Abstract
Objective
This study aimed to explore the association between pan-immune-inflammation value and heart failure outcomes using data from the nationally representative National Health and Nutrition Examination Survey database.
Methods
We conduct a cross-sectional cohort analysis using National Health and Nutrition Examination Survey data, including participants aged ≥20 years with available pan-immune-inflammation value data. We performed smooth curve fitting and threshold analysis, using both linear and non-linear regression models, to assess dose–response relationship and explore the continuous effect of pan-immune-inflammation value on heart failure outcomes.
Results
Our analysis revealed a significant and independent non-linear association between elevated pan-immune-inflammation value levels and an increased risk of heart failure occurrence. After adjustment for multiple covariates, these findings remained consistent and each increment unit in logarithmic pan-immune-inflammation value is associated with 34% increase in the risk of heart failure occurrence. Furthermore, we identified an inflection point of logarithmic pan-immune-inflammation value = 5.98 as a critical threshold. Stratified analyses revealed that the association between pan-immune-inflammation value and heart failure occurrence remains consistent across different subgroups.
Conclusions
This study confirmed the clinical value of pan-immune-inflammation value as a novel inflammatory biomarker in the assessment and monitoring of heart failure.
Keywords
Introduction
Heart failure (HF) is a global clinical syndrome of pandemic concern, with more than 56 million people affected worldwide. Its occurrence is expected to raise due to improved survival rates facilitated by evidence-based treatments and longer expectancy of general population. 1 HF is the final stage of heart disease and has a high rate of readmission and mortality, with approximately 50% of patients re-admitted within 1 year following their initial diagnosis and more than 50% dying within 5 years of survival. 2 The poor prognosis of HF imposes a huge medical burden on both patients and global healthcare systems. Therefore, it is necessary to strengthen the early identification and treatment of HF, so as to reduce the rate of readmission and mortality of patients.
There is evidence that patients with systemic inflammatory disorders have a substantially higher risk of developing cardiovascular disease than the general population, suggesting that inflammatory variables can be used as predictive indicators for cardiovascular disease. 3 Lately, the integrated prognostic scores depending on hematological parameters within peripheral blood counts have elicited substantial interest as indirect indicators of cancer-related inflammatory responses.4–6 The pan-immune-inflammation value (PIV), a newly developed comprehensive biomarker that combines lymphocyte, neutrophil, monocyte, and platelet counts, 7 has not witnessed extensive exploration of its potential applications since its initial conceptualization. PIV has been confirmed to be proficient in evaluating the risk and prognosis of multiple malignancies, like colorectal, breast, and laryngeal tumors. It is also advantageous in overseeing hypertension and serves as a manifestation of human immunological response and inflammation.8–11 It is widely acknowledged that inflammatory factors can activate remodeling cascades and induce pathological alterations during the progression of HF.12,13 However, the relationship between PIV and HF is currently unclear, as is the subsequent dose–response relationship.
Therefore, using a nationally representative dataset from the 2011–2020 National Health and Nutrition Examination Survey (NHANES), we conducted a cross-sectional analysis to elucidate the relationship between PIV and HF, and further assess the role of PIV in the occurrence of HF.
Methods
Study population and STROBE statement
During the period from 2011 to 2020, data were gathered from the NHANES, which is a nationwide cross-sectional study managed by the Centers for Disease Control and Prevention (CDC) with the aim of assessing the health and nutritional conditions of the American population. The study protocol was reviewed and approved by the Research Ethics Review Board of the National Research Ethics Committee. Written informed consent was obtained from all participants involved in this research, and they all followed the NHANES survey procedures, thus guaranteeing strict compliance with ethical standards. Detailed information about the study methodology and NHANES-related data can be retrieved from the official website of the CDC (https://www.cdc.gov/nchs/nhanes/). For the purposes of our study, we selectively extracted the obtainable data of those individuals who took part in the 2011–2020 cycles of the NHANES study. The initial sample consisted of 54,717 individuals. Following the exclusion of 22,868 individuals under the age of 20 years, 3112 with incomplete heart HF status and PIV data, as well as 17,233 participants with missing covariate data or who were pregnant, the final study cohort included 11,504 eligible participants (Figure 1). The reporting of this study conformed to STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. 14
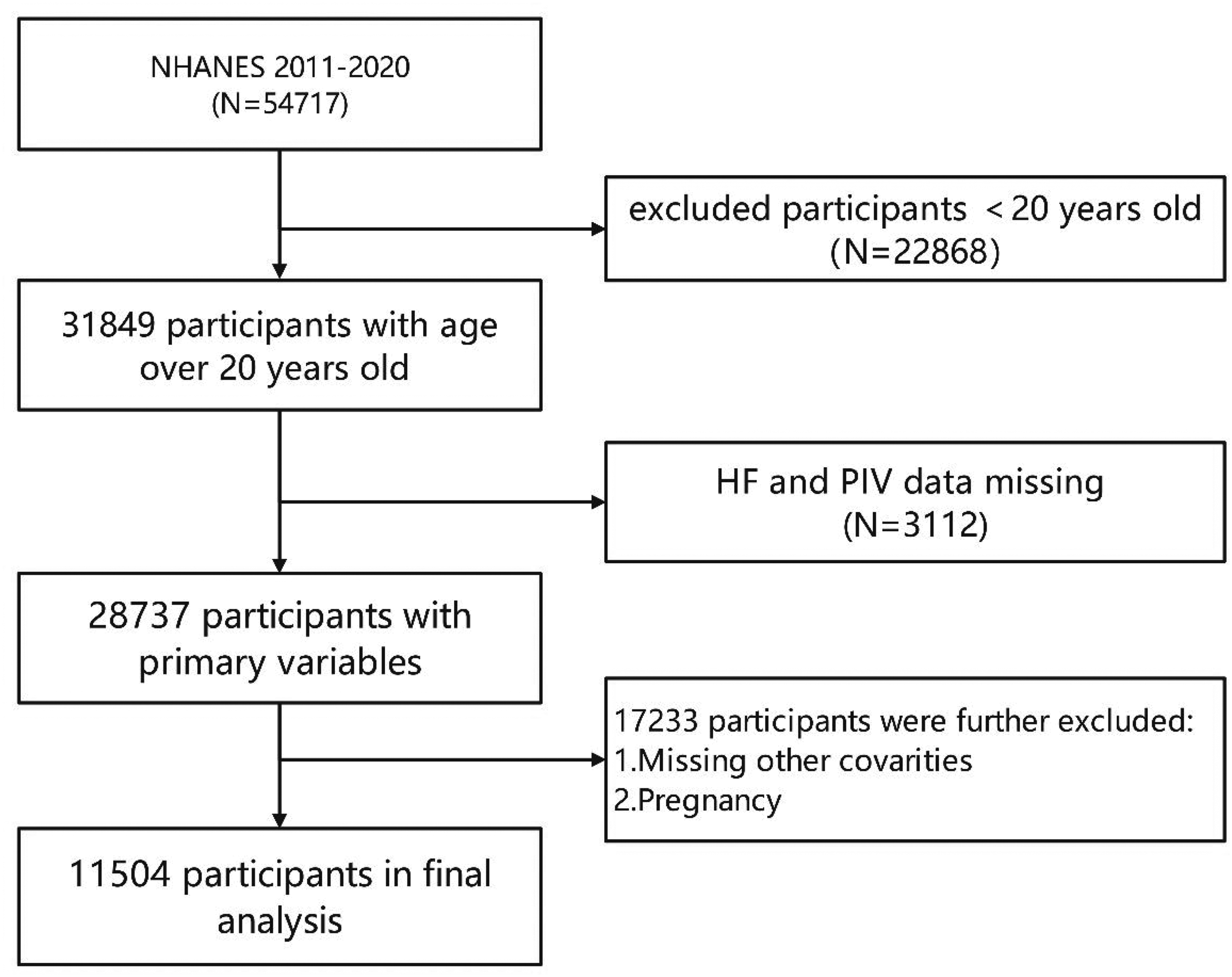
Flowchart diagram of patients’ selection in this study. HF: heart failure; NHANES, National Health and Nutrition Examination Survey; PIV: pan-immune-inflammation value.
Assessment of HF
HF data were gathered through personal interviews using a standardized questionnaire. Participants were deemed to have HF if they responded affirmatively to the query, “Has a doctor or healthcare specialist ever informed you that you have HF?” The interpretation of questionnaire data for diagnosing HF remains unclear, making it difficult to effectively identify the condition without access to clinical features, physical examination, diagnostic tests, and functional assessment. Previous articles have also adopted the response from this questionnaire as a method of defining HF.15–17
Calculation of the PIV
The Beckman Coulter DxH 800 apparatus, installed in the NHANES mobile examination center, is capable of conducting a comprehensive analysis of blood cellular constituents for each participant and generating complete blood cell count (CBC) reports for blood specimens. The PIV is derived from meticulously examined blood samples, with stringent standardized collection procedures being followed to ensure the accuracy and reliability of the collected data. The computation of the PIV primarily relies on the results of the CBC evaluation. Specifically, the counts of platelets, neutrophils, and lymphocytes are precisely measured at a density of 1000 cells per microliter. The PIV is then calculated accurately using the following formula: PIV = (platelet count × neutrophil count ×monocyte count)/lymphocyte count.
Covariates definition
To improve the reliability of our examination on the connection between PIV and HF, we deliberately selected a set of variables that could potentially interfere with the association. In terms of sociodemographic factors, our investigation covered the age range of the participants, their sex, racial diversity, body mass index (BMI) categories (<18.5, 18.5–24.9, 25–29.9, and ≥30 kg/m2), educational attainment (categorized as less than high school, high school graduate or higher), and a measure of household economic status, using the household poverty-to-income ratio (PIR) as an indicator. The specific PIR ranges are ≤1.35, 1.35–1.85, and >1.85. When it comes to laboratory examinations, we focused on several key biomarkers, including total cholesterol, high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), and triglyceride levels. The assessment of comorbidities was based on whether the participants had been diagnosed by a medical professional with conditions such as stroke, diabetes, hypertension, asthma, coronary heart disease, angina, or myocardial infarction. Regarding the definition of smoking status, we followed the convention that individuals were considered smokers if their lifetime cumulative cigarette consumption exceeded 100 cigarettes. For detailed procedures and standards for assessing all of these covariates, readers may refer to the NHANES guidelines (www.cdc.gov/nchs/nhanes/).
Statistical analysis
All statistical analyses were performed using R (version 4.1.2) and EmpowerStats (version 4.2). The criterion of significance difference was set at p < 0.05. Participants were divided into two groups based on their HF status. Continuous data are presented as mean ± SD, whereas categorical data are presented as percentages. The PIV data are unevenly distributed and their values were logarithmic (ln) transformed before conducting statistical analysis. The categorical variables were compared using chi-square test, whereas the association between continuous variables was analyzed through weighted linear regression model. To delve deeper into the association between PIV and HF, we constructed three multivariate logistic regression models: model 1 (a non-adjusted model), model 2 (a model adjusted only for gender, age, race, education, and PIR), and model 3 (a fully adjusted model that included all potential confounding factors). To explore the threshold effects of PIV and HF, we adopted a two-piecewise linear regression model with a smoothing curve fitting, and used the log-likelihood ratio test to verify the non-linear effect and identify the inflection point value. In addition, we performed a subgroup analysis, stratified by gender, race, age group, educational degrees, BMI, as well as the presence or absence of stroke, diabetes, hypertension, asthma, coronary heart disease, angina, heart attack, and smoking status, to examine in detail the differences and similarities in different settings. Subsequently, we performed interaction tests to assess whether there were significant differences in effects among these subgroups.
Results
Baseline characteristics
Table 1 summarizes the baseline characteristics of the participants. This study incorporated 11,504 participants, among whom 389 were diagnosed with HF (3.38%). The average age of participants was 49.99 ± 17.37 years and included 5598 (48.67%) males and 5906 (51.33%) females. There were significant differences existed in demographic profiles and baseline clinical characteristics among patients with HF and those without HF. Specifically, HF group exhibited a higher proportion of males, elderly individuals, non-Hispanics, smokers, and individuals with lower educational degrees and poverty income ratio. Similarly, the proportion of individuals diagnosed with diabetes, coronary artery disease, angina pectoris, asthma, hypertension, heart attack, and stroke were found to be significantly higher in the HF group than that in the non-HF group. Additionally, individuals with HF showed higher levels of BMI, triglyceride, and PIV levels, whereas they showed decreased concentrations of HDL, LDL, and total cholesterol.
Baseline characteristics of selected individuals according to HF status.

Mean ± SD for continuous variables: the p value was calculated by a linear regression model. % for categorical variables; the p value was calculated by a chi-square test.
BMI: body mass index; HDL: high-density lipoprotein; HF: heart failure; LDL: low-density lipoprotein; ln-PIV: logarithmic pan-immune-inflammation value; PIR: poverty-to-income ratio.
Association between PIV and HF
Table 2 shows the association between ln-PIV and HF, revealing that an increase in ln-PIV level is associated with an elevated likelihood of HF occurrence. In the crude model (model 1), ln-PIV is positively associated with HF (odds ratio [OR] = 2.06; 95% confidence interval [CI], 1.78–2.37, p < 0.0001). These significant associations were maintained in model 2 after it was adjusted for gender, age, race, education, BMI, and PIR. After adjusting for all covariates, each increment unit in ln-PIV is associated with 34% increase in the risk of HF occurrence (OR = 1.34; 95% CI, 1.14–1.58, p = 0.0004). Further stratifying ln-PIV into tertiles and employing multivariate logistic regression analysis, we found that individuals with the highest ln-PIV tertile exhibited a statistically significant 90% elevated risk of HF occurrence compared with those in the lowest tertile (OR = 1.90; 95% CI, 1.38–2.61, p < 0.0001) in a fully adjusted model.
Association between ln-PIV and HF.
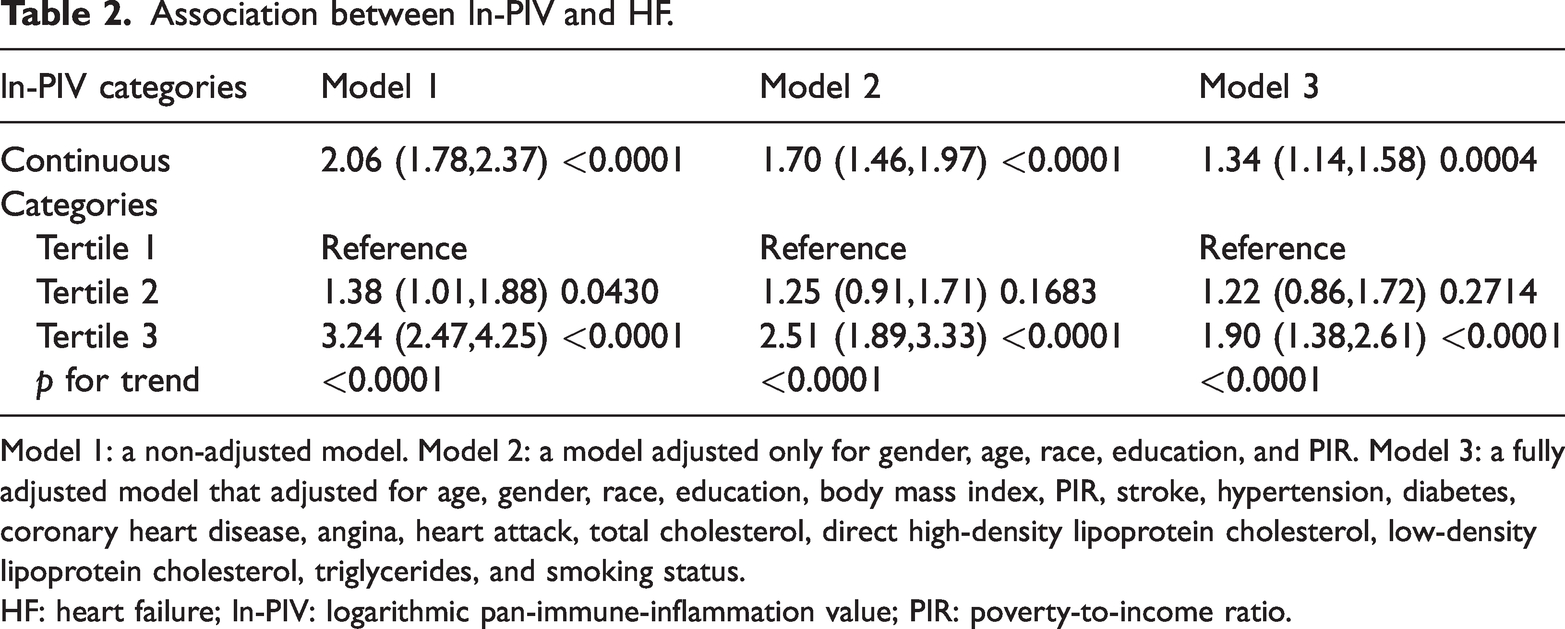
Model 1: a non-adjusted model. Model 2: a model adjusted only for gender, age, race, education, and PIR. Model 3: a fully adjusted model that adjusted for age, gender, race, education, body mass index, PIR, stroke, hypertension, diabetes, coronary heart disease, angina, heart attack, total cholesterol, direct high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, triglycerides, and smoking status.
HF: heart failure; ln-PIV: logarithmic pan-immune-inflammation value; PIR: poverty-to-income ratio.
Stratified analysis
Stratified analyses revealed that the correlation between PIV and HF occurrence remains consistent across different subgroups (Figure 2). This indicates that variables including gender, ethnicity, age, educational degrees, BMI categories, stroke, hypertension, diabetes, asthma, coronary artery disease, angina, heart attack, and smoking status did not exert a significant influence on this positive correlation (p > 0.05 for all subgroups).

Stratified analysis for the association between logarithmic pan-immune-inflammation value and heart failure. BMI: body mass index; CI: confidence interval; OR: odds ratio.
Non-linear relationship and threshold analysis
The analysis of a non-linear relationship and threshold effect were performed through the application of smooth curve fitting (Figure 3). We identified an inflection point of ln-PIV = 5.98 as a critical threshold and quantify the non-linear effects (Table 3). Below this threshold, each increment unit in ln-PIV was associated with a 66% increase in the risk of HF (OR = 1.66; 95% CI, 1.28–2.14, p < 0.0001). However, above the threshold, there was no significant association between ln-PIV and the risk of HF (OR = 0.89; 95% CI, 0.59–1.35, p = 0.5880). This suggests that PIV have a threshold effect on HF occurrence. After reaching a particular threshold, additional elevations in PIV may not introduce any further hazards.

Relationship between ln-PIV and heart failure by smooth curve fitting. CI: confidence interval; ln-PIV: logarithmic pan-immune-inflammation value; OR: odds ratio.
Threshold effect for the relationship between ln-PIV and HF.

CI: confidence interval; HF: heart failure; ln-PIV: logarithmic pan-immune-inflammation value.
Discussion
The relationship between HF and inflammation is intricate, and involves interactions between heart and multiple peripheral organs. Inflammatory chemokines and cytokines contribute to persistent low-grade inflammation and impose detrimental effects on organic structures beyond the heart. 18 Notably, extensive research has established the involvement of the proinflammatory cytokines, particularly TNF-α, IL-1, and IL-6, as essential components in the intricate mechanisms underlying the pathogenesis of HF. 19 Additionally, inflammatory responders including neutrophils, monocytes, lymphocytes, and platelets are also activated and recruited during the development of HF. And novel inflammatory scores of monocyte-to-lymphocyte ratio, neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, and systemic immune-inflammation index have been investigated and shown promising potential in predicting HF and subsequent all-cause mortality. Thus, as an indicator integrates the four major immune cells, PIV has received great attention ever since it was first proposed.
The current study is the first investigation designed to explore the association between PIV and the risk of HF, using a comprehensive analysis of epidemiological data from US adults. PIV, which serves as a promising cancer biomarker, has attracted significant interest due to its prognostic potential in various malignancies, including colorectal and breast cancer. Epidemiological evidence suggests that inflammation plays a pivotal role in the risk and progression of several chronic diseases. It is well established that heightened inflammatory responses can exacerbate cancer development by promoting tumor growth and metastasis. As such, PIV acts as a biomarker that encapsulates not only the physiological state of inflammation but also the immune status of individuals, thus bridging a gap between cancer biology and cardiovascular health. Recent research indicates that PIV predicts enhanced chemotherapy responses in Turkish patients with breast cancer, highlighting its potential utility in personalizing treatment strategies. 20 Similarly, studies conducted by Sahin et al. 21 have established that lower PIV levels correlate with improved survival outcomes among HER2-positive patients undergoing trastuzumab therapy. The ability of PIV to provide valuable prognostic insights is underscored by findings from the Valentino (Phase II Randomized Study of Panitumumab Plus FOLFIRI Followed by Panitumumab Plus Fluorouracil/Leucovorin as Maintenance Therapy in RAS Wild-Type Metastatic Colorectal Cancer) and TRIBE (Phase III Randomized Trial of FOLFOXIRI Plus Bevacizumab vs. FOLFIRI Plus Bevacizumab as First-Line Treatment for Metastatic Colorectal Cancer) trials, which reaffirmed its significance in predicting outcomes for patients with metastatic colorectal cancer receiving first-line treatments. 7 The relevance of PIV extends beyond oncology into the realm of cardiovascular diseases. Elevated PIV levels have been documented in individuals experiencing the coronary slow flow phenomenon, a condition associated with impaired coronary microcirculation. 22 This association suggests that systemic inflammation, as indicated by high PIV levels, may contribute to coronary artery pathophysiology, potentially leading to adverse cardiac events. Furthermore, a high PIV level has been identified as an independent predictor of ischemic complications following percutaneous coronary intervention in patients with ST-segment elevation myocardial infarction, highlighting its potential role in risk stratification for cardiac patients. 23 Moreover, studies have shown that elevated PIV is related to increased mortality risks among hypertensive patients, emphasizing its role as an important biomarker for assessing cardiovascular risk in this population. 11 Recent research has found that elevated levels of PIV are significantly associated with poor long-term outcomes in patients with acute decompensated HF, including higher mortality and readmission rates.24,25 Our research further demonstrated that PIV may be considered an independent risk factor for HF. This finding would be even more accurate if more confounders were taken into account.
The precise mechanisms underlying novel immune markers in the development and progression of HF remain incompletely understood. However, some related research in this field has garnered recognition. Leukocytes contribute to both the initiation and perpetuation of an inflammatory cascade that can exacerbate cardiac damage, further compromising cardiac output and leading to a vicious cycle of decline. 26 The inflammatory response in HF is triggered by a myriad of factors, including myocardial injury, ischemia–reperfusion injury, and neurohormonal activation. These stimuli activate resident cardiac immune cells, such as macrophages and dendritic cells, which release a barrage of cytokines and chemokines that recruit additional leukocytes to the heart. 27 Neutrophils, the first responders, rapidly infiltrate the myocardium, where they engage in phagocytosis of damaged tissue and pathogens, releasing reactive oxygen species and proteases that can exacerbate cardiac injury. 28 As the inflammatory response progresses, monocytes migrate to the heart and differentiate into macrophages, which further amplify the inflammatory cascade by secreting cytokines and chemokines that recruit additional immune cells. These macrophages can also polarize into distinct phenotypes, with M1 macrophages promoting inflammation and tissue damage, whereas M2 macrophages participating in tissue repair and resolution of inflammation. 29 Lymphocytes, particularly T cells, also play a significant role in the inflammatory response in HF. Activated T cells can infiltrate the myocardium, recognizing antigens presented by antigen-presenting cells and mediating both adaptive immune responses and direct cytotoxicity toward cardiac cells. 30 Moreover, the production of autoantibodies against cardiac proteins by B cells has been implicated in certain forms of HF, further exacerbating the inflammatory process. 31 Platelets, traditionally recognized for their hemostatic function, have emerged as dynamic players in the inflammatory cascade that characterizes HF. 32 Upon activation by various stimuli, including endothelial injury, oxidative stress, and neurohormonal dysregulation, platelets release a myriad of cytokines, chemokines, and growth factors that serve as potent signals for the recruitment and activation of leukocytes within the cardiac tissue. 33 Furthermore, the inflammatory milieu triggers the activation of the neurohormonal cascades, notably the renin-angiotensin-aldosterone axis and the sympathetic nervous system, which further perpetuate cardiac dysfunction and inflammation. 34 We hypothesize that the association between inflammatory markers and HF deserves further investigation, and our findings may yield broader public implications within the field of HF.
Our research has certain strengths and constraints. Initially, the study was based on a sample of adults from the United States that was ethnically diverse and nationally representative. This enabled us to conduct subgroup analyses, thereby minimizing residual confounding. Additionally, our study uncovered a dose–response relationship by carrying out non-linear and threshold effect analyses. However, this study also had some constraints. Firstly, there were limited information about participants’ clinical features, physical examination, diagnostic tests, and functional assessment, and the diagnosis of HF was obtained through self-reported medical questionnaires completed during personal interviews. Second, due to the limitations of the database, we were precluded from incorporating other potential covariates that might potentially impact the association between the PIV and HF. Third, the inherent nature of the cross-sectional study design restricts the conclusions drawn to an association rather than establishing a causal relationship. Consequently, future longitudinal studies are crucial to reveal the underlying causal relationship and to evaluate the effectiveness of anti-inflammatory treatment in HF.
Conclusion
Therefore, our results demonstrated that PIV, as a non-invasive and easily measurable hematological indicator, may serve as a valuable biomarker in the assessment and monitoring of HF. However, further research is warranted to clarify the precise mechanisms underlying this association and to explore its potential clinical applications in risk stratification, early detection, and management of HF.
Abbreviations
body mass index cholesterol complete blood cell count Centers for Disease Control and Prevention confidence interval high-density lipoprotein heart failure low-density lipoprotein National Health and Nutrition Examination Survey poverty-to-income ratio pan-immune-inflammation value odds ratio Strengthening the Reporting of Observational Studies in Epidemiology
Footnotes
Acknowledgements
The author expresses gratitude to all NHANES participants for their valuable contributions to this study.
Author contributions
Data availability statement
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors have no funding and conflicts of interest to disclose.
