Abstract
Neurobrucellosis is a severe and rare complication of human brucellosis, particularly in the pediatric population. It manifests with diverse clinical presentations, with meningoencephalitis being the most common. Limited cases have been reported in Saudi Arabia. Here, we present the case of an 11-year-old boy diagnosed with neurobrucellosis who developed diplopia, inward deviation of the left eye, and ophthalmoplegia. Cerebrospinal fluid analysis revealed pleocytosis, elevated protein levels, and high opening pressure. Brain magnetic resonance imaging demonstrated microabscesses with nodular enhancement, dural thickening in the quadrigeminal cistern, and swelling with edema of the left optic nerve. To the best of our knowledge, this is the first reported case of a patient with brain microabscesses secondary to Brucella infection in Saudi Arabia. This case highlights the need for heightened awareness of neurobrucellosis as a differential diagnosis in children presenting with unusual neurological symptoms in endemic regions.
Introduction
Brucellosis, caused by Brucella species, is the most common zoonotic disease globally and is endemic in parts of Africa, western Asia, Latin America, and the Mediterranean region.1,2 Transmission typically occurs through contact with infected animals or consumption of unpasteurized dairy products. 2 Human brucellosis can affect multiple organ systems, leading to diverse clinical presentations.2–4
Neurobrucellosis (NB) is a rare but severe complication of brucellosis, occurring in approximately 3%–5% of all brucellosis cases.1,5 It can present at any disease stage with manifestations such as meningoencephalitis, cranial neuropathies, psychiatric symptoms, and myelitis.2,5 Diagnosis of NB relies on neurological symptoms, cerebrospinal fluid (CSF) analysis, serological testing, and imaging findings.5,6 Slow bacterial growth in cultures and nonspecific symptoms contribute to significant delays in diagnosis. 7
Here, we present a pediatric case of NB from Saudi Arabia characterized by brain microabscesses, highlighting the importance of clinical suspicion in endemic areas.
Case presentation
An adolescent male patient aged <12 years with a negative medical history presented with complaints of bone and muscle pain that began 2 months prior to his hospital visit. These symptoms were accompanied with frontal headaches, vomiting, and double vision over the past 2 weeks. The frontal headache was sudden in onset and associated with neck pain, which was particularly severe in the early morning. Vomiting occurred three to four times daily, primarily upon waking. The patient also exhibited ophthalmoplegia in both eyes, experienced transient episodes of aphasia twice, and had intermittent fever responsive to antipyretics.
On examination, the following vital signs were observed: temperature, 37.6°C; heart rate, 98 bpm; respiratory rate, 23 bpm; and blood pressure, 104/68 mmHg. The patient was conscious, alert, and oriented, with normal power, tone, reflexes (abdominal reflexes; planter response; biceps, triceps, knee, and ankle reflexes), and intact sensation in the upper and lower limbs. Cerebellar examination, including diadochokinesis, point-to-point movement, Gait and Romberg’s test, was unremarkable.
On initial assessment, impaired ocular motility was noted and broadly referred to as ophthalmoplegia. Upon detailed ophthalmology examination, the findings were as follows: vision 20/30 in both eyes (OU), right eye apparent esotropia, and left abducens (sixth cranial nerve) palsy. Fundus examination revealed bilateral hyperemic, mildly elevated optic discs, indicating mild papilledema.
Cranial nerve examination revealed left abducens nerve palsy, whereas other cranial nerves were normal. There were no signs of abnormal movements, ataxia, or systemic symptoms such as weight loss, night sweats, chest pain, or palpitation.
The patient had no history of regular medication use, recent travel, or contact with a new housemaid. Meningeal signs were absent, and there was no hepatosplenomegaly or lymphadenopathy.
The patient reported a history of raw milk ingestion and contact with camels. Additionally, two siblings and his mother had previously been diagnosed with brucellosis. Based on the clinical presentation and family history, NB was strongly suspected.
Laboratory investigations revealed moderate neutropenia with a total leukocyte count of 4400/μL (21% polymorphs and 67% lymphocytes), normal platelet count, and normal hemoglobin levels. Liver and renal function tests were within normal ranges. The C-reactive protein level was 0.4 (normal range: 0–5) mg/L, erythrocyte sedimentation rate was 21 (normal range: 2–30) mm/h, and the purified protein derivative skin test for tuberculosis was negative.
A brain CT performed before lumbar puncture showed no acute brain insult, with normal brain parenchyma and no midline shift.
CSF analysis (Table 1) revealed a CSF opening pressure of 25 cm H2O and lymphocytic pleocytosis with a white blood cell count of 140/mL (normal range: 0–5/mL), comprising 86% lymphocytes and 6% polymorphonuclear cells. Protein levels were elevated at 597 (150–450) mg/L, and glucose levels were reduced at 1.4 (2.2–3.9) mmol/L, with a simultaneous blood glucose level of 3.5 (3.6–6.0) mmol/L. CSF Gram stain showed no microorganisms. CSF Ziehl–Neelsen stain for tuberculosis and Löwenstein–Jensen (LJ) cultures for tuberculosis were also negative. CSF multiplex polymerase chain reaction (PCR) using the BioFire FilmArray panel was negative for viruses, bacteria, and Cryptococcus neoformans; moreover, the tuberculosis PCR test was negative. A second lumbar puncture was performed after 1 week of therapy, and the results are shown in Table 1. The recorded CSF opening pressure was 25 cm H2O. The standard threshold for elevated CSF opening pressure is typically set at >28 cm H2O in adults, and values above 20–25 cm H2O are considered elevated in pediatric populations, particularly in the presence of clinical findings such as bilateral papilledema and left sixth nerve palsy, indicating elevated intracranial pressure. This interpretation is supported by the patient’s rapid improvement in ocular findings (squint) following therapeutic lumbar puncture.
CSF analyses results for the patient.

CSF: cerebrospinal fluid; RBC: red blood cell; WBC: white blood cell.
Brain magnetic resonance imaging (MRI) with contrast revealed nodular enhancement with dural thickening in the quadrigeminal cistern. A central hypointense, nonenhanced area with diffusion restriction, consistent with a microabscess, was observed. Additional findings included linear enhancement along the inner surface of the orbit and thickening with edema of the left optic nerve (Figure 1). A standard agglutination test for Brucella spp., BRUCELLACAPT®, is a single-step immunocapture assay for the detection of total anti-Brucella antibodies. It consists of U-bottom well-strips coated with anti-human immunoglobulins. This test showed significant titers of 1:640 for both Brucella melitensis and B. abortus in the serum; however, the test was not performed on the CSF sample because it was not validated in our laboratory. An automated blood culture system was used, and multiple blood cultures were sent, all of which returned negative. Enzyme-linked immunosorbent assay (ELISA) for anti-Brucella antibodies was not available for the serum or CSF.

Magnetic resonance imaging findings of the patient. (a) There is linear enhancement along the inner surface of the orbits representing uveitis/scleritis and edema of the left optic nerve and (b) there is nodular enhancement with mild dural thickening on the quadrigeminal cistern involving the left cerebellar tentorial leaflet, with a central hypointense nonenhanced area showing mild diffusion restriction representing a microabscess.
Based on these findings, the diagnosis of NB was confirmed. The patient was initiated on a regimen of quadruple antibiotic therapy, consisting of ceftriaxone (100 mg/kg/day BID), gentamicin (7.5 mg/kg/day TID), rifampin (15 mg/kg/day BID), and doxycycline (5 mg/kg/day BID), along with dexamethasone (0.6 mg/kg/day QID for 5 days). Clinical improvement was observed within the first week, with complete resolution of the squint. Intravenous antibiotics were continued for 1 month, after which the patient was transitioned to oral antibiotics for a total duration of 6 months. Follow-up MRI after 1 month of therapy showed resolution of the previously noted abnormalities (Figure 2). Figure 3 summarizes the timeline of the patient’s clinical course, diagnostic interventions, and treatment milestones.
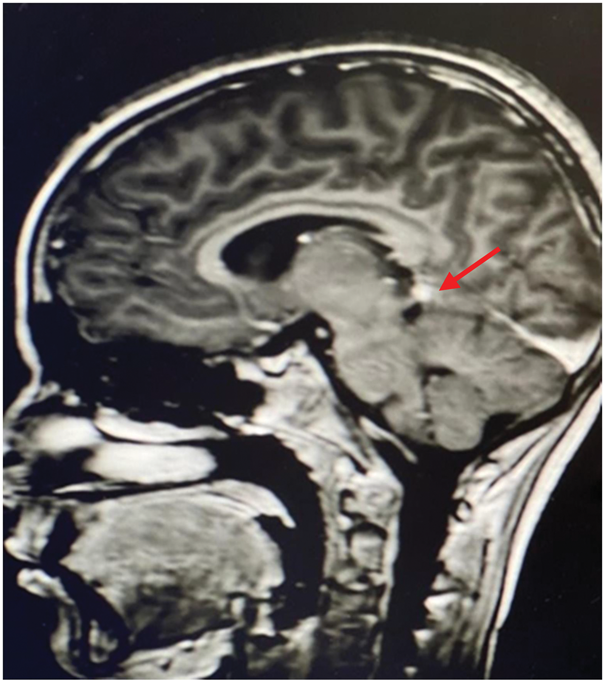
Interval regression of the dural nodule on the quadrigeminal cistern involving the left cerebellar tentorial leaflet, with no diffusion restriction, indicating radiological healing.

Flowchart summarizing the clinical course, diagnostic interventions, and treatment milestones in a pediatric case of neurobrucellosis. Key events are shown from symptom onset (Day 60) through hospital admission, diagnostic LP, brain MRI, initiation of quadruple antibiotic therapy, clinical improvement, discharge, follow-up imaging, and completion of total therapy at 6 months (Day 180). LP: lumbar puncture; MRI: magnetic resonance imaging.
Discussion
This case involves an 11-year-old boy with a history of raw milk ingestion and camel contact, presenting with systemic symptoms such as bone and muscle pain (nonspecific) and neurological symptoms including frontal headache, vomiting, double vision, ophthalmoplegia, and transient aphasia. The patient’s family history of brucellosis further supported the suspicion of NB based on his clinical presentation.
NB is a rare complication of brucellosis, with meningitis being the most prevalent manifestation. A survey of 187 cases across 37 articles revealed that 37% of NB cases presented with meningeal irritation, which can occur in both acute and chronic forms of the disease. 1 Similarly, meningoencephalitis involvement was observed in a NB case previously reported in Saudi Arabia (Table 2), different from the present case.
Previously reported cases of pediatric neurobrucellosis from Saudi Arabia.
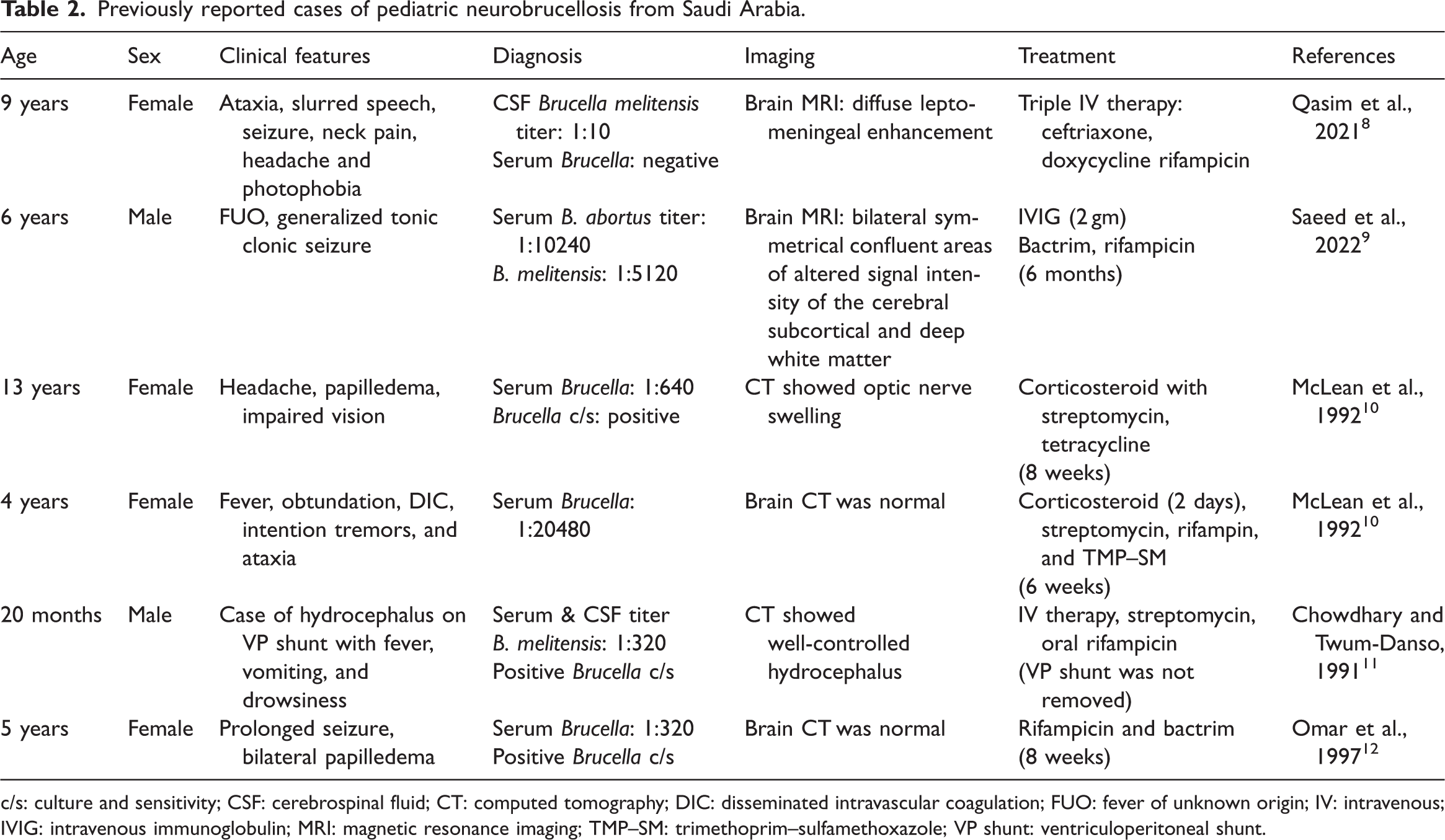
c/s: culture and sensitivity; CSF: cerebrospinal fluid; CT: computed tomography; DIC: disseminated intravascular coagulation; FUO: fever of unknown origin; IV: intravenous; IVIG: intravenous immunoglobulin; MRI: magnetic resonance imaging; TMP–SM: trimethoprim–sulfamethoxazole; VP shunt: ventriculoperitoneal shunt.
In this case, the diagnosis was established using a combination of clinical suspicion, positive agglutination with Coombs tests, brain MRI, and CSF analysis. Diagnosing NB is challenging due to its nonspecific clinical presentation, requiring a high index of suspicion, particularly in endemic areas. 13 Diagnostic criteria include the following: neurological symptoms and signs unexplained by other diseases, positive blood or CSF culture for Brucella organisms, increased CSF protein and lymphocytic pleocytosis, and improvement in clinical and CSF parameters following specific antibiotic therapy. 13
The CSF analysis in this case revealed low glucose levels, high protein levels, and lymphocytic pleocytosis (86% lymphocytes and 6% polymorphonuclear cells). However, these findings are nonspecific and can also occur in other chronic meningitis etiologies such as neurocryptococcosis and tuberculosis. 14 Infections such as neurocryptococcosis and tuberculosis were ruled out through negative CSF multiplex PCR, Ziehl–Neelsen staining, and cultures.
The patient’s standard agglutination test showed significant titers of 1:640 for B. melitensis and B. abortus in the serum using the Coombs test. ELISA (IgM and IgG) kits were not available at our institution during the time of diagnosis. It is recommended to use a combination of diagnostic tests, such as standard tube agglutination with Coombs or ELISA, to improve diagnostic accuracy. 15
Definitive diagnosis of brucellosis requires bacterial isolation from the blood, CSF, bone marrow, or other tissues. However, the sensitivity of these cultures is low, ranging from 20% to 28%, with the lowest isolation rate observed in CSF samples (10%). 16 Automated cultures, which detect bacterial growth through CO2 production, offer slightly higher sensitivity (25%) compared with conventional cultures in CSF. 16 Therefore, serological testing is considered the cornerstone for diagnosing NB. 2
Brain MRI findings in this case demonstrated nodular enhancement with dural thickening in the quadrigeminal cistern, along with a central hypointense nonenhanced area showing diffusion restriction consistent with a microabscess. Additional findings included swelling and edema of the left optic nerve. Imaging studies, such as the Istanbul-3 study, reported MRI abnormalities in 45% of NB cases, including meningeal enhancement, arachnoiditis, granulomas, white matter lesions, brain abscesses, cranial nerve involvement, and vascular changes. 17 Differential diagnoses for such MRI findings include infections such as tuberculosis and granulomatous diseases such as sarcoidosis. 17
The patient in this case was treated with a regimen of quadruple antibiotics—ceftriaxone, gentamicin, rifampin, and doxycycline—along with dexamethasone for 5 days. NB poses unique challenges due to the ability of Brucella to evade the immune system and proliferate throughout the body, making recurrence common and treatment difficult. 18 There are no randomized controlled trials to guide the choice, dosage, or duration of antibiotic therapy for NB. However, sufficient antibiotic penetration into the central nervous system is crucial. Treatment typically involves a combination of three antibiotics administered until clinical symptoms resolve and CSF parameters normalize. 19
The patient showed significant clinical improvement within 1 week, with resolution of the squint and improvement of CSF analysis and MRI findings after 1 month of treatment. It is recommended that patients be monitored every 3 months to detect potential relapses, which occur in 5%–15% of cases, usually within the first 6 months post-treatment. 20 If clinical symptoms persist or CSF abnormalities remain, extending antibiotic therapy for an additional 6 months is advised. 20
This case highlights the importance of early detection and timely intervention in NB, particularly in endemic regions, to prevent long-term neurological sequelae.
Conclusions
Brucellosis, with its diverse clinical presentations, can mimic various systemic illnesses, leading to diagnostic delays and potentially severe complications. NB, although rare, should be considered in the differential diagnosis of patients presenting with unexplained neurological symptoms, particularly in endemic regions, as its nonspecific presentation often results in misinterpretation and missed diagnoses. Timely recognition and prompt initiation of appropriate antibiotic therapy are critical in managing NB and minimizing its long-term neurological sequelae. Physicians must maintain a high index of suspicion and utilize a combination of clinical, serological, microbiological, and imaging studies to confirm the diagnosis. Early intervention remains pivotal in improving patient outcomes and preventing relapses.
Footnotes
Acknowledgements
The authors acknowledge our patient and his family for their kind participation in this work.
Authors’ contributions
All authors contributed substantially to writing of the manuscript, literature review, concept and design, acquisition and interpretation of data, drafting of the article, critical revision of the article for important intellectual content, and final approval of the version to be published.
Data availability statement
All data and materials related to the study are included in the current manuscript.
Declaration of conflicting interests
All authors declare no competing interests related to the study.
Funding
No funds were available for the current research.
