Abstract
Limb-shaking transient ischemic attack (LS-TIA) is a rare clinical manifestation of steno-occlusive carotid lesions. Reports of LS-TIA caused by posterior circulation ischemia are rare. Here, the case of a 56-year-old male patient with a history of epilepsy, who presented with dizziness, nausea and vomiting, is reported. Initial brain magnetic resonance imaging (MRI) indicated no signs of acute ischemia, and the patient’s symptoms had not relieved despite drug administration to treat dizziness. During a vestibular function test on the next day, he developed sudden left-limb shaking without disturbance of consciousness or facial twitching. Considering a seizure, an anti-epileptic drug was administered, but no improvement was observed. Eight hours later, the patient experienced left-limb paralysis, and re-examination of the MRI indicated brainstem infarction. The patient recovered well with antiplatelet and antiatherosclerotic therapy. While LS-TIA is a rare manifestation of TIA of the carotid arterial system, posterior circulation ischemia may also manifest as LS-TIA. If a patient presents with limb shaking due to postural changes despite a history of epilepsy, LS-TIA should be highly suspected.
Keywords
Introduction
Limb-shaking transient ischemic attack (LS-TIA) is a clinically rare type of TIA that is similar to focal motor seizures. Often misdiagnosed, LS-TIA might progress to severe cerebral infarction if not treated in time. Limb shaking might be a red-flag for critical haemodynamic compromise, and early recognition may lead to timely intervention and reduced future risk of stroke.1,2 Here, the case of a patient with brainstem infarction, who had a history of epilepsy and experienced limb shaking due to posture changes, is described. Although the limb shaking was mistaken for a seizure after onset, the cause was subsequently diagnosed and the patient recovered well with antiplatelet and antiatherosclerotic therapy.
Case report
A 56-year-old male patient presented to the Emergency Department of Tianjin Huanhu Hospital, Tianjin, China in July 2021, with dizziness, nausea, vomiting and unsteady gait for the previous 6.5 h. The patient had histories of hypertension, cerebral infarction, and basilar artery stent implantation. He had been diagnosed with epilepsy for more than 3 years, which manifested as automatisms, including involuntary actions of putting-on and taking-off clothes, walking around and talking to himself, accompanied by impaired consciousness. The epilepsy had been treated by 450 mg oxcarbazepine, orally, twice daily, and seizures had not recurred during the previous 6 months (approximately). On presentation, the patient’s blood pressure was 120/85 mmHg. Neurological examination revealed no abnormal findings. Brain magnetic resonance imaging (MRI) indicated no signs of acute ischemia but revealed chronic lacunar infraction in the bilateral cerebellar hemisphere and encephalomalacia in the right temporo-occipital region (Figure 1 and 2). Magnetic resonance angiography indicated occlusion of the right vertebral artery (Figure 3 and 4). The patient’s symptoms had not relieved with administration of 12 mg betahistine and 100 mg gastrodin capsule, orally, three times daily, to treat dizziness. He was referred to the otorhinolaryngology outpatient department the next day, because the dizziness was related to postural changes.
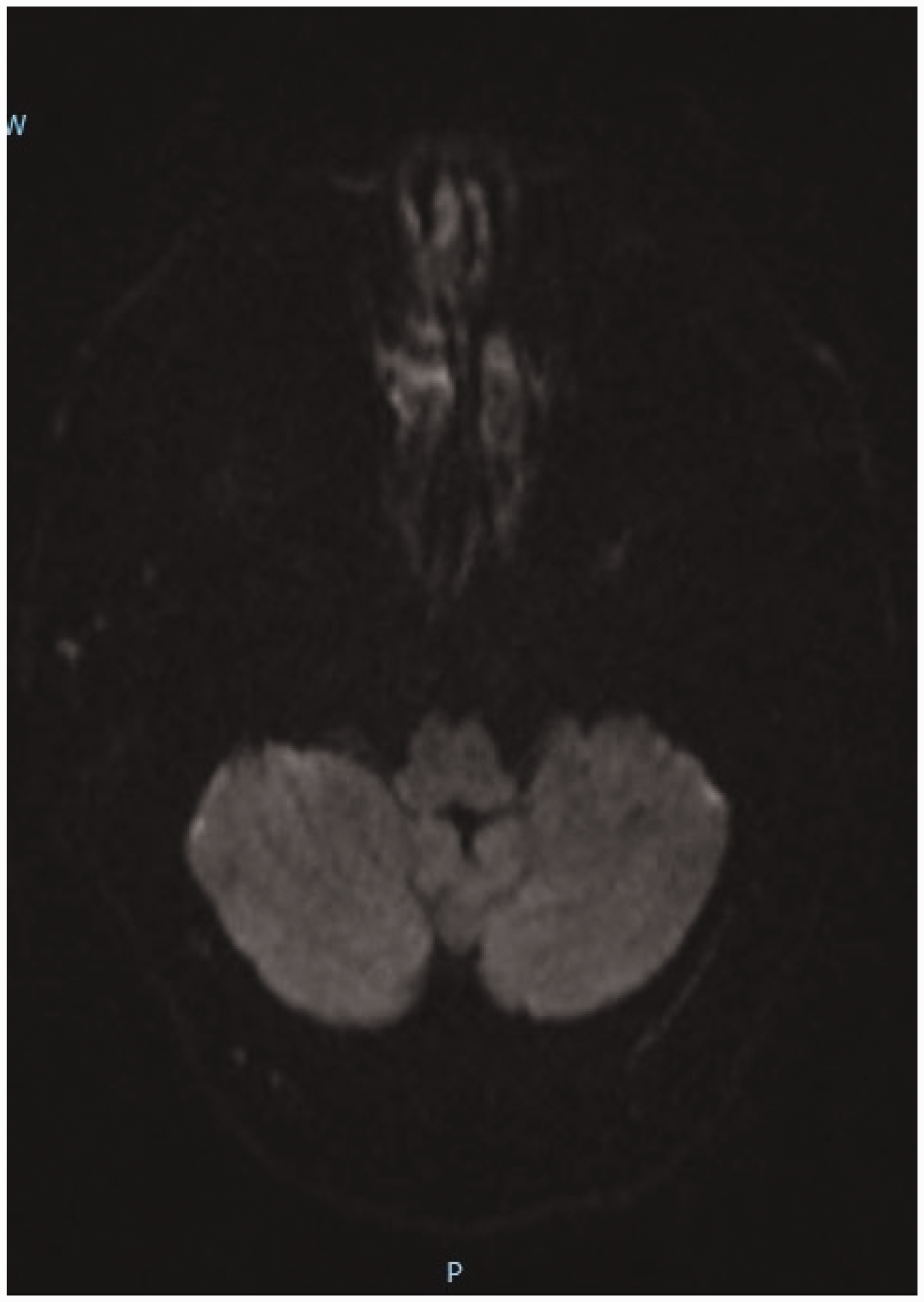
Initial brain magnetic resonance imaging (MRI) in a 56-year-old male patient with a 3-year history of epilepsy who presented with dizziness, nausea and vomiting. No signs of acute ischemia were revealed, but encephalomalacia was observed in the right temporo-occipital region. Axial head diffusion-weighted MRI.

Initial brain magnetic resonance imaging (MRI) in a 56-year-old male patient with a 3-year history of epilepsy who presented with dizziness, nausea and vomiting. No signs of acute ischemia were revealed, but encephalomalacia was observed in the right temporo-occipital region. Axial head fluid-attenuated inversion recovery (flair) MRI.

Magnetic resonance angiography (MRA) in a 56-year-old male patient with a 3-year history of epilepsy who presented with dizziness, nausea and vomiting: MRA indicated occlusion of the right vertebral artery. Three-dimensional time-of-flight MRA.

Magnetic resonance angiography (MRA) in a 56-year-old male patient with a 3-year history of epilepsy who presented with dizziness, nausea and vomiting: MRA indicated occlusion of the right vertebral artery. Three-dimensional time-of-flight MRA.
During vestibular function tests, the patient developed sudden left-limb shaking without limb weakness, tonic-clonic jerks, disturbance of consciousness or facial convulsions. The patient was immediately transferred back to the emergency department. His blood pressure was 110/70 mmHg, and no abnormality was found in blood routine examination, renal and liver function, or electrolyte and glucose levels. He had not taken any dose of his antiepileptic drug (AED) after the onset of symptoms approximately 6.5 h before presentation to hospital. Considering a seizure, 200 mg phenobarbital, intramuscular injection, and 400 mg sodium valproate, intravenous drip, were successively administered, but the shaking did not improve and lasted for around 4 h. The patient then fell asleep without limb shaking. After 4 h, he woke up and discovered that his left limb was weak and difficult to lift, but there was no shaking. Neurological examination revealed left-limb paralysis. The left upper limb could only move horizontally, the left lower limb could be slightly lifted against gravity but not resistance, and left Babinski sign was positive. He presented with no cranial nerve deficit or other positive signs. Re-examination of the brain MRI indicated acute cerebral infarction of the right medulla oblongata (Figure 5). The patient was immediately administered 200 mg aspirin and 20 mg atorvastatin, orally, once daily, and fluid replacement therapy. He was then hospitalized, followed by initiation of anticoagulation therapy with 10 mg argatroban, intravenous drip, twice daily, in addition to the medications mentioned above. His blood pressure was maintained at a higher level (systolic blood pressure: 140–150 mmHg and diastolic blood pressure: 90–105 mmHg) with avoidance of all antihypertensive medications. An electroencephalogram (EEG) showed no abnormal findings, and 450 mg oxcarbazepine, orally, was continued twice daily. Computed tomography angiography indicated right vertebral artery occlusion, basilar artery stent implantation and mild left vertebral artery stenosis (Figure 6 –8). The patient’s limb weakness improved after antiplatelet and antiatherosclerotic therapy. The limb shaking did not recur and he benefitted from avoiding hypotension and lying down. During 8 months of follow-up, the patient could lift and hold objects using the left upper limb and walk independently, and the limb shaking did not recur. The patient provided verbal informed consent to treatment. All patient details were de-identified for this case report and the reporting of this study conforms to CARE guidelines. 3

Re-examination of brain magnetic resonance images in a 56-year-old male patient with a 3-year history of epilepsy who presented with dizziness, nausea and vomiting, indicated acute cerebral infarction of the right medulla oblongata.
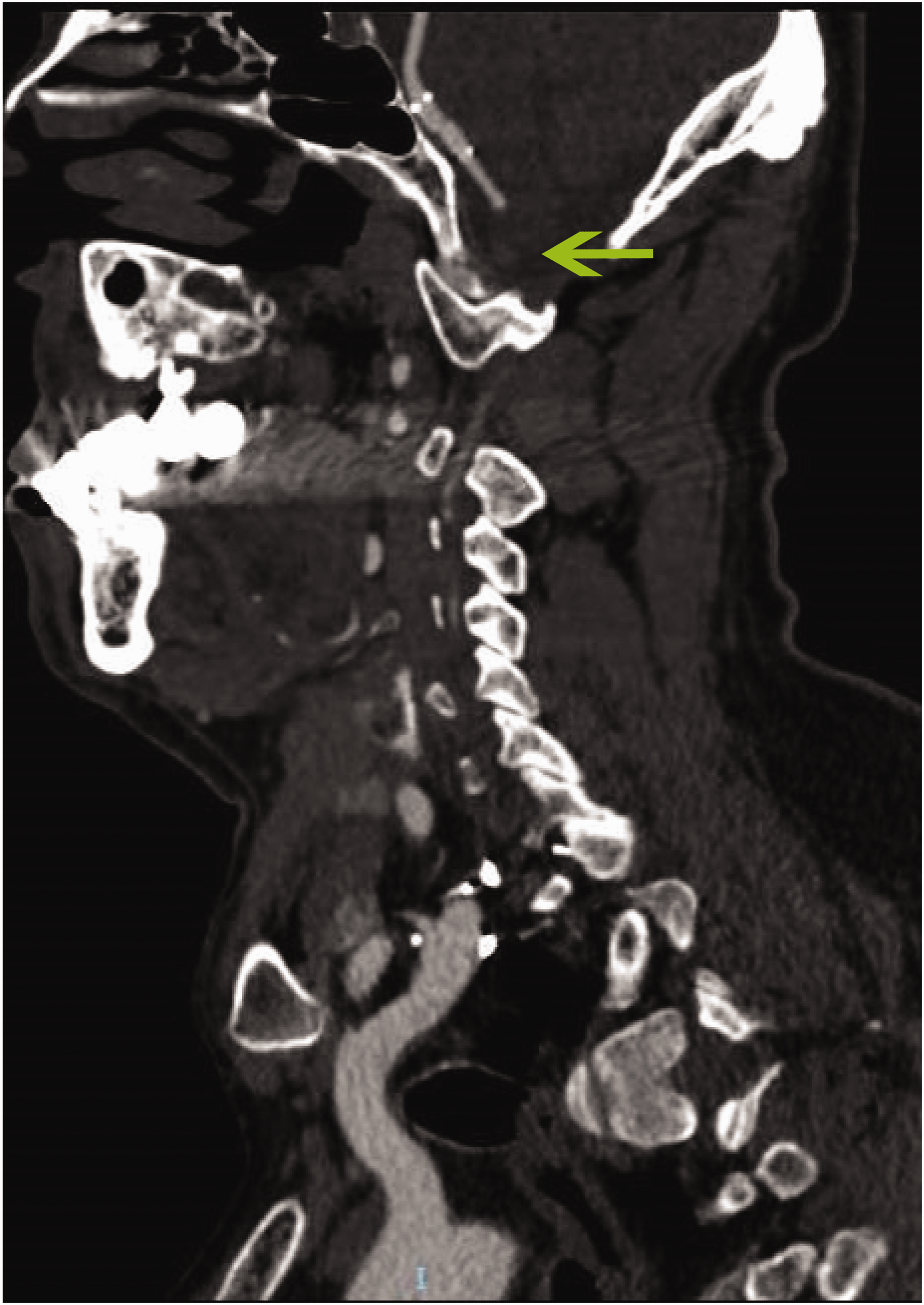
Computed tomography angiography (CTA) in a 56-year-old male patient with a 3-year history of epilepsy who presented with dizziness, nausea and vomiting. CTA indicated right vertebral artery occlusion and basilar artery stent implantation. Sagittal plane view around the right vertebral artery.
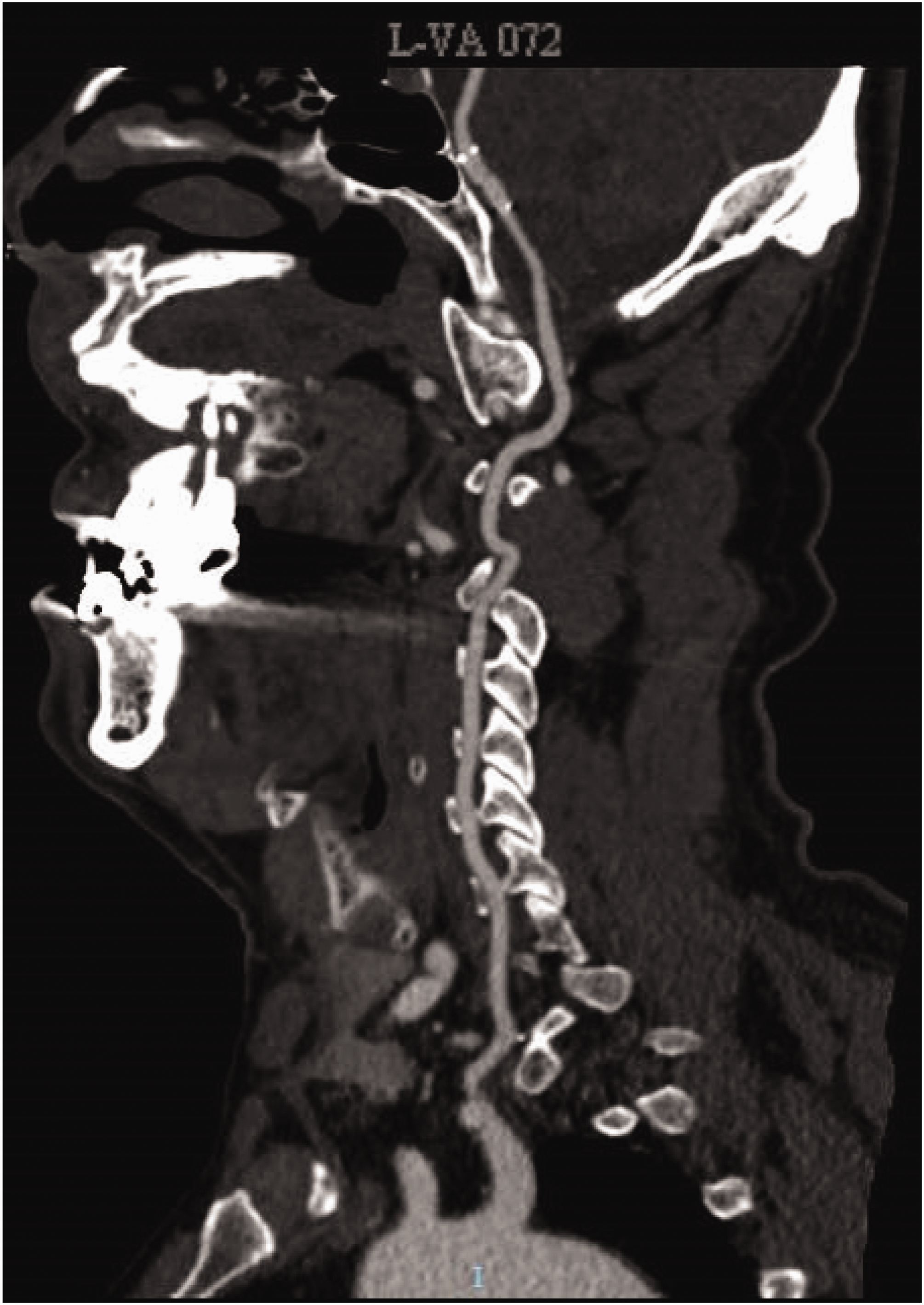
Computed tomography angiography (CTA) in a 56-year-old male patient with a 3-year history of epilepsy who presented with dizziness, nausea and vomiting. CTA indicated right vertebral artery occlusion and basilar artery stent implantation. Sagittal plane view around the left vertebral artery.

Computed tomography angiography (CTA) in a 56-year-old male patient with a 3-year history of epilepsy who presented with dizziness, nausea and vomiting. CTA indicated right vertebral artery occlusion and basilar artery stent implantation. Three-dimensional CTA image.
Discussion
Since the first report of transient limb-shaking syndrome associated with contralateral carotid stenosis by Fisher in 1962, 4 scholars have increasingly focused on this exceptional TIA, which is believed to be a rare manifestation of TIA caused by steno-occlusive carotid lesions. 5 The present study validated that posterior circulation ischemia may also manifest as LS-TIA, which has been rarely reported previously. Limb shaking is characterized by transient, repetitive, involuntary shaking of one or both limbs and a relative sparing of the face. LS-TIA is often mistaken for focal seizure; however, LS-TIA shows no Jacksonian march and no involvement of facial muscles or trunk, and no head or eye deviation, incontinence, or tongue bite. LS-TIA is often associated with postural change, whereas motor seizures are not. Involuntary shaking of the limb occurs without disturbance of consciousness and tonic-clonic jerks. Epileptic discharges are not shown in EEG recording during episodes and interludes.6,7 The shaking is not controlled by AEDs,6,7 but may be controlled with antiplatelet drugs. In the present case, the patient’s shaking lasted for several hours and was not controlled by an AED, and epileptic discharges were not found on surface EEG without deep electrodes. Therefore, there should be no doubt about the diagnosis of LS-TIA. However, it is worthwhile completing a thorough examination, particularly deep electrode examination, to rule out epilepsy as the cause.
Episodes of LS-TIA usually last for several seconds or minutes, while shaking may also last for several hours. 8 In the present case, the patient’s limb shaking lasted for 4 hours before sleep. The patient presented with continuous shaking of the left limb, which was not controlled after AED, and was found to have occlusion of the contralateral vertebral artery. Limb-shaking TIA is thought to be associated with high-grade stenosis or occlusion of the internal carotid artery (ICA), but in rare cases, posterior circulation ischemia may also manifest as limb shaking, and the compensatory function of posterior circulation ischemia may be worse. The present study reports only one case and further studies with larger sample sizes are warranted in this regard.
In the present case, the patient had a history of epilepsy and missed taking the AED after symptom onset, so could easily have been misdiagnosed as having a seizure. However, the limb shaking did not improve after administration of the AED, and the patient progressed to having a severe cerebral infarction. Of note, the patient had experienced automatism without limb shaking previously. On the present occasion, he presented with limb shaking purely due to postural changes, without disturbance of consciousness or other symptoms. Therefore, an early recognition may lead to timely intervention and reduced future risk of stroke.
Although the patient’s initial brain MRI indicated no signs of acute ischemia after symptom onset, magnetic resonance angiography indicated occlusion of the right vertebral artery, which was most likely a sign of posterior circulation ischemia. The patient developed sudden left-limb shaking due to postural changes and experienced left-limb paralysis later. Re-examination of the brain MRI indicated acute cerebral infarction of the right medulla oblongata that was due to occlusion of the right vertebral artery. Therefore, it is important to start an adequate diagnostic workup for LS-TIA, including imaging studies of the brain, in the emergency department.
Hypoperfusion is recognized as the generally accepted pathogenesis of LS-TIA. Initially, the brain is able to compensate for hypoperfusion by anastomoses of the Willis’ circle and dilation of the surrounding vessels through an autoregulation mechanism. The maximum vasodilatation of intercranial arteries causes the exhaustion of vasomotor reactivity that fails to maintain cerebral perfusion and outstrips an inherent cerebral autoregulatory reserve, and induces transient ischemia in certain situations. The cerebral vasomotor reserve capacity is decreased because of long-term chronic stenosis or occlusion of the intracranial or extracranial artery.9,10 Shaking is often triggered by postural changes, prolonged standing and walking, hyperextension of the neck and excessive lowering of the blood pressure, which can aggravate brain diminished perfusion of the frontal subcortical motor pathways. The physiological mechanism of LS-TIA in steno-occlusive carotid lesions is likely to be cortical ischemia and hypoxia that lead to subcortical disinhibition and neuronal hyperexcitability and excessive limb movements, which is similar to the mechanism of bilateral spastic twitching in patients with syncope. To date, LS-TIA has not been observed in patients with a normal internal carotid artery system, yet shaking occurred in the present patient with overt posterior circulation ischemia. The patient had occlusion of the right vertebral artery and right brainstem infarction after limb shaking, without stenosis or occlusion of the intracranial or extracranial artery. This indicates hypoperfusion in the posterior circulation and the physiological mechanism may be different from carotid lesions. A small sample study showed that patients with moyamoya angiopathy complicated by posterior cerebral artery (PCA) stenosis were more likely to develop LS-TIA due to diminished maintenance of the anterior circulation by leptomeningeal collaterals from the PCA. 11 However, the physiological mechanism of limb shaking in brainstem infarction remains unclear. It may be that spinal myoclonic jerks, which are similar to shakings, are induced by ischemia of the cervical spinal cord due to occlusion of the right vertebral artery. The present report contributes to a growing body of evidence that hypoperfusion-related cerebral ischemia is not solely confined to carotid disease, but may include posterior circulation ischemia. Further studies are needed to investigate more patients with LS-TIA involving posterior circulation ischemia, to fully elucidate the pathophysiological mechanism.
The management principle of LS-TIA is to increase cerebral blood flow, improve hypoperfusion, and minimize stroke risk. Treatment of limb-shaking TIAs include conservative drug-based therapy and surgical revascularization. 12 The conservative drugs that reduce embolic risk and prevent atherosclerotic disease progression, such as antiplatelet and hypolipidemic drugs, were effective in the present case. Surgical revascularization is also effective in abolishing the attacks, and revascularization of the responsible artery may work better than conservative drug-based therapy.13,14 Carotid endarterectomy is the treatment of choice to reduce stroke risk if LS-TIA is complicated by serious extracranial ICA stenosis in patients under 70 years old. 15 Extracranial-intracranial bypass surgery has not been shown to reduce long-term stroke risk associated with TIAs in patients with complete ICA occlusion. However, the present patient did not receive surgical revascularization and recovered well with antiplatelet and antiatherosclerotic therapy; there was no recurrence of limb shaking during follow-up. Thus, treatment should be individualized by detailed arterial imaging, or depending on the location of the cerebrovascular pathology.
No difference in mortality rate has been shown between patients on TIA prophylactic medications and those not on TIA medications. There was no significant difference in treatment outcomes in patients who experienced TIA who were treated in the emergency department and discharged home versus those admitted to hospital for additional treatment. 16 Therefore, patients with symptoms of TIA need not be admitted to hospital, except when such patient symptoms fail to resolve in the emergency room.
In conclusion, LS-TIA is a special manifestation of steno-occlusive carotid lesions, although posterior circulation ischemia may also manifest as limb shaking. In addition, patients with limb shaking due to postural changes should be suspected of LS-TIA, and MRI and EEG should be performed carefully to avoid misdiagnosis.
Research Data
Research Data for Limb-shaking transient ischemic attack in posterior circulation ischemia: a case report
Research Data for Limb-shaking transient ischemic attack in posterior circulation ischemia: a case report by Zhihong Zhao, Jie Qin in Journal of International Medical Research
Footnotes
Author contributions
Zhihong Zhao collected patient information, drafted the manuscript and collected the figures; Jie Qin edited the manuscript. Both authors read and approved the final manuscript.
Date accessibility statement
Data are available on request from the authors.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
