Abstract
Objectives
Schizophrenia is a debilitating psychiatric illness that is often understudied due to stigma and cultural barriers. This report presents a clinical descriptive evaluation of patients with schizophrenia in Saudi Arabia, focusing on demographics, clinical information, and treatment efficacy and safety.
Methods
This single-center cross-sectional study included 42 patients on antipsychotics for at least 6 months, with a male-to-female ratio of 1:1 and a mean age of 36 years.
Results
Most patients were single (64.3%) and unemployed (51.2%), with 52.4% having only a high school education level. The median duration of antipsychotic treatment was 21.2 (1.7–227.5) months. Antipsychotic combination therapy was the most common intervention (59.5%). Efficacy results from the Positive and Negative Syndrome Scale showed complete remission in 4.8%, partial remission in 11.9%, moderate remission in 33.3%, and no improvement in 50% of the patients. Safety evaluation via the modified Simpson–Angus Scale indicated that 71.4% of the patients had normal scores, 26.2% experienced minimal movement disorders, and only 2.4% had clinically significant movement symptoms.
Conclusion
This preliminary investigation examined the demographics and clinical features of patients with schizophrenia as well as efficacy and tolerability of relevant treatments. Larger studies are needed to understand this population comprehensively.
Introduction
Schizophrenia is a serious mental illness that throws a person’s entire mental state into disarray. It is characterized by a mix of positive symptoms, negative symptoms, and cognitive features, including distorted thinking and memory, changes in emotions, altered perception of reality, and disrupted behavior such as social withdrawal and apathy. 1 It is a chronic and substantially debilitating condition, which may involve repeated episodes of worsening symptoms.1,2 The main sign of schizophrenia is psychosis, which includes delusions, auditory hallucinations, and fixed false beliefs.2,3
Schizophrenia has a lifetime risk of approximately 0.5%–1.0%. Owing to its early onset and proclivity for chronicity, its prevalence is relatively high in both men and women. 2 A substantial number of people (∼24 million or 1 in 300) live with schizophrenia worldwide. 4 This number rises to 1 in 222 among adults (0.45%).4–6 Based on a cross-sectional study at six hospitals in the five main regions of Saudi Arabia, the most frequently reported psychiatric diagnosis among inpatients was schizophrenia (55.8%), followed by bipolar disorder (23.3%) and major depressive disorder (7.2%). In contrast, the most common psychiatric diagnosis among outpatients was major depressive disorder (29.3%), followed by schizophrenia (28.9%). 7 However, based on a more recent Saudi National Mental Health Survey, schizophrenia was considered as one of the mental disorders with the least 12-month persistent prevalence (<2.1%). Comparatively, anxiety disorders were ranked as the most common class among lifetime prevalence disorders (12.3%). Notably, the prevalence of both conditions was slightly higher than the estimates obtained by the World Mental Health Surveys Initiative in other high-income countries (8.3%, range: 6.5%–13.7%). 8
This illness has a massive social and economic impact, and its effects on patients and their families can be extremely detrimental. 1 Genetics is a vital contributor to schizophrenia, likely responsible for 80% of the cases. However, environmental and psychosocial factors also play a crucial role.1,9 Additionally, emerging evidence links the gut microbiome to schizophrenia, and disturbances to the gut microbiome can disrupt the gut–brain axis and lead to psychosis. 10
Schizophrenia should be managed using a comprehensive approach. This may include medication (primarily antipsychotics) and psychosocial interventions such as psychoeducation, which can improve the patient’s understanding of their illness. Additionally, family support and programs promoting independent living skills (psychosocial rehabilitation) can be crucial for overall well-being. 11 The course of treatment for a patient is charted by considering several factors, including illness acuity/severity, patient’s history of medications, effectiveness of various medications and their side effects, and patient’s preferences and ability to follow the treatment plan. 1
There are two main classes of drugs used to treat schizophrenia, first-generation antipsychotics (FGAs) and second-generation antipsychotics (SGAs).1,12 SGAs are the preferred line of treatment over FGAs in clinical practice, as most of the guidelines recommend SGAs as the treatment of choice. However, FGAs are less costly and are still used worldwide.1,13,14
Studies in Saudi Arabia consistently highlight the prominent role of antipsychotics in treating mental health conditions. One study by Alosaimi et al. 6 found antipsychotics to be the most prescribed medication, exceeding others by 6.6%. Furthermore, a more recent study by Alsabhan et al. 15 suggests a preference for atypical antipsychotics for treating schizophrenia (68%). Supporting this preference, Zubair et al. 16 found atypical antipsychotics to be superior to FGAs (typical antipsychotics) in terms of success rates, relapse rates, and tolerability for patients with schizophrenia.
While research 13 has suggested that some antipsychotics are slightly more effective than others, these differences are often minor. Overall, various studies have not found significant differences between typical and atypical antipsychotics about how well they treat immediate symptoms or how often people stop taking them. 16 However, the types of side effects do differ. Interestingly, one meta-analysis found that atypical antipsychotics like olanzapine, amisulpride, and risperidone may be more effective than typical medications. 17 Further supporting the use of atypical antipsychotics, Leucht et al. 18 reported lower treatment failure rates (53%) and relapse rates (14%) compared with typical antipsychotics (70% and 23%), respectively.
Schizophrenia treatment effectiveness can be measured using the Positive and Negative Syndrome Scale (PANSS); this scale evaluates a wide range of symptoms experienced by schizophrenia patients. 19 A successful treatment response typically involves a marked improvement in these symptoms, reflected by a minimum 20%–50% reduction in the patient’s initial PANSS score. 19 Many studies endorse PANSS as a reliable measure for evaluating antipsychotic efficacy in clinical trials. 20
Antipsychotics, although effective, can cause movement disorders as a serious side effect. These disorders include parkinsonism, akathisia, tardive dyskinesia, and dystonia. 21 Although newer atypical antipsychotics offer a considerable advantage over FGAs with their reduced side effect profile, they still carry some potential risks. 22 Research by Weng et al. 23 suggested that FGAs are more likely to cause movement disorders compared with newer ones. Although atypical antipsychotics are generally considered to have fewer side effects, a previous study suggested a more cautious interpretation. A study by Hugenholtz et al. 24 found that some clinical trials did not compare antipsychotics fairly. In these trials, patients were given higher doses of typical antipsychotics than recommended, with no additional medications that could help manage extrapyramidal side effects (EPS). This difference in treatment might explain why atypical antipsychotics seemed safer. Studies have also suggested that some patients are more prone to these antipsychotic-induced movement disorders (e.g. antipsychotic-induced parkinsonism (AIP)). These factors include younger or older age, female sex, and treatment with conventional antipsychotics. 23 The Simpson–Angus Scale (SAS) is a 10-item rating scale that is commonly used in both clinical settings and research to assess the severity of EPS. 25
Moreover, antipsychotics, particularly SGAs, are reportedly associated with significant metabolic side effects, including weight gain, obesity, diabetes mellitus, and dyslipidemia.26,27 These adverse effects not only increase the risk of cardiovascular disease and diabetes but can also impair patient adherence to treatment. 26
This study investigated the prescribing patterns of antipsychotics and their relationship with various patient demographics (e.g. age and sex) and clinical factors (e.g. illness severity and medical history). Additionally, this study used established tools to assess the safety and effectiveness of the medications.
Objectives
The current study focuses on three main objectives. First, this study aimed to describe the pattern of antipsychotics (types and dosages) in Saudi patients with schizophrenia, considering patients’ demographics and clinical factors. The second aim was to provide an early description of the effectiveness of antipsychotics using the PANSS. 19 Finally, we provided preliminary data about treatment safety, particularly by determining the frequency of EPS associated with different medications using the modified Simpson–Angus Scale (MSAS). 25 In the future, we will examine existing data on metabolic problems.
Methods
Study design
This single-center cross-sectional study enrolled 42 individuals with schizophrenia who were recruited from the Security Forces Hospital (SFH) in Riyadh, Saudi Arabia. The study was performed from January 2023 to October 2023. The reporting of this study conforms to Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 28 Written informed consent was obtained from the patients after interviewing them in their scheduled psychiatry clinic. Psychiatric consultants (MِِA and NAB) then administered two validated assessment tools, the PANSS and MSAS, to evaluate the effectiveness and safety of the current antipsychotic therapy. Additional demographic and clinical information, such as concurrent medications, coexisting medical conditions, antipsychotic dosage and duration, medical history, laboratory results, social status, education level, and sex, was retrieved from the patient’s electronic medical records maintained at the clinic. This study was conducted in accordance with the Helsinki Declaration of 1975 as revised in 202413.
Participants
This pilot study recruited 42 of the 50 outpatients diagnosed with schizophrenia (based on The Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria) between January 2023 and October 2023 from the SFH in Saudi Arabia. Participants were included if they were aged between 18 and 65 years, had been taking antipsychotics continuously for at least 6 weeks, had at least an elementary school education level, and did not have visual or auditory impairments. Patients were excluded if they had dementia or substance-induced mental disorders or refused to participate in the study. The chlorpromazine-equivalent (CPZE) daily dose was calculated according to published guidelines. 29
Measures
PANSS
The PANSS, a 30-question interview, is the leading tool for assessing schizophrenia and antipsychotic effectiveness. It combines elements from other scales and uses a detailed 7-point rating system (1 = absent, 7 = extreme) to determine symptom severity. 19 This tool is considered the gold standard for evaluating the efficacy of antipsychotics as well as their effectiveness in clinical practice. 20 The PANSS requires a 30–40-min interview in which the patient is assessed for the presence and severity of psychopathology. The PANSS instrument provides a complete definition of each item as well as detailed anchoring criteria for each of the seven rating points: 1 = absent, 2 = minimal, 3 = mild, 4 =moderate, 5 = moderate–severe, 6 = severe, and 7 = extreme. In the 30-item scale, 7 items were related to positive symptoms (hallucinations, delusions, and other distortions of reality), 7 to negative symptoms (flattened emotions, social withdrawal, and lack of motivation), and 16 to general psychopathology (anxiety, depression, and other mental health concerns). Additionally, a composite scale for effectiveness was derived by subdividing the total score of positive symptoms by 7, the total score of negative symptoms by 7, the total score of positive and negative symptoms by 14, and the total score of general symptoms by 16. In this study, patients’ effectiveness was classified into four categories: (a) complete remission, if the mean score was ≤2; (b) partial remission, if the mean score was 2.1–3.9; (c) moderate remission, if the mean score was 4; and (d) no improvement, if the mean score was >4. These standards were previously described in a consensus report on remission based on proposed criteria and rationale for schizophrenia symptomatology improvement. 30
MSAS
Antipsychotics, although effective for schizophrenia, can disrupt movement due to blocking dopamine in the brain’s nigrostriatal pathway, leading to Parkinson’s-like symptoms. This side effect, known as drug-induced parkinsonism (DIP), can be serious. The motor dysfunction caused by DIP can be as severe as those in Parkinson’s disease and may continue even after stopping the medication. 31 The MSAS combats this by assessing these side effects through a 10-item rating scale system.20,25 This widely used 10-point rating system (valuable in both research and clinical settings) condenses the original 10-item assessment in SAS into a more efficient way for detecting severity of movement disorders linked to antipsychotics. 20 It evaluates the core aspects of movement such as gait abnormalities, stiffness in limbs (arms, shoulders, elbows, and wrists), neck mobility, facial muscle control, tremors, excessive drooling, and even akathisia (restlessness and an urge to move). Furthermore, this focused approach allows for a quicker assessment of potential side effects from these medications. This scale assigns scores to movement disorders according to severity on a 5-point scale (0–4), with descriptive anchors for each point and a clearly described examination procedure for each item. A total score of 0–40 is calculated. Scores below 3 indicate normal function, whereas scores of 3–5 suggest a minimal issue. Scores ranging from 6 to 11 signify a clinically relevant movement disorder, and scores of 12 and above indicate a severe degree of movement disorder. This scoring system helps healthcare professionals assess the impact of antipsychotics and determine if adjustments are needed.
Statistical analysis
Statistical analysis was performed using SPSS software version 29.0.1.1. Descriptive statistics were calculated for all variables in the study, and the normality of the variables within each group was verified by the Kolmogorov–Smirnov test. Thereafter, a parametric test was employed when the variable followed a normal distribution, whereas a nonparametric test was used when the variable did not follow the normal hypothesis. The parametric tests used were the analysis of variance to compare three means or more and the Student’s t test to compare two means. The nonparametric test used was the Spearman test for the correlation between two continuous variables. Multivariate regressions, univariate, chi-square, and Fisher’s exact test were conducted to examine the correlation of the total PANSS score or the total MSAS score as dependent variables, with continuous or categorical study variables that showed P-values of <0.1 in the bivariate analysis as independent variables, to eliminate potential confounding factors as much as possible. A P-value of ≤0.05 was considered to indicate statistical significance.
Results
Study participants
Baseline characteristics of study participants
Table 1 demonstrates the demographic characteristics of the study participants. A total of 42 patients with schizophrenia were included, with an average age of 36 ± 11.08 years. The ratio of males to females in the study was 1:1, and most of them were single (64.3%). Approximately 62% of the participants had a high school education level, and 51.2% were unemployed. Antipsychotics were used by all participants.
Characteristics of patients with schizophrenia in the sample.

CPZE: Chlorpromazine-equivalent.
As shown in Figure 1, paliperidone (10%) was the most common single medication, followed by risperidone (7%), aripiprazole (5%), and quetiapine (5%). Remarkably, the majority of patients with schizophrenia administered combination therapy (59.5%), with aripiprazole–risperidone being the most frequent combination (12%) in the current study (Figure 1).

Pattern of most frequently used antipsychotics either as a monotherapy or a combined therapy (more than one).
As depicted in Table 1, over two-thirds of the patients (66.7%) used medications other than antipsychotics. These included antidepressants (N = 15 (35.71%)), anticonvulsants (N = 7 (16.6%)), and beta-blockers (N = 3 (7.14%)). Notably, anticholinergics (such as procyclidine) were used by over half (N = 22 (52.38%)) of the patients, whereas benzodiazepines were used by only 14.3%. The median (range) duration of antipsychotic treatment was 21.2 (227.5 ) months, with typical antipsychotics having a lower average dose of 100 (975) mg and atypical antipsychotics having a higher average dose of 600 (15433.33) mg.
Baseline clinical laboratory data
The clinical laboratory data of the study participants are presented in Table 2. The study found a high prevalence of obesity in patients with schizophrenia, with an average body mass index exceeding 30. Additionally, 73% of the participants had elevated prolactin levels, a hormone potentially impacted by antipsychotics. However, the other laboratory tests included routine blood tests such as lipid profiles, blood sugar, and blood cell counts, all of which were within normal ranges.
Clinical laboratory investigation.

BMI: body mass index; HDL: high-density lipoprotein; LDL: low-density lipoprotein; HGB: hemoglobin; MCV: mean corpuscular volume; HCT: hematocrit; FBG: fasting blood glucose.
Normal ranges: BMI (18.5–24.9 kg/m2); prolactin level for males (86–324 μIU/L) and females (102–496 μIU/L); total cholesterol (0.00–5.19 mmol/L); triglyceride (desirable <1.7 mmol/L); LDL (0.00–2.59); HDL (>1.68 mmol/L); HGB for males (14–18 g/dL) and females (12–16 g/dL); HCT for males (41%–50%) and females (36%–48%); MCV (75–95 fL), FBG (4.1–6.1 mmol/L).
Bold values indicate abnormal levels.
Study measures
PANSS observed scores
The study assessed patients’ symptoms using PANSS. Most PANSS scores (positive symptoms, negative symptoms, total positive and negative symptoms, and composite scale) were normally distributed according to the Kolmogorov–Smirnov test, as shown in Table 3, and averaged as follows: (a) positive symptoms (29 ± 8.62); (b) negative symptoms (28 ± 8.79); (c) total positive and negative symptoms (57 ± 14.9); (d) general symptoms (46.69 ± 14.2); and (e) overall score (3.8 ± 0.8).
Results of the PANSS.

Kolmogorov–Smirnov test.
PANSS: Positive and Negative Syndrome Scale.
The study then classified patients based on their total PANSS score, with 4.8% achieving complete remission (lowest score), 11.9% achieving partial remission (mild symptoms), 33.3% showing moderate improvement, and a significant proportion (50%) of patients showing no improvement based on their PANSS score (Figure 2). These results indicate that although some patients benefited from treatment, a substantial proportion of patients required further evaluation or medication adjustments.

Bar chart of Positive and Negative Syndrome Scale (PANSS) classification of participants according to the mean score of improvement in symptoms (complete remission, mean score ≤2; partial remission, mean score = 2.1–3.9; moderate remission, mean score = 4; no improvement, mean score >4).
MSAS observed scores
The study used MSAS to assess movement disorders potentially caused by antipsychotics. The average score was 2 (standard deviation, 1.95), with a median score of 1. The scores ranged from 0 to 8. Interestingly, the distribution of these scores leaned toward higher values (positively skewed), as shown in Figure 3. According to the classification of MSAS severity score, most patients (71.4%, N = 30) had a score below 3, indicating normal movement. Moreover, 11 (26.2%) patients scored between 3 and 5, suggesting a minimal degree of movement disorder. Only one (2.4%) patient had a score exceeding 5, which is considered a clinically significant degree of movement disorder according to the MSAS scoring system.

Histogram of modified Simpson–Angus Scale (MSAS) score of severity. The scores were classified as follows: <3 (normal), 3–5 (patient has a minimal degree of movement disorder), and 6–11 (patient experiences a clinically significant degree of movement disorder).
Baseline characteristics associated with PANSS scores
Univariate analysis of mean PANSS scores
As shown in in Table 4, univariate analysis of continuous variables associated with mean PANSS scores revealed a possible association between symptom severity (PANSS score) and movement disorders (MSAS score). Patients with more severe symptoms tended to experience slightly more movement problems. However, this weak positive correlation was not statistically significant (P = 0.067, borderline significant). No other statistically significant associations were found between the PANSS score and other factors, including CPZE daily dose, typical and atypical CPZE, age, diagnosis year, or onset age of schizophrenia. Although some variables showed negative trends, potentially indicating less severe symptoms with higher values, none of them reached statistical significance (P > 0.05).
Univariate correlation between the PANSS overall mean score and population characteristics (continuous variables).
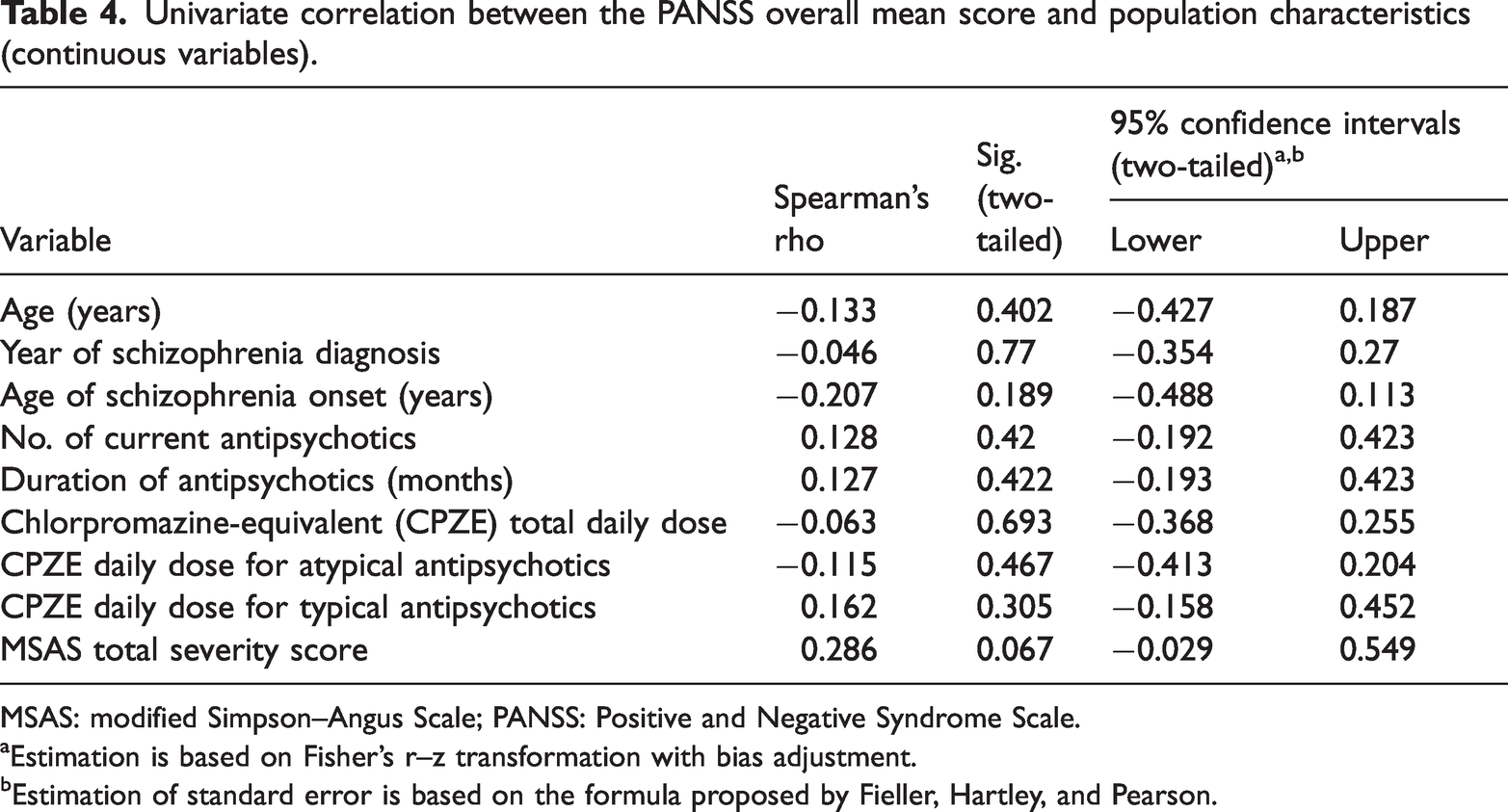
MSAS: modified Simpson–Angus Scale; PANSS: Positive and Negative Syndrome Scale.
Estimation is based on Fisher’s r–z transformation with bias adjustment.
Estimation of standard error is based on the formula proposed by Fieller, Hartley, and Pearson.
Univariate analysis of PANSS effectiveness category
Table 5 represents the univariate correlation between the effectiveness category (based on the mean overall score) and categorical variables. Interestingly, males showed a significantly better response to antipsychotic treatment compared with females (P < 0.05), whereas other categorical variables were not shown to have any association with the effectiveness category.
Univariate correlation between PANSS overall score (effectiveness category) and population characteristics (categorical variables).
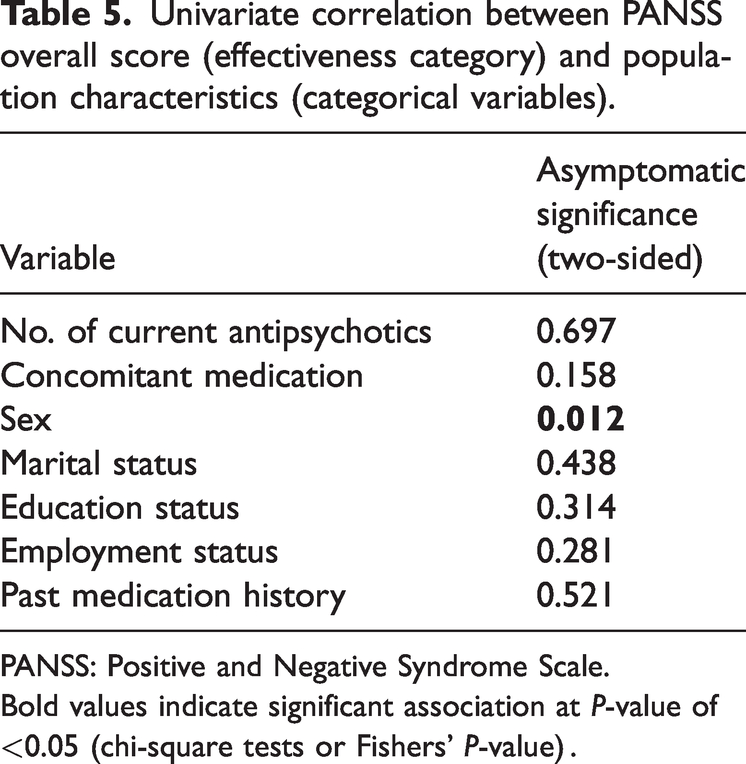
PANSS: Positive and Negative Syndrome Scale.
Bold values indicate significant association at P-value of <0.05 (chi-square tests or Fishers’ P-value) .
Generalized linear model (GLM) analysis of variables
GLM analysis revealed sex as a significant predictor of treatment response in schizophrenia (Tables 6 and 7). Males were more likely to respond to treatment and have less severe symptoms compared with females, with a high statistically significant probability (P = 0.023, odds ratio (OR) = 0.128, 95% confidence interval (CI): 0.22–0.758). This effect was particularly evident for positive and negative symptoms (P = 0.025, OR = 0.107, 95% CI: 0.015–0.75).
Predictors of no improvement based on PANSS overall score (effectiveness category) using a generalized linear model.

PANSS: Positive and Negative Syndrome Scale; OR: odds ratio.
The model was obtained by using generalized linear regression.
Bold values indicate significant association at P-value <0.05.
Predictors of no improvement based on negative, positive, total, and general symptoms (effectiveness category) using a generalized linear model.

The model was obtained using generalized linear regression.
Bold values indicate significant association at P-value <0.05.
Meanwhile, another significant finding was the association between education level and treatment outcome. Patients with only elementary/primary school education level were more likely to experience no improvement in general symptoms (P = 0.023, OR = 161.714, 95% CI: 2.014–12983.68). Although not statistically significant, high school education level also showed a trend toward worse outcomes (P = 0.06, OR = 46.386, 95% CI: 0.845–2546.14). Other variables showed trends indicating that they influence treatment response (positive or negative effects on different symptom categories); however, these trends were not statistically significant (P > 0.05).
Discussion
This cross-sectional study investigated antipsychotic prescribing patterns among a sample of Saudi patients with schizophrenia, revealing the effectiveness and safety of antipsychotics utilizing established tools.
Analysis of demographic characteristics showed that the mean and standard deviation of the age of the study patients was 35.69 (±11.084) years, which was comparable to that reported in other previous studies.32,33 Schizophrenia typically manifests itself between the late teens and the mid-30s. In the present study, schizophrenia onset was mostly found to start at the age of 23.79 ± 44 years, which falls within the established range of schizophrenia onset. Intriguingly, 16% of the participants with schizophrenia reported a family history of the disorder, highlighting the potential role of genetics, which is a well-established risk factor for the condition.
Antipsychotics are the core of schizophrenia management; the physician must assess numerous individual factors of the patient to select an appropriate antipsychotic, based on its efficacy, safety, and tolerability to patients as well as its cost-effectiveness.32,34 SGAs are preferred over FGAs. Researchers have reported that SGAs are either similarly effective or modestly better than FGAs in terms of EPS and efficacy, as proven by various clinical trials.32,35 In contrast, our study revealed that atypical antipsychotics were prescribed to most of the study participants.
Approximately 88% of the patients used SGAs such as paliperidone, risperidone, or aripiprazole as either monotherapy or in combination treatment, such as the combination of aripiprazole with risperidone, which was more common than other combination forms, with no consideration for the administration route. Another study showed a similar pattern of prescribing antipsychotics at King Saud University Medical City in Saudi Arabia. Aripiprazole was the most frequent antipsychotic drug prescribed to patients with schizophrenia, followed by haloperidol and risperidone, 15 which aligned with our findings.
Antipsychotic doses in our study were converted to CPZE doses. The median CPZE daily dose of antipsychotics was 600 (range: 15433.3) mg, which is considered one of the factors associated with successful dose reduction and neurocognitive improvement, as suggested by Tani et al. 36 According to Tani et al., 36 lowering antipsychotic doses beyond 200 mg/day (CPZE) might improve cognitive function in patients with schizophrenia. However, this benefit comes with a potential worsening of symptoms. Interestingly, a subgroup analysis within this study found that maintaining antipsychotic doses above 200 mg/day CPZE while gradually reducing the dose might not worsen relapse risk compared with keeping a steady dose. Patients with chronic schizophrenia may also benefit from antipsychotic dose reduction. Studies, including the study by Tani et al., 36 have suggested that patients’ cognitive function can be preserved even after 10 years of illness, making them suitable candidates for dose reduction.37,38 Our participants, with a median illness duration of 7.5 years (range of up to 38 years), fall within this window.
Over a median of 1.77 years (21.2 months), participants received antipsychotics in our study. However, the total treatment time spanned nearly 44 years (227.5 months), suggesting significant individual differences in treatment duration. Early identifying antipsychotic nonresponders allows for quicker treatment adjustments, avoiding treatment with ineffective medications. 39 This study includes patients who received antipsychotics for at least 6 weeks, allowing sufficient time for clinical response to develop and accurately judge the effectiveness. This timeframe aligns with the recommendations of both the National Institute for Health and Care Excellence and the British Association of Psychiatry.40,41
In the current study, the post-treatment mean for PANSS overall general symptoms was 3.79 ± 0.86, suggesting moderate remission in response to their antipsychotic therapy. Yet we further found that effectiveness level is rated mostly as no improvements by 50% of cases in terms of either positive, negative, and total (positive and negative) mean scores (29 ± 8.62, 28 ± 8.79, and 57 ± 14.9, respectively). The lower the mean score, the better the improvement in schizophrenia symptoms. This observed discrepancy in the level of improvement among the three subdomains in the PANSS tool could be attributed to several factors. First, our research investigated the effectiveness and tolerability of six atypical antipsychotics (risperidone, olanzapine, aripiprazole, paliperidone, quetiapine, and clozapine) and four typical antipsychotics (haloperidol, chlorpromazine, zuclopenthixol, and trifluoperazine), which are known to produce variable effectiveness levels based on previous clinical trials.32,35,42 The study randomly enrolled 964 patients to receive olanzapine, risperidone, aripiprazole, or amisulpride. The nonresponder percentage was significantly higher in the aripiprazole group than in the other treatment groups (P < 0.001). All four antipsychotics significantly reduced PANSS total scores from baseline to week 8 (P < 0.01). In our multivariate analysis, subgroup analysis based on antipsychotic class or combinations was impossible due to small proportions within similar groups.
Second, our enrolled patients were at different durations of antipsychotic therapy, though mostly were in an advanced stage of management, which could partially explain the elevated mean scores obtained by their evaluators as rated on the three PANSS subdomains. According to Long et al., 42 the best way to predict whether patients would not respond to antipsychotics by week 8 was to look for minimal improvement in their PANSS scores at earlier weeks (<10% reduction at week 2 and <20% reduction at week 4). In the same study, among participants receiving atypical antipsychotics, 74% exhibited a positive response after 8 weeks, compared with 35.7% who did not respond. However, a systematic review and meta-analysis of randomized trials by Kishimoto et al. 43 indicated the potential of relapse and treatment failure among patients receiving antipsychotic therapy for a longer period than 12 months, though these outcomes were estimated to have higher trends among FGAs compared with SGAs. In our study, a greater proportion of patients (approximately 67%) were receiving antipsychotics for more than 12 months. This result could indicate the possibility of relapse and reduced effectiveness observed in some symptoms within either negative, positive, or combined subdomains in PANSS, thus elevating the individual mean scores of these constructs to a higher level than the overall PANSS measure.
Third, our study used similar antipsychotics (mostly atypical SGAs in 88% of participants), which are assumed to produce similar antipsychotic effectiveness. This assumption is based on findings from a previous clinical trial displaying similar overall score reductions (23%–26% in PANSS score) after 12 weeks across three SGD medications (olanzapine, risperidone, and aripiprazole), thus suggesting that all three drugs were equally efficacious. 32 In contrast, a more comprehensive analysis of rigorously selected placebo-controlled, double-blinded studies by Huhn et al., 14 including 32 oral antipsychotics for schizophrenia, revealed important differences in efficacy among all SGD drugs, and as compared with placebo. Clozapine, amisulpride, olanzapine, and risperidone significantly outperformed others for overall symptoms. Amisulpride, risperidone, olanzapine, paliperidone, and haloperidol were top performers for positive symptoms, whereas clozapine, amisulpride, and olanzapine were most effective for negative symptoms, with zotepine and risperidone showing lesser improvement. This conclusion suggests varying effectiveness among antipsychotics within SGDs, highlighting several particularly beneficial options for different symptom categories. 14 Interestingly, our research delves deeper than previous studies that focused on overall PANSS scores. 32 We examine individual aspects of the scale, negative symptoms, positive symptoms, and general psychopathology to facilitate comparisons with some other studies. 14 This comprehensive approach was thought to potentially lead us to gain a broader understanding and uncover new insights that have not been clearly resolved in other studies.14,32 Additionally, the serious concern is that 50% of patients still did not achieve satisfactory symptom improvement (PANSS) for positive or negative symptoms in our real-world practice. It could be attributed to our participants’ elevated baseline symptom severity (unmeasured in the current study), which may likely have influenced the response trajectories to different SGA drugs. Therefore, a careful individualization approach in drug selection based on initial baseline PANSS scores and regular monitoring of individual subdomain score reduction was undoubtedly needed in our future healthcare in this area of psychiatry practice.
Lastly, antipsychotic adherence is widely recognized as critical in evaluating clinical results, effectiveness, and safety. A recent study by Shilbayeh et al. 44 employed the Medication Adherence Rating Scale to assess patient adherence to antipsychotic treatment. Their findings revealed that adherence levels were predominantly categorized as low, with 64.4% of the sample study not adhering to treatment. Our current study, examining 42 patients from that pool, might also be affected by this adherence issue, which could explain limited symptom improvement in some of our patients.
Our analysis also revealed a weak positive association between total scores on the MSAS and PANSS; this suggests that for some patients, the long-term benefits of antipsychotics might be outweighed by the potential side effects. Although this finding only reached borderline statistical significance (P = 0.067), it warrants further investigation, particularly considering the novelty of this research on the association between antipsychotic side effects and symptoms’ severity.
Separately, various factors, including age, education, and social support, influenced the response to antipsychotic treatment, and sex emerged as the strongest predictor. Men, in particular, showed significantly better overall improvement (P = 0.023) and greater reduction in both positive and negative symptoms (P = 0.025) compared with women in analyses using GLM. Notably, education level also impacted outcomes, with individuals having limited education being most likely to see no improvement (P = 0.023) and those with some high school education showing a trend toward worse outcomes (P = 0.06).
However, a recent meta-analysis by Storosum et al., 45 encompassing 29 placebo-controlled studies, diverges from our findings regarding sex differences in antipsychotic response for schizophrenia. Their analysis suggests that women experienced a larger reduction in symptoms than men, based on ratings using the Brief Psychiatric Rating Scale. Our study yielded different results. Furthermore, research suggests a connection between educational level and medication compliance in individuals diagnosed with schizophrenia. This association might be explained by stronger cognitive abilities and a deeper understanding of the illness among those with higher education.46,47 However, it is crucial to acknowledge that other factors, such as socioeconomic background and medication characteristics, can also significantly influence treatment adherence. Additionally, some studies have raised concerns that limited disease insight and knowledge can potentially affect the accuracy of assessments performed in schizophrenia trials. 48
Furthermore, compliance with medication can be negatively impacted by antipsychotic-induced movement disorders (extrapyramidal symptoms) and adverse effects of antipsychotics, ultimately affecting the course of the illness. We assessed AIP using MSAS. This condition is characterized by slowness of movement (bradykinesia), trembling (tremor), stiffness (rigidity), and a hunched posture, in addition to problems such as difficulty walking (gait disturbance), excessive drooling (salivation), and seborrheic dermatitis. 49 AIP is prevalent in 19%–36% of patients with schizophrenia. 50 Most of our study participants (71.4%) showed no or minimal parkinsonism-like symptoms (tremor and rigidity), whereas 28.6% were experiencing EPS (such as dystonia, parkinsonism, and akathisia) or long-lasting and chronic tardive dyskinesia and dystonia. AIP can severely impair activities of daily life, and it can be treated or at least alleviated by anticholinergic drugs such as procyclidine, which were commonly prescribed in our study to prevent or treat the abovementioned side effects, with 52.38% of the patients taking procyclidine solely for symptom relief. Although treatments for EPS vary, the study suggested that anticholinergics were frequently used in this setting, even for preventive measures. Notably, atypical SGAs are generally considered less likely to cause EPS than typical FGAs. 51 In the present study, 88% of the participants were receiving atypical antipsychotics.
Multiple studies have investigated AIP across various medications. However, their findings vary considerably. A study by Gibert et al. 52 investigated the tolerability of quetiapine in an open-label, multicenter setting with 1961 participants. They used the SAS scale to evaluate tolerability. The results showed a significant decrease in average SAS scores, from a baseline of 6.8 (95% CI: 6.5–7.1) to 2.8 (95% CI: 2.6–3.0) at the 6-month interval (P < 0.05), indicating a substantial improvement in EPS among the patients. Moreover, in a separate longitudinal study by Jeste et al. 51 involving 122 patients, researchers compared the development of AIP between risperidone and haloperidol. They used the MSAS to assess both groups. Although the haloperidol group initially had a lower average score for EPS, statistical analysis revealed a higher risk of developing AIP over time in patients taking haloperidol than in those on risperidone (P < 0.05). This study suggests a link between medication and AIP development; however, it is possible that genetic variations also play a role in individual susceptibility to AIP. 50 We have not investigated this aspect in our current research.
Although FGAs effectively treat psychosis, they are often associated with side effects such as involuntary movements and increased prolactin levels. Newer medications (SGAs) tend to cause more weight gain and drowsiness.14,15 However, a surprising finding emerged in our study. Despite using SGAs, which are generally linked to lower prolactin elevation, nearly half of the participants (52%) exhibited an abnormally high level of prolactin.
Employing both PANSS and MSAS, our research is the first to provide a comprehensive picture of the therapeutic potential and safety profiles of diverse antipsychotics in Saudi Arabia. This study, using PANSS and MSAS, sheds light on patient responses to various treatments. It highlights improvement in some patients and also reveals a concerning subgroup that shows no improvement despite medication. A more extensive and frequent follow-up study is warranted to uncover the reasons behind this lack of response, ultimately aiming to personalize and improve care for those suffering from this psychotic disorder.
This study has potential limitations. Although we included all available antipsychotics in our single-center study, the small sample size of the participants restricts the applicability of our findings to broader Saudi settings. Furthermore, due to the cross-sectional design and short data collection period, our study might not fully capture the long-term effectiveness and safety profile of the investigated antipsychotics. Furthermore, limited data in certain categories may have led to some inconclusive results. Although the use of GLMs is appropriate, some studies reported that ORs have wide CIs, likely due to the small sample size. We need more exhaustive multicenter studies to validate our findings. Additionally, our study’s focus on outpatients potentially excludes patients with more severe schizophrenia, thus limiting the generalizability of our findings.
Conclusion
The preliminary findings of this study revealed the unsatisfactory antipsychotic effectiveness in patients with chronic schizophrenia, which warrants urgent further large-scale investigations to identify underlying significant demographic, clinical, and dosage regimen factors as predictors of optimum effectiveness.
Summary points
Male sex was significantly associated with reduced overall PANSS score as well as an improvement in the effectiveness of the total symptoms. Approximately 52% of the patients were maintained on anticholinergic medications for their EPS. This finding could be a reason why most of them have not had any EPS during antipsychotic therapy, revealing a positive result. A noticeable increase in prolactin levels in 52.3% of the patients warrants further investigation of the causes and mechanisms underlying this high prevalence. A positive correlation was noted between total MSAS and PANSS scores, indicating some degree of no benefit-risk ratio for chronic antipsychotic use in our patients.
Footnotes
Acknowledgments
The current work was supported by the Princess Nourah bint Abdulrahman University Researchers Supporting Project, number (PNURSP2025R814), Princess Nourah bint Abdulrahman University, P.O. Box 84428, Riyadh 11671, Saudi Arabia.
Authors’ contributions
Conceptualization, S.S., M.A., H.A.A, and N.A.; Methodology, S.A., R.S., O.G., K.M.A., and N.A.; Validation, A.Y.B., F.A., and S.S.; Formal analysis, A.Y.B., S.S., and F.A.; Recruitment & clinical investigation, S.A., N.A., M.A., and F.A.; Resources, S.S., N.A.M., K.M.A., and H.A.A.; Data curation, S.A., F.A., and A.Y.B.; Writing–original draft preparation, F.A., A.Y.B., K.M.A., H.A.A., K.M.A., and S.A.; Writing–review & editing, all authors; Visualization, F.A., N.A.M., and H.A.A.; Supervision, S.S. and O.G.; Project administration, S.S.; Funding acquisition, H.A.A. All authors have revised and approved the final manuscript.
Data availability
Required information is available upon request to the authors.
Declaration of conflicting of interests
None.
Ethical considerations
The study was approved by the Institutional Review Board (IRB) committee of Security Forces Hospital (SFH) in Saudi Arabia (IRB accreditation number (H-01 -R-069)). All procedures were performed in accordance with the National Ethical Standards Committee guidelines. All enrolled patients showed the capacity to understand and complete the questionnaire and provide written and verbal informed consent.
Funding
Not applicable.
