Abstract
Objective
Observational studies suggest a link between hernia and inflammatory cytokines, but randomized trials are limited by ethical and cost constraints. In this study, we used bidirectional Mendelian randomization (MR) to investigate the causal relationship between inflammatory cytokines and five types of hernia, aiming to inform preventive and therapeutic strategies.
Methods
We selected 41 inflammatory factors and five types of hernia as instrumental variables, using data from the IEU Open GWAS database including individuals of European descent. The primary analysis used the inverse variance weighted method with false discovery rate (FDR) adjustment. Additional MR methods and sensitivity analyses ensured robustness. Reverse MR was used to assess potential reverse causality.
Results
After FDR adjustment, stem cell growth factor beta (SCGFb) was causally associated with diaphragmatic hernia (odds ratio = 0.884, 95% confidence interval: 0.819–0.955). Reverse MR indicated that diaphragmatic hernia may increase interferon gamma-induced protein 10 (IP10) and monokine induced by interferon-gamma (MIG), and ventral hernia may elevate macrophage inflammatory protein-1b (MIP1b). Sensitivity analyses confirmed robustness.
Conclusion
SCGFb may protect against diaphragmatic hernia, and IP10, MIG, and MIP1b are involved in hernia development, suggesting the therapeutic potential of targeting these cytokines. Further studies are needed.
Keywords
Introduction
Hernia is a common surgical condition where an internal organ or tissue protrudes through a weak area of muscle or connective tissue. The International Classification of Diseases Tenth Revision (ICD-10) categorizes hernia into various types, primarily inguinal, femoral, umbilical, ventral, and diaphragmatic hernia. 1 These defects necessitate surgical intervention owing to the risk of complications such as incarceration and strangulation. 2 Although the use of prosthetic mesh in tension-free repair substantially reduces recurrence rates compared with suturing alone, it is less effective in minimizing postoperative complications. 3 Additionally, emergency hernia surgery is associated with higher mortality, increased patient morbidity, and extended hospital stays, adding a considerable burden to health care systems. 4
Despite numerous epidemiological studies investigating the roles of age, sex, smoking, obesity, and comorbidities in hernia development, the precise molecular mechanisms remain unclear.5,6 Inflammation, a crucial immune defense mechanism, 7 has been increasingly linked to hernia formation, progression, and surgical healing.8–10 Research indicates that inflammatory cytokines and pathways may play a significant role in hernia pathogenesis. For instance, excessive high mobility group box-1 (HMGB-1) in incisional hernias may lead to an exaggerated inflammatory response and activation of pathogenic fibroblasts, resulting in extracellular matrix disorganization and abdominal wall weakness.11–13 Additionally, elevated levels of cytokines such as epidermal growth factor, interferon (IFN)-α, interleukin (IL)-6, IL-8, and tumor necrosis factor (TNF)-α have been observed in patients with congenital diaphragmatic hernia (CDH), highlighting the role of inflammation in hernia development.14,15 However, another study demonstrated decreased IL-8 levels in patients with CDH. 16 This apparent inconsistency underscores the complexity of inflammation’s role and the need for causal inference tools to clarify these associations. Variability in epidemiological study results owing to differing methods and the presence of confounders complicates determination of whether inflammatory cytokines are a cause or consequence of hernia. 17 Ethical constraints and the high cost of randomized controlled trials further limit the ability to explore these associations. 18
Mendelian randomization (MR), in which genetic variations are leveraged to explore causal relationships, offers a robust method to address these challenges. 19 Although recent MR studies have linked obesity and smoking with hernia risk,20–22 a comprehensive analysis of the causal relationship between inflammatory cytokines and hernia risk has yet to be conducted. Using bidirectional MR, our study not only fills a critical gap in understanding the inflammatory underpinnings of hernia pathogenesis but also paves the way for developing targeted therapeutic strategies and preventive measures for hernia.
Methods
Study design
Figure 1 briefly depicts the overall study design of this two-sample bidirectional MR analysis. In this study, we used data from genome-wide association studies (GWAS) on 41 circulating inflammatory cytokines and five types of hernia to explore causal relationships. Comprehensive details about these datasets can be found in Tables S1 and S2. The overall process adheres to the three core assumptions of MR, namely, (1) relevance assumption: the genetic instruments are strongly associated with the exposure factor; (2) independence assumption: the genetic instruments are independent of confounders in the exposure–outcome relationship; (3) exclusivity assumption: the genetic instruments affect the outcome only through the exposure.23–25

Overview of study design and three core assumptions in Mendelian randomization.
Data source
The genetic predictors for the 41 circulating inflammatory cytokines in this MR investigation were derived from an extensive cytokine-related GWAS meta-analysis. The Cardiovascular Risk in Young Finns Study and two waves of the FINRISK investigations (FINRISK1997 and FINRISK2002), which included data for 8293 participants, were included in that meta-analysis. 26 That study investigated the genetic determinants of inflammatory cytokine levels using a robust two-step inverse adjustment to standardize the spectrum of 41 cytokine markers. To guarantee solid and trustworthy results, primary factors including age, sex, and the 10 leading genetic principal components were changed during the study. The data on hernia were derived from FinnGen Biobank, a sizable collaboration that combines genetic and health registry data from approximately 500,000 Finnish biobank participants. 27 According to ICD-10 classification, this dataset includes detailed information on 17,096 cases of inguinal hernia, 700 cases of femoral hernia, 4224 cases of umbilical hernia, 3737 cases of ventral hernia, and 5741 cases of diaphragmatic hernia, alongside 190,557 controls of European ancestry. No overlap was observed as the data for circulating inflammatory cytokines and hernia specimens were collected from different consortiums.
Instrumental variable selection
We used a rigorous screening procedure to ensure that our instrumental variables were valid and robust. First, rather than using the stricter p < 5 × 10−8 criteria, we applied a genome-wide significance threshold of p < 5 × 10−6 to collect enough single nucleotide polymorphisms (SNPs) for the subsequent MR analysis. Second, to guarantee independence of the chosen SNPs and avoid linkage disequilibrium, we applied the clumping algorithm with a threshold of r2 < 0.001 and a clumping window size of 10,000 kb. Third, SNPs with incompatible alleles and palindromic SNPs were excluded to prevent errors related to strand orientation or allele coding inconsistencies. Fourth, we implemented the MR Pleiotropy RESidual Sum and Outlier (MR-PRESSO) method to find and eliminate outlier SNPs that could introduce bias owing to horizontal pleiotropy.
28
Finally, we evaluated the strength of each remaining SNP as an instrumental variable by calculating the F-statistic using the formula
Mendelian randomization (MR) analyses
In our MR analysis, we primarily used the inverse variance weighted (IVW) method, which combines Wald estimates for each genetic variant to assess the causal effect of genetically predicted exposure on the outcome. This method provides a robust causal analysis. 31 To ensure consistency and reliability, we complemented IVW with three additional methods: MR-Egger, weighted median, and MR-PRESSO. Results were considered meaningful if the IVW method yielded a statistically significant p-value and the β-values from the other methods were consistent in direction. For binary outcomes, we reported odds ratios (ORs), and for continuous outcomes, β-values. This distinction allowed us to accurately quantify effect sizes. To account for multiple testing, we applied the Benjamini–Hochberg false discovery rate (FDR) correction, considering FDR-adjusted p-values below 10% to be statistically significant. 32
Sensitivity analysis
To ensure robustness of the MR findings, we used a comprehensive suite of sensitivity analyses. These included the Cochran Q test, MR-Egger intercept, MR-PRESSO test, and leave-one-out analyses. The Cochran Q test was used to assess heterogeneity among the individual SNP estimates, with a significant p-value (<0.05) indicating the presence of heterogeneity. The MR-Egger intercept test was conducted to detect horizontal pleiotropy. 33 The MR-PRESSO test was used to further evaluate horizontal pleiotropy and identify potential pleiotropic outliers, providing adjusted estimates after excluding such outliers. 28 Leave-one-out analyses was applied to assess the influence of individual SNPs on the overall results. All analyses were executed using R software version 4.3.2 (www.r-project.org) with the R packages “Two-sampleMR” and “MR-PRESSO,” ensuring a comprehensive and robust exploration of the causal relationships in our study.
Neither ethics approval nor informed consent were required owing to the nature of this MR study using data only.
Results
When screened at a significance threshold of p < 5 × 10−6, the F-statistic values for the 41 inflammatory cytokine SNPs ranged from 11.16 to 789.15, and the F values for the hernia SNPs ranged from 20.89 to 143.94. All instrumental variables had F values >10, indicating no weak instrumental variable bias. Comprehensive information on the selected SNPs is available in Tables S3–S6. The bidirectional MR analysis results are presented in Figure 2 and Tables S7 and S8. The sensitivity analysis results are presented in Tables S9 and S10.

IVW findings of causal relationships between 41 circulating inflammatory cytokines and five types of hernia. (a) Causal relationship of circulating inflammatory cytokines to hernia and (b) causal relationship of hernia to circulating inflammatory cytokines. IVW, Inverse variance weighted.
Causal effect of circulating inflammatory cytokines on the risk of hernia
As shown in Figure 2(a) and Table S7, the IVW method identified 16 associations between inflammatory factors and the risk of five types of hernia with p-values <0.05, suggesting possible causal relationships. However, with an FDR threshold of <10%, only the associations of stem cell growth factor beta (SCGFb) and beta nerve growth factor (bNGF) with diaphragmatic hernia remained significant. Upon further analysis, bNGF was excluded because its MR Egger’s β-value was in the opposite direction to those obtained using the other three MR methods. Consequently, SCGFb was the only significantly associated inflammatory factor found to be negatively correlated with the development of diaphragmatic hernia, with an OR of 0.884 (95% confidence interval [CI]: 0.819–0.955; p = 0.0016; FDR = 0.0656) as illustrated in Figure 3(a).
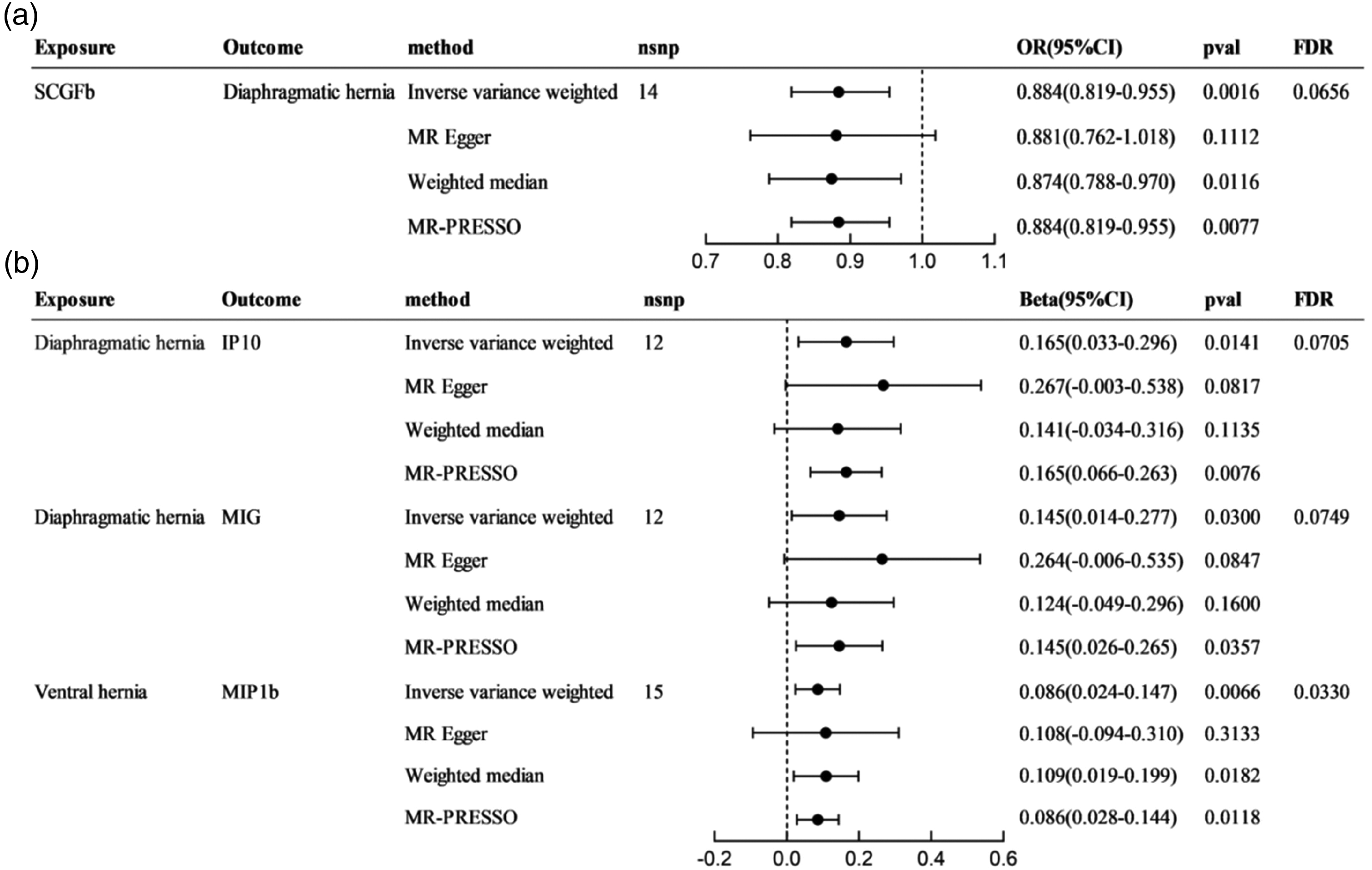
Forest plots of statistically significant results from bidirectional Mendelian randomization. (a) Forest plots for causal effect of circulating inflammatory cytokines on hernia and (b) forest plots for causal effect for hernia on circulating inflammatory cytokines.
As detailed in Table 1, there was no evidence of heterogeneity (Cochran’s Q = 14.520, p = 0.338) or horizontal pleiotropy (MR-Egger intercept test p = 0.951). The MR-PRESSO test further supported these findings, with no significant outliers identified. Leave-one-out analysis indicated the robustness and consistence of the results, as depicted in Figure 4.
Sensitivity analyses between circulating inflammatory cytokines and hernia.

SCGFb, stem cell growth factor beta; IP10, interferon gamma-induced protein 10; MIG, monokine induced by interferon-gamma; MIP1b, macrophage inflammatory protein-1b; MR-PRESSO, Mendelian Randomization Pleiotropy RESidual Sum and Outlier.
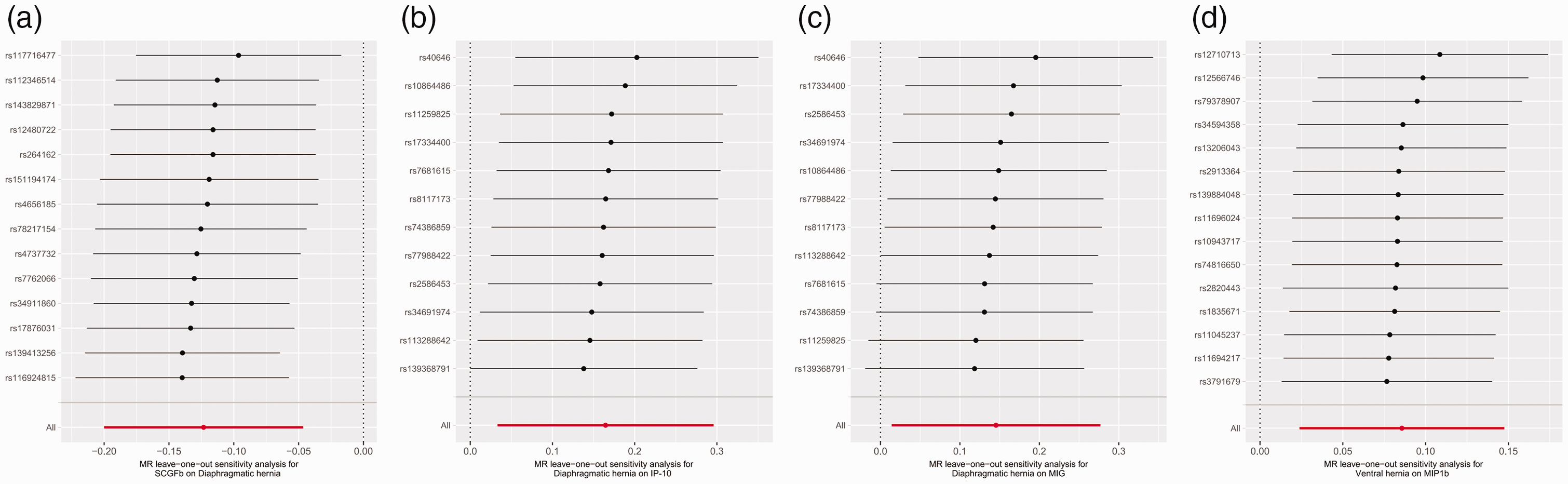
Results of leave-one-out sensitivity analysis. (a) SCGFb in diaphragmatic hernia. (b) Diaphragmatic hernia in IP-10. (c) Diaphragmatic hernia in MIG and (d) Ventral hernia in MIP1b. SCGFb, stem cell growth factor beta; IP10, interferon gamma-induced protein 10; MIG, monokine induced by interferon-gamma; MIP1b, macrophage inflammatory protein-1b.
Overall, our MR analysis provided significant evidence of a causal connection between circulating SCGFb levels and the risk of diaphragmatic hernia. The findings were robust across various sensitivity analyses, confirming the reliability of the association. No evidence was found to suggest that other circulating inflammatory cytokines are relevant to the risk of hernia.
Causal effect of hernia on levels of circulating inflammatory cytokines
According to Figure 2(b) and Table S8, several associations between hernia and the level of inflammatory factors were identified using the IVW method with a p-value of <0.05. Specifically, diaphragmatic hernia was associated with interferon gamma-induced protein 10 (IP10) and monokine induced by interferon-gamma (MIG); femoral hernia was associated with growth-regulated-alpha and IL-1 receptor antagonist; inguinal hernia was associated with TNF-related apoptosis-inducing ligand; and ventral hernia was associated with macrophage inflammatory protein-1b (MIP1b). However, after FDR adjustment, only the associations between diaphragmatic hernia and IP10, diaphragmatic hernia and MIG, and ventral hernia and MIP1b remained significant. Importantly, the direction of the IVW β-values for these associations was matched with findings from the other three complementary MR methods, indicating robust results. For diaphragmatic hernia, a positive correlation was observed with IP10 (β: 0.165; 95% CI: 0.033–0.296; p = 0.0141; FDR =0.0705) and MIG (β: 0.145; 95% CI: 0.014–0.277; p = 0.0300; FDR = 0.0749). Similarly, ventral hernia showed a positive correlation with MIP1b (β: 0.086; 95% CI: 0.024–0.147; p = 0.0066; FDR = 0.0330), as illustrated in Figure 3(b).
As shown in Table 1, the p-values for both the Cochran’s Q test and MR-Egger intercept test exceeded 0.05, suggesting no significant heterogeneity or horizontal pleiotropy. Leave-one-out analysis (Figure 4) for the associations of diaphragmatic hernia with IP10 and ventral hernia with MIP1b confirmed the robustness of these associations, with all results remaining on the same side of zero. However, for the association between diaphragmatic hernia and MIG, the CIs of the effect estimates crossed zero when certain SNPs were removed. Despite this, the variations were minimal, with most effect estimates clustering on the same side of zero. This suggests that individual SNPs did not significantly affect the overall results, maintaining stability in the overall effect estimates. Furthermore, the MR-PRESSO global test did not identify any significant outliers.
In summary, our reverse MR analysis identified significant positive correlations between diaphragmatic hernia and both IP10 and MIG, as well as between ventral hernia and MIP1b. These associations were supported by various sensitivity analyses, affirming the reliability of these findings, except for the association between diaphragmatic hernia and MIG, which exhibited slight instability upon the exclusion of certain SNPs.
Discussion
Inflammatory factors, which are broadly categorized into chemokines, growth factors, interleukins, and other immune-related factors, play crucial roles in the body’s inflammatory and immune responses. Chemokines, such as IP10, MIG and MIP-1b, are involved in the recruitment of immune cells in injury or infection, and they have been implicated in various pathological conditions, including chronic inflammation and fibrosis. 34 Growth factors, including SCGFb, promote cell proliferation, tissue repair, and angiogenesis; however, when dysregulated, these can contribute to pathological fibrosis and impaired tissue remodeling. 35 Interleukins, such as IL-6 and IL-8, are central mediators of both acute and chronic inflammation, influencing the balance between pro-inflammatory and anti-inflammatory pathways. 36 Other factors also play important roles in immune modulation and tissue repair. In the case of hernia, excessive or prolonged inflammation may lead to the disruption of collagen synthesis, reduced tissue repair, and ultimately the formation of hernia. 10 Therefore, targeting key inflammatory cytokines through reducing their levels or inhibiting their progressive increase could help mitigate the harmful effects of excessive inflammation, preventing tissue damage and potentially offering new therapeutic approaches to hernia prevention and management.
In this study, we performed a bidirectional two-sample MR analysis to explore the causal relationships between 41 circulating inflammatory factors and five types of hernia. We identified SCGFb as causally linked to a reduced risk of diaphragmatic hernia, aligning with its roles in cellular growth and tissue repair. 37 Conversely, reverse MR analysis showed that diaphragmatic hernia was associated with increased levels of IP10 and MIG, and ventral hernia was linked to elevated MIP1b levels. No causal associations were found between the remaining cytokines and hernia. However, this lack of significance does not imply that these cytokines are irrelevant to hernia pathogenesis. Cytokines such as IL-6, IL-8, and TNF-α are well recognized for their roles in autoimmune diseases, infections, and chronic inflammation. Several factors may explain this result. First, cytokine pathways often exhibit biological redundancy, meaning the effects of certain cytokines might be less pronounced in hernia development owing to compensation by other cytokines. Additionally, some cytokines are primarily involved in acute inflammatory responses and may have a more transient or context-specific role, which might not have been captured in our analysis owing to its reliance on genetic predisposition and steady-state levels of circulating cytokines. These findings emphasize the complexity of immune responses in hernia pathogenesis and suggest that the development of hernia is likely influenced by a network of cytokines, some of which may not show clear causal relationships in large-scale genetic studies.
SCGFb is a cytokine involved in hematopoiesis and cellular growth, playing a key role in immune modulation and tissue repair. Although direct research on SCGFb’s link to diaphragmatic hernia is limited, its protective effects may stem from its ability to reduce inflammation, promote angiogenesis, and enhance collagen production. SCGFb has been shown to induce macrophage polarization toward an anti-inflammatory M2 phenotype, 38 aiding in the removal of debris and mitigating inflammation. 39 It also supports stem cell function and improves endothelial recovery, ensuring proper oxygen and nutrient supply to damaged tissues, thereby facilitating healing. 40 Additionally, SCGFb promotes fibroblast differentiation and collagen production, 41 contributing to the formation of a new extracellular matrix, essential for tissue repair. 42 Our study suggests that higher SCGFb levels lower the risk of diaphragmatic hernia, thereby guiding clinical risk stratification of diaphragmatic hernia and pointing toward the exploration of SCGFb-based cytokine-targeted therapies. However, further mechanistic studies are necessary to elucidate the underlying processes.
As members of the CXC chemokine subfamily, IP10 and MIG play critical roles in immune and inflammatory responses. 43 Studies have identified these as key indicators of disease activity and severity. 44 IP10, typically low in healthy individuals, can be significantly elevated in response to IFN-γ, binding to CXCR3 and triggering severe inflammation or immunosuppression. 45 Severe cases of neonatal CDH are linked to persistent IP10 elevation, associated with intrathoracic hypoxia and chronic inflammation. 14 Similarly, MIG (CXCL9) is induced by IFN-γ and recruits T lymphocytes to inflammation sites, further exacerbating tissue injury and fibrosis in diaphragmatic hernia.46,47 Additionally, diaphragmatic hernia might lead to secondary complications such as infections or impaired lung function, further contributing to the upregulation of IP10 and MIG. 48 These chemokines not only indicate the severity of diaphragmatic hernia but also serve as biomarkers for monitoring treatment effectiveness and managing high-risk patients. Recently, next-generation sequencing analyses in CDH have demonstrated unexpectedly high activity in inflammatory pathways within lung tissues or cells, underscoring the complex interplay between systemic and local inflammation in this condition. 49 Our findings may therefore provide valuable preliminary insights for future research aimed at unraveling the contributions of inflammatory cytokines to the pathology of CDH, complementing tissue-specific investigations.
MIP1b (CCL4), another inflammatory chemokine, is released during infection or inflammation. 50 Mechanical stress and tissue injury from ventral hernia can trigger MIP1b release, attracting macrophages, 51 which aid in debris removal and tissue repair while also amplifying the inflammatory response. 50 Elevated MIP1b levels have been correlated with various illnesses, including eosinophilic airway inflammation and severe infections like COVID-19, indicating its potential as a biomarker for disease severity.52,53 Further research is needed to explore the therapeutic potential of targeting MIP1b in clinical settings.
Despite the valuable insights provided by our MR study, several limitations should be noted. First, the relatively lenient p-value threshold of <5 × 10−6 for selecting genetic instruments could increase the risk of false positives, although FDR correction was applied to address this issue. Second, our study population was predominantly of European descent, which limits the generalizability of our findings to other ethnic groups. Differences in genetic and environmental factors across populations may influence the relationships between inflammatory factors and hernia, necessitating further studies in diverse cohorts. Third, cytokine levels are dynamic and can vary with different physiological and pathological conditions whereas MR analysis relies on static genetic associations, which may not fully capture the temporal variability and complexity of cytokine regulation. Thus, we cannot distinguish whether the observed cytokine changes are the cause of the hernia or a secondary effect of physiologic changes caused by hernia itself. This distinction is particularly important in CDH, where the overall health of the patient is often severely compromised, which may result in a different cytokine profile. In addition, the cytokine changes observed in these patients are more likely to reflect downstream effects of the condition, including inflammation secondary to pulmonary dysplasia, cardiac abnormalities, and infections, rather than the primary drivers of herniation. Future studies should include stratification of congenital and acquired hernias to better understand these differences. Fourth, our study focused exclusively on peripheral circulating inflammatory factors and did not include tissue-level data that could provide insights into local inflammatory processes within herniated tissues. Finally, the lack of comprehensive databases linking hernia with related adverse events or complications prevented us from exploring the impact of inflammatory cytokines on hernia-related outcomes. These limitations underscore the need to interpret our results with caution and emphasize the necessity for validating the findings in well-powered experimental studies and clinical research.
Conclusion
This study provides the first bidirectional MR evidence of causal relationships between inflammatory cytokines and hernia subtypes, highlighting the protective potential of SCGFb and the association of IP10, MIG, and MIP1b with disease progression. These findings deepen our understanding of hernia pathophysiology and open avenues for cytokine-targeted therapies, such as those involving monoclonal antibodies or small-molecule inhibitors.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251315923 - Supplemental material for Causal relationship between circulating inflammatory cytokines and the risk of hernia: a bidirectional Mendelian randomization study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251315923 for Causal relationship between circulating inflammatory cytokines and the risk of hernia: a bidirectional Mendelian randomization study by Yaqin Qi, Changjiu Li, Xingyue Gao and Fangjie Zhang in Journal of International Medical Research
Footnotes
Acknowledgements
We extend our heartfelt appreciation to all investigators across relevant GWAS consortia and technicians associated with the IEU Open GWAS Project for their important contributions to data curation, collection, and summarization in the GWAS summary database (![]() ). We also extend our sincere thanks to scientists in various fields for their pivotal role in establishing several crucial open-source software projects related to this dataset, thereby facilitating the public availability of GWAS data.
). We also extend our sincere thanks to scientists in various fields for their pivotal role in establishing several crucial open-source software projects related to this dataset, thereby facilitating the public availability of GWAS data.
Author contributions
Yaqin Qi mainly contributed to the primary design and execution of formal analyses and authored the initial draft of the manuscript. Changjiu Li undertook data curation and validated the dataset. Xingyue Gao contributed to manuscript revision and scrutinized the results. Fangjie Zhang supervised the entire project. All the authors have thoroughly reviewed, critically discussed, and reached a consensus on the final version of the manuscript for publication.
Data availability statement
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research was funded by the Natural Science Foundation of Zhejiang Province (grant number LGF20H030007), 2023 General Research Projects of Zhejiang Provincial Department of Education (grant number Y202351300) 2024 Science and Technology Program of Traditional Chinese Medicine in Zhejiang Province (grant number 2024ZL715).
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
