasizing the necessity for agent- and population-specific data with plausible molecular reasons for these effects. Several reasons may have contributed to the relatively high failure rate of Group K. Despite its robust dissociative properties, intranasal ketamine has a bitter taste and can cause nasal pain, likely leading to partial ejection or limited absorption via the nasal mucosa. 21 Furthermore, ketamine’s sedative qualities alone may be inadequate in some pediatric groups, particularly nervous or uncooperative children who require rapid and thorough sedation. Similarly, intranasal midazolam, although frequently used for its anxiolytic and sedative properties, has a relatively short duration of action and may have limited effectiveness when used as monotherapy in situations involving procedural stress. 22 Individual mucosal variables and drug delivery strategy impact the pharmacokinetics of intranasal midazolam, which can lead to different clinical effects. 23 The absence of sedation failure in Group C shows that ketamine and midazolam synergistically provide complimentary advantages, with midazolam providing anxiolysis and causing amnesia and ketamine providing analgesia and deeper sedation, resulting in more consistent sedation results.15,24 Prior studies have demonstrated enhanced sedation success and fewer adverse behavioral reactions when ketamine and midazolam are combined via the intranasal route. 25
Baseline hemodynamic and respiratory parameters were similar between the three groups, and no clinically significant differences were noted following intranasal medication delivery. These findings are consistent with those of previous studies showing that intranasal midazolam and ketamine have hemodynamic stability.18,19 The considerably higher HRs observed in Group M are most likely due to the cohort’s lower mean age, which is consistent with age-appropriate physiological norms. 20 Systolic and diastolic BPs were nonsignificantly higher in Groups K and C than in Group M, most likely due to the sympathomimetic effects of ketamine, as previously described.26,27
Respiratory measures, such as SpO2 and RR, remained within physiological limits, with no significant variations between the three groups. No child exhibited desaturation (SpO2 ≤ 92%), supporting the findings of Ibrahim et al., who reported no major respiratory compromise with intranasal ketamine during sedation for magnetic resonance imaging. 28 In contrast, Abrams et al. documented temporary desaturation in 2 of the 10 participants receiving intranasal sedation for dental operations, underscoring the necessity for procedure-specific safety data. 29 Ketamine has been linked to moderate respiratory depression due to decreased partial pressure of arterial carbon dioxide reactivity; however, this was not observed in the current investigation. 30
There were no significant variations in the PSSS ratings at the time of adequate sedation between the three groups. However, compared with Group M, Group C achieved adequate sedation within a short period. Compared with Group K, Group C achieved adequate sedation within a nonsignificantly shorter period. Group M had the shortest recovery and discharge periods, which is consistent with midazolam’s known pharmacokinetic profile. These results are consistent with those reported by Bahetwar et al., who found that intranasal ketamine had a faster onset and a longer recovery period than midazolam. 24
Inconsistencies in the reported onset and recovery periods may be due to interstudy variations such as dosage and intranasal administration systems. For example, Alp et al. used a lower ketamine dosage (4 mg/kg) and observed a considerably quicker onset of sedation with midazolam, which contradicted the findings of the current study. 31 The increased ketamine dosage utilized in the current trial might explain the prolonged recovery times and enhanced sedative effectiveness. This emphasizes the vital need of consistent dosage guidelines and delivery techniques in pediatric intranasal sedation studies.
Parental separation and IV cannulation acceptance ratings further confirmed Group C’s performance, with 70% of the children displaying excellent separation and 90% exhibiting good-to-excellent cannulation tolerance. Although ketamine has been linked to improved procedural acceptability due to its analgesic effects, this was not observed in the current study, with 28.6% of the children in Group K exhibiting poor cannulation acceptance. These findings contradict those of Gharde et al., who ascribed improved cannulation tolerance to ketamine's analgesic effects. 15 This disparity might be explained by interindividual variation in pediatric anxiety and pain expression, which can be impacted by both psychological and developmental variables. 32
Adverse events included bradycardia (40%), tachycardia (28%), and excessive salivation (8%). These were modest, self-limiting events and did not necessitate medical attention. No statistically significant variations in adverse event occurrence were noted between the three groups. These findings are consistent with those of previous research indicating the safety of intranasal midazolam, ketamine, and their combination.29,33 In another study, hypersalivation was more prevalent in ketamine-treated groups, as predicted, given its known cholinergic effects. 34
Despite a growing body of research on intranasal sedation in pediatric populations, there is a scarcity of data related to low-resource settings, where IV access may be more difficult and general anesthesia infrastructure is restricted. This study helps bridge this gap by establishing that intranasal sedation with ketamine, midazolam, or their combination is both practical and safe for pediatric patients scheduled to undergo CT in a low-resource environment. Nonetheless, differences in onset, duration, and recovery outcomes between studies underscore the need for further research targeted to the limits of low-resource settings. Standardized protocols for intranasal sedation, which include medication selection, concentration, volume, and administration technique, are critical for improving procedural sedation regimens. Future multicenter studies with larger samples and standardized evaluation tools will be required to identify optimal practices that are scalable and resource-constrained.
A practical problem in intranasal medication administration is maintaining adequate mucosal absorption while preventing overflow or first-pass metabolism caused by swallowing. Volumes >1 mL per nostril are normally avoided. The total amount provided in this trial was 1.25 mL in each nostril, which may have had an influence on drug absorption efficiency, especially in young infants.35–37 Future research should prioritize the use of concentrated formulations and delivery systems that allow for accurate dosing with little volume.
This study compares the effectiveness of ketamine, midazolam, and their combination for procedural sedation in children undergoing CT. Although the results show that the groups had varied sedation outcomes, some limitations must be noted, particularly considering the study's statistical power and analytical technique.
After excluding individuals who had failed sedation, a post-hoc power analysis found that the remaining sample still had statistical power greater than 80%. This shows that the study’s sensitivity was sufficient to identify variations in sedation ratings across the three groups. However, although statistically sound, this conclusion should be interpreted with caution. The exclusion of individuals with unsuccessful sedation increases selection bias and restricts the therapeutic relevance of the findings. These omitted individuals constitute an essential subset of the target population, and their results are directly relevant to real-world sedative effectiveness. The assumption that missingness due to unsuccessful sedation is fully random could be unwarranted, although a comparison of baseline characteristics between excluded and included patients revealed no significant differences (Table 2). These failures may be due to the sedative medications utilized; therefore, their omission may have resulted in an overestimation of the agents’ efficacy. Although the revised power confirms the ability to detect group differences among individuals who completed the study, it may not truly reflect the overall clinical effectiveness of the therapies.
Another methodological issue is the use of ANOVA to compare sedation scores, which are naturally ordinal rather than continuous variables. Although ANOVA is a robust method, given the study’s small and uneven group sizes, breaches of its assumptions are possible.
Conclusions
Intranasal administration of midazolam, ketamine, and their combination provided effective and safe sedation for pediatric patients scheduled to undergo CT, with no significant cardiorespiratory compromise. The combination regimen achieved the highest sedation success rate and improved cooperation during parental separation and IV cannulation. Midazolam alone resulted in faster recovery and discharge times. While all interventions were generally well-tolerated, the higher incidence of poor cannulation acceptance in Group K, potential methodological limitations, and potential effect of intranasal volume on drug absorption represent some of the study limitations. These findings support the feasibility of intranasal sedation in low-resource settings. Further studies are needed to refine dosing strategies and administration techniques for optimized outcomes.
Footnotes
Acknowledgements
We wish to acknowledge the staff of the Department of Anaesthesia, Korle-Bu Teaching Hospital, for their immense support during the conduct of this study.
Authors’ contributions
EOD, BNA, and RD developed the concept and wrote the manuscript. RE and GA analyzed the data. GIOA, OSD, LBA, AA, and NMV contributed to the writing and review of different sections of the manuscript. Prior to submission, all the authors were involved in the review of the final manuscript.
Declaration of conflict of interests
The authors declare no conflict of interests.
Data availability statement
The datasets used and analyzed in this study are accessible from the corresponding author upon reasonable request.
Funding
This study was self-funded.
