Abstract
Objective
In this study, we examined whether levosimendan could lower B-type natriuretic peptide (BNP) and improve cardiac function in patients with heart failure who have diuretic resistance and low ejection fraction.
Methods
We conducted a cohort study among 120 participants from September 2022 to August 2023. We investigated the efficacy of treatment with levosimendan in patients with HF who exhibited diuretic resistance and had a low ejection fraction. All patients received recombinant human neuronal natriuretic peptide (rhBNP) and the treatment group additionally received levosimendan. In addition to the primary endpoints, safety measures were assessed, encompassing the monitoring of blood pressure, heart rate, and serum creatinine levels.
Results
Patients in the treatment and control groups were similar in age, sickness duration, weight, and sex ratio. Medication use, New York Heart Association classification, ejection fraction percentage, and other factors were evenly distributed. The treatment group showed increased urine volume and weight loss. Both groups had similar post-treatment systolic blood pressure, heart rate, and serum creatinine levels, indicating good safety.
Conclusion
Levosimendan in combination with rhBNP may improve diuretic resistance in patients with HF and low ejection fraction. Further multicenter trials are needed to confirm our findings.
Keywords
Introduction
Heart failure (HF) and cardiac dysfunction, which affect over 26 million people globally, are major global public health concerns. HF is on the rise worldwide as the population ages, and cardiac dysfunction is becoming increasingly more prevalent. 1 It is critical to recognize HF and cardiac failure in forensic and clinical medicine as these result in substantial morbidity and fatality rates. For inpatients, HF and cardiac diseases can be detected using clinical tests, such as electrocardiography and echocardiography. 2 HF and heart function can be assessed after death, but this highly challenging for forensic pathologists if there are no clinical medical records or no tests available. 3 It is particularly challenging to diagnose HF or heart failure after death if there are no typical morphological abnormalities. Numerous forensic research studies have shown that both NT-proBNP and BNP may be used to assess heart health in deceased individuals. Several postmortem and animal investigation materials have been used in these investigations. These may be applied in forensic medicine as postmortem biomarkers to identify cardiac dysfunction and HF. 4
Clinical evaluation of HF and cardiac dysfunction often use the key markers brain natriuretic peptide (BNP) and N-terminal pro-brain natriuretic peptide (NT-proBNP). 5 The most reliable biomarkers for determining the etiology of HF and heart dysfunction are BNP and NT-proBNP, according to the American College of Cardiology Foundation/American Heart Association and the European Society of Cardiology (ESC). These biomarkers may also be used to determine the degree of cardiac disease, to direct pertinent therapeutic approaches and estimate patient prognosis. The pathophysiology of BNP and HF is represented schematically in Figure 1.
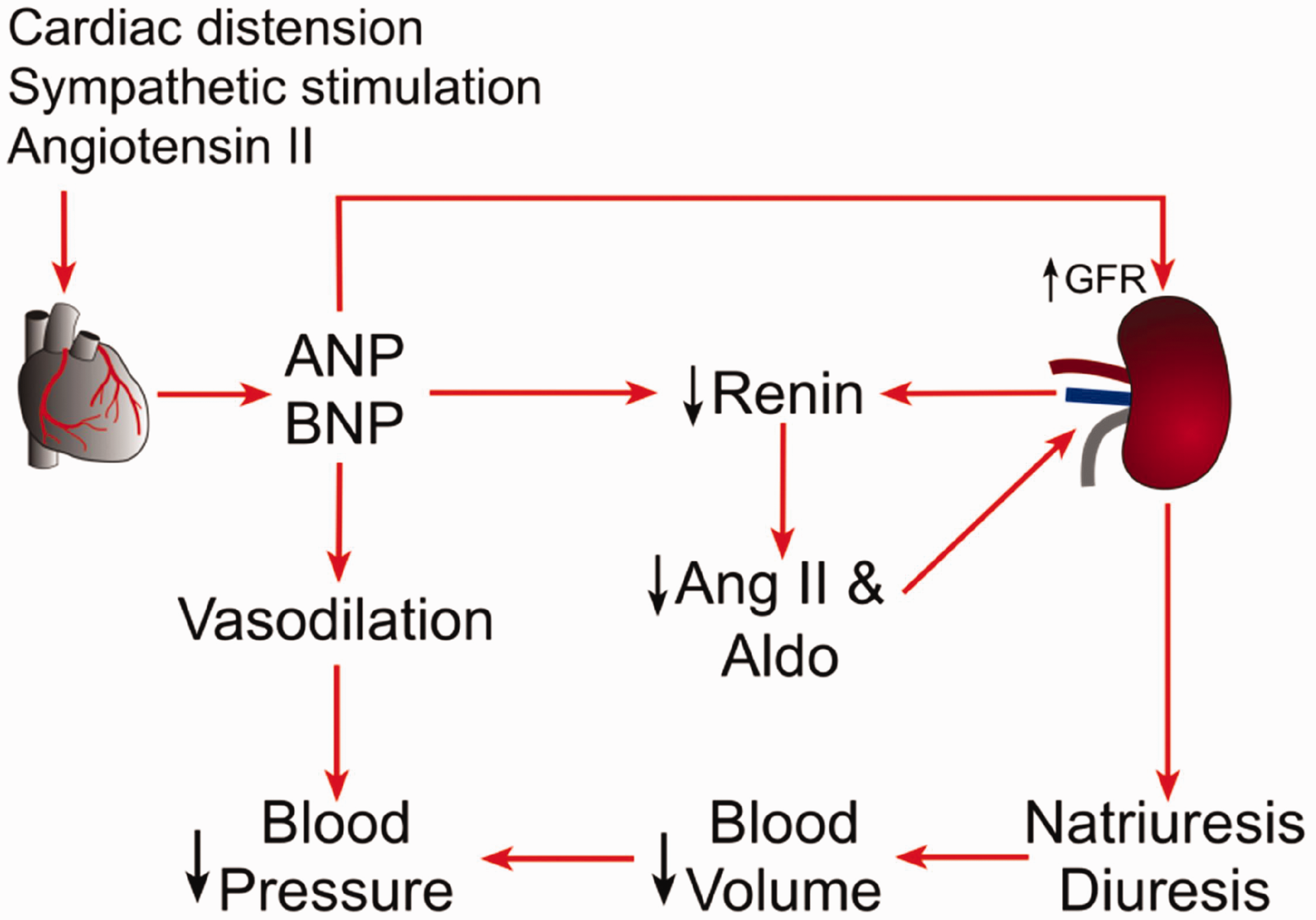
Pathophysiological relationship between B-type natriuretic peptide and heart failure.
Approximately 1% to 2% of adults in the United States are affected by HF, a multifactorial systemic illness. Depending upon the ejection fraction (EF), patients with HF may be divided into two groups, those with HF and normal EF or preserved EF (HFpEF). 6 Following the 2016 ESC recommendations for the diagnosis and management of chronic and acute conditions including long-term HF, all individuals with suspected acute HF should have their BNP and NT-proBNP levels measured. 7 For NT-proBNP and BNP, the upper limit of normal cutoff value in an acute scenario is 100 pg/mL and 300 pg/mL, respectively; in a non-acute context, these cutoffs are 35 pg/mL and 125 pg/mL, respectively. BNP levels can aid clinicians in differentiating between HF and other causes of dyspnea. BNP levels below 100 pg/mL are indicative of other causes of dyspnea rather than HF. With BNP levels between 100 and 500 pg/mL, HF should be diagnosed according to clinical judgment. With BNP ≥500 pg/mL, HF and cardiac dysfunction are likely and immediate HF treatment is advised. 8 Heart dysfunction and HF may coexist with other conditions indicative of heart failure, which can be used as a diagnostic tool. These conditions include cardiomyopathy, several types of arrhythmia, and ischemic heart disease. 9
According to past studies, treating acute ischemic heart disease more aggressively with the aim to lower ventricular wall tension has been proposed using BNP levels as an indicator. The degree of left ventricular dysfunction may be reflected in the association between acute ischemic heart disease and an increase in BNP levels. In patients with unstable coronary artery disease, BNP and NT-proBNP are equally effective predictors of adverse cardiovascular events. 10 In patients with acute myocardial infarction, BNP and NT-proBNP levels are assessed as well as biomarkers of myocardial damage including myoglobin, creatine kinase MB, and cardiac troponin T. NT-proBNP may be a more accurate diagnostic biomarker than BNP as it can remain elevated for as long as 12 weeks, on average. When determining the severity of ischemic heart disease and its prognosis in patients experiencing acute coronary syndrome, BNP and NT-proBNP can be helpful indicators; additionally, these are extremely accurate and precise markers of the severity of myocardial infarction. 11
NT-pro and BNPBNP are crucial in the diagnosis of HF, but these are also very useful in determining the extent and prognosis of HF. According to the results of Doppler echocardiography, BNP and NT-proBNP are the best independent predictors HFpEF. 12 The grading system used by the New York Heart Association (NYHA) includes NYHA classes I through IV according to increasing plasma BNP concentrations, which may indicate that plasma BNP levels are increased with increased HF severity. 13 In patients with cardiovascular disorders, decreased in plasma levels of NT-proBNP and BNP indicate an improvement in their clinical condition. The mortality risk is positively correlated with BNP or NT-proBNP values in cardiac dysfunction. 14
The synthetic endogenous hormone recombinant human neuronal natriuretic peptide (rhBNP) has the same sequence of amino acids as BNP. rhBNP is an established treatment for managing HF symptoms, particularly in patients with low EF as it efficiently lowers both preload and afterload by directly widening blood channels. Nesiritide is an effective rhBNP with several biological properties that are similar to those of endogenous BNP. These properties include facilitating natriuresis and diuresis, inhibiting the renin–angiotensin–aldosterone system, boosting heart output, reducing wedge pressure within pulmonary capillaries, and improving cardiac diastolic as well as systolic function. Nesiritide was approved in 2001 by the US Food and Drug Administration for the management of acutely decompensated HF. 15
The most successful therapies for advanced HF are heart transplantation and mechanical circulatory support. Patients must take medication when waiting for these treatments to maintain stable vital signs. Intravenous inotropic support includes vasopressors like inodilators such as levosimendan, dopamine, norepinephrine, and epinephrine, as well as other drugs. Levosimendan, a calcium-sensitizing cardiotonic drug, can improve myocardial contractility while also widening coronary and peripheral blood arteries. 16 The impact of pre- and post-load on the body can be mitigated by levosimendan via the activation of adenosine triphosphate (ATP), an enzyme that is sensitive to cell membrane potassium channels and enhances the responsiveness of myocardial systolic pressure protein to calcium. Additionally, levosimendan can avoid myocardial apoptosis and does not raise myocardial oxygen demand or remodeling. 17
In this study, we aimed to determine whether levosimendan can lower BNP and improve cardiac function in patients with HF who have diuretic resistance and low EF.
Methods
This cohort study was conducted at the cardiac care unit of Xingtai People’s Hospital China from September 2022 to August 2023. We aimed to assess the effectiveness of a treatment intervention with levosimendan among patients diagnosed with HF who demonstrated resistance to diuretics and had a low EF. Diuretic resistance is operationally defined as the need to administer more than two daily doses of loop diuretics. The diagnosis of HF adhered to the criteria delineated in the 2018 Chinese guidelines for the diagnosis and management of HF. The sample size was calculated using the World Health Organization calculator. Patients who met the criteria for eligibility were randomly assigned at a 1:1 ratio to either the control group or the treatment group with an equal number of research participants in each group. To avoid bias, a blinded technique was used.
Patients in both groups were given a complete pharmacological regimen for HF, which included thiazide diuretics, cardiotonic medicines, and vasodilators. As part of the admission process, all patients were given a furosemide injection with the objective of attaining a urine output above 0.5 mL/kg/hour. The experimental group received levosimendan in addition to the initial diuretic and rhBNP therapy; the control group only received rhBNP. Including rhBNP in both arms ensured that the potential effects of rhBNP were controlled to facilitate accurate analysis of the additional impact of levosimendan when given in combination with rhBNP. Participants were followed for a duration of 24 hours. The effectiveness of levosimenden was assessed by measuring the outcome variables 24-hour urine volume as well as changes in body weight and dyspnea score before and after the 24-hour treatment period. We compared the outcomes both within and between the two groups. Safety outcomes included changes in systolic blood pressure, heart rate, and serum creatinine levels before and after the administration of treatment.
The inclusion criteria were patients with HF and an EF <40%, and a NYHA functional classification I–IV. The exclusion criteria were patients diagnosed with acute coronary syndrome and those with an estimated glomerular filtration rate (eGFR) below 30 mL/minute/1.73 m2 indicating serious renal damage, as well as patients with systemic infection.
Ethics statement
Ethical approval was obtained from Xingtai People’s Hospital prior to the study (approval number: 2022-XT7-01, date: 2023-8-7). We de-identified all patient details to maintain patients’ privacy. Written informed consent was obtained from all participants. This study was conducted in accordance with the principles of the Declaration of Helsinki. The reporting of this study confirms the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 3
Statistical analysis
The statistical analysis was conducted using IBM SPSS 22.0 (IBM Corp., Armonk, NY, USA). Demographic characteristics are expressed as mean ± standard deviation or frequency and percentage. The chi-square test or Fischer’s exact test was used as an independent t-test to compare two groups, assuming that the data followed a normal distribution and had equal variance. The Mann–Whitney test was used for data with a non-normal distribution. A paired t-test was used to compare the data within a particular group. A significance level of P < 0.05 was deemed to indicate statistical significance.
Results
The demographic and baseline characteristics of the control (n = 60) and treatment (n = 60) groups are shown in Table 1. The sex distribution was balanced, with 53.33% men in the control group and 63.33% in the treatment group. Age, disease course, and weight were similar between groups. Most patients in both groups took angiotensin-converting enzyme inhibitors and angiotensin receptor blockers, beta-blockers, and aldosterone receptor antagonists, with no significant differences. Most patients in both groups were categorized as class III or class IV for NYHA cardiac performance, with no significant difference. No significant differences were found in EF percentages or eGFR. The two groups had similar distributions of dilated heart disease, coronary heart disease, rheumatic heart disease, atrial fibrillation, and arterial hypertension. The baseline characteristics and demographics of the control and treatment groups were similar, making the study comparisons credible (Table 1).
Demographic and baseline characteristics of patients.
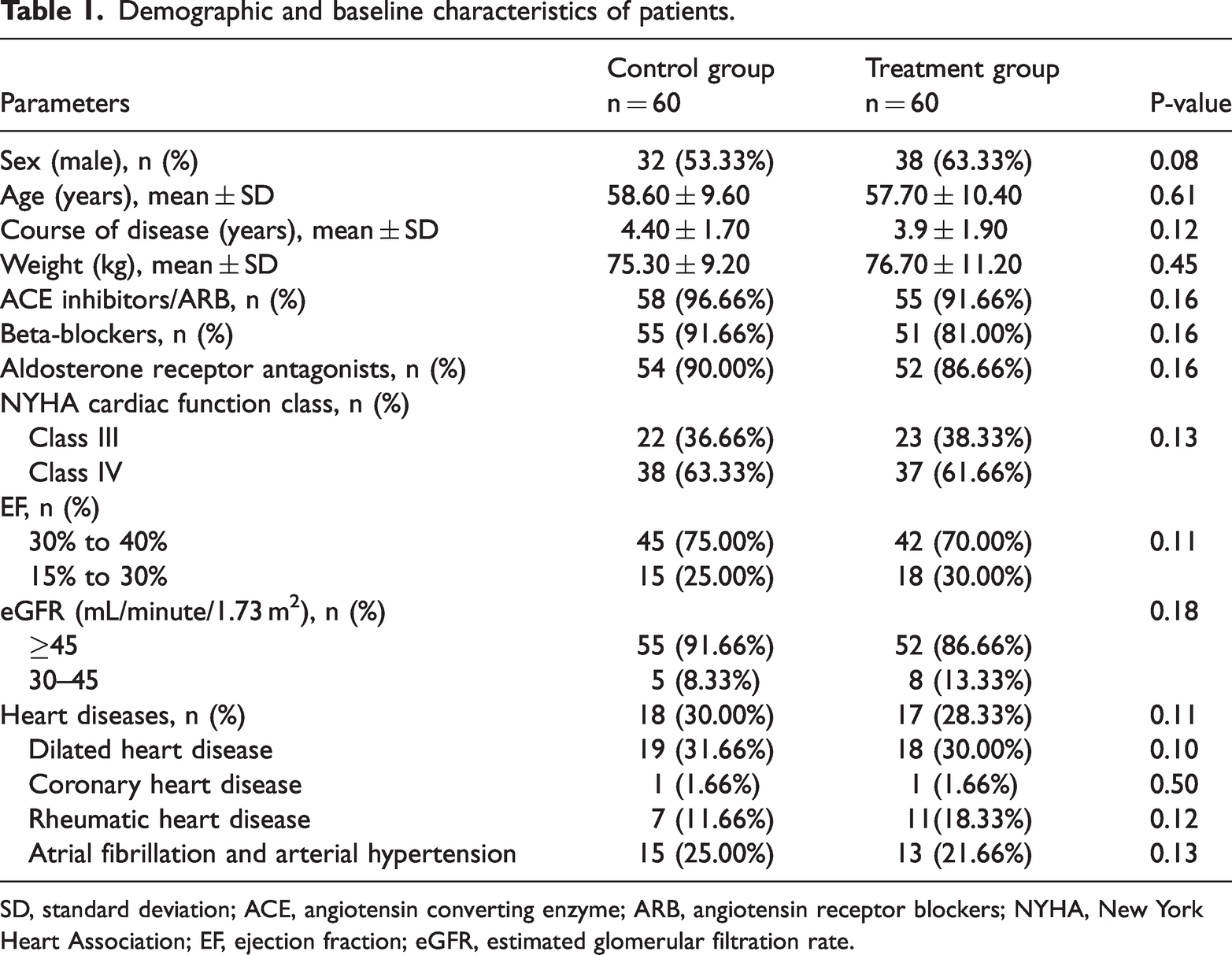
SD, standard deviation; ACE, angiotensin converting enzyme; ARB, angiotensin receptor blockers; NYHA, New York Heart Association; EF, ejection fraction; eGFR, estimated glomerular filtration rate.
Table 2 shows the difference in treatment effectiveness between the control and levosimendan groups before and after treatment. Before treatment, both groups had similar urine volumes: 749 ± 125 mL for the control group and 779 ± 109 mL for the treatment group. Post-treatment, urine volume in the treatment group increased significantly to 1879 ± 119 mL (P = 0.045) whereas the control group remained unchanged. After treatment, weight in the control group increased from 73.29 ± 8.90 kg to 73.8 ± 9.08 kg; patients in the treatment group had a decrease in weight from 81.69 ± 12.11 kg to 79.1 ± 4.9 kg. Before treatment, dyspnea scores were identical between groups; however, after treatment, the levosimendan group had a larger increase (to 5.41 ± 2.31) than the control group (to 4.37 ± 2.08). While losing weight, the treatment group showed considerable improvements in urine volume but a somewhat larger rise in dyspnea than the control group.
Findings for other indicators in each group.

Table 3 shows the pre- and post-treatment safety indices for the control and treatment groups. Both groups exhibited nearly identical systolic blood pressure levels before treatment: 119.1 ± 10.5 mmHg for the control group and 120.3 ± 13.8 for the treatment group. Post-treatment, systolic blood pressure in the control group increased slightly to 119.5 ± 11.5 mmHg whereas that in the treatment group remained stable at 119.8 ± 18.4 mmHg. Both groups had comparable heart rates before treatment: 77 ± 17 beats/minute in the control group and 79 ± 15 beats/minute in the treatment group. Post-treatment, heart rate in the control group increased to 79 ± 15 beats/minute whereas that in the treatment group declined to 76 ± 16 beats/minute. Serum creatinine levels were similar before treatment, with the control group at 82.39 ± 16.81 µmol/L and the treatment group at 81.45 ± 19.87. Both groups experienced minimal changes after treatment, with serum creatinine levels rising to 84.39 ± 17.17 µmol/L in the control group and lowering to 79.81 ± 19.65 µmol/L in the treatment group. Both groups maintained stable systolic blood pressure, heart rate, and serum creatinine levels, suggesting that the treatment did not significantly affect these safety markers.
Pre- and post-treatment efficacy and safety in each group.

Discussion
The most prevalent disease in the industrialized world is HF. Levosimendan (Simdax) is a novel injectable drug that works by producing myofilaments that are more sensitive via activation of potassium ATP-dependent receptors in vascular smooth muscle and the conversion of calcium to provide inotropic and vasodilator effects. Levosimendan infusion enhances the boosting of cardiac output by raising the heart rate and stroke volume while lowering pulmonary capillary wedge tension. The drug contains an active metabolite with an approximately 80-hour half-life; therefore, hemodynamic effects from infusions spanning 6 to 24 hours persist for 7 to 10 days. 18 When compared with placebo and dobutamine, a single levosimendan injection lasting 6 to 24 hours led to hemodynamic shifts, symptomatic relief, and a decrease in left ventricular systolic dysfunction over the subsequent 2 to 4 weeks among patients with severe HF, improving rates of morbidity and mortality. 19 There was no decline in this early advantage over 6 months, according to long-term follow-up. Although approved in many countries, levosimendan is not licensed in North America for treating decompensated HF. Additional research evaluating the effects of levosimendan in patients with acute HF and left ventricular systolic dysfunction including placebo and dobutamine are currently being conducted. Levosimendan may replace other therapies as the accepted approach for treating serious cardiac failure if its advantages are further confirmed. 20
Patients with decompensated HF have higher BNP concentrations than healthy individuals. A meta-analysis assessed the effectiveness and safety of levosimendan in treating left HF. Among individuals with decompensated HF, levosimendan treatment showed better cardiac performance and a substantial decrease in plasma BNP levels. 21 In people with advanced HF, levosimendan is typically used; however, no studies have clarified the mechanisms underlying the benefits of levosimendan in people with severe HF. Using clinical indicators, studies have assessed the effectiveness of levosimendan. 22 Among individuals who have advanced HF, intravenous levosimendan may lower BNP levels and raise left ventricular EF, which may lower mortality with the shortest follow-up time possible. A unique positive inotropic calcium sensitizer drug, levosimendan is used to treat acute left cardiac disease. One study used tissue Doppler echocardiography to assess the effect of levosimendan. That study comprised individuals with ischemic cardiomyopathy and acute decompensated reduction in the EF of the left ventricle to <40% owing to coronary artery disease. Maximum systolic, maximum early (Ea), maximum late (Aa), and peak diastolic annular velocities, as well as the Ea/Aa ratio from the tricuspid lateral annulus were assessed using tissue Doppler echocardiography; systolic pulmonary arterial pressure was also assessed before and 24 hours after therapy. Right ventricular systolic and diastolic performance was enhanced with levosimendan treatment. 23 Late left ventricular heart disease is typically complicated by right ventricular (RV) dysfunction, which worsens the patient prognosis. The novel indicator levosimendan enhances hemodynamics or the systolic and diastolic activity of the left ventricle in patients experiencing severe HF. 24 However, the impact on RV function in these patients has not been well evaluated. In a randomized experiment, 54 individuals with advanced HF were evaluated to investigate the effects of levosimendan and a placebo on various right ventricular systolic and diastolic parameters, as well as echocardiographic indicators of left ventricular systolic dysfunction. 25 Only the levosimendan group saw a substantial increase in maximum tricuspid annular velocity (S wave) during tissue Doppler imaging. Following levosimendan treatment, both the RV and late diastolic velocities (E/A) began to increase as the early diastolic velocity (E wave) does ratio on tissue Doppler imaging was dramatically increased. Individuals receiving levosimendan had a considerable reduction in systolic pulmonary arterial pressure. 26 Levosimendan decreases the interleukin (IL)-6 to IL-10 ratio and is altered by BNP concentrations to favor the latter, which helps to control the neurohormonal and inflammatory state. Levosimendan may provide further therapeutic benefits by enhancing systolic and diastolic RV performance in patients with advanced HF. 27
HF causes an increase in the release of BNP. Plasma levels of BNP and NT-proBNP are decreased following effective pharmacologic therapy for HF, suggesting that measurement of plasma BNP may be useful in assessing treatment. Levosimendan has been found to reduce both long- and short-term mortality among individuals with serious HF; however, current findings from extensive clinical trials have been fairly contentious. 28 To clear up the ambiguity surrounding the effect of levosimendan on the prognosis of individuals with decompensated HF, further clinical investigations are required.
Levosimendan has been found to be more effective in relieving symptoms than dobutamine, according to earlier trials contrasting the two drugs. Secondary measures taken immediately after or during a dobutamine injection have been used in echocardiographic investigations evaluating the long-lasting left ventricular diastolic functions that are affected by continuous dobutamine and levosimendan infusions. 29
Study limitations
The endpoint analysis in this study concentrating on 24-hour urine volume and its single-center design are the main study limitations. Caution is advised in generalizing the findings to a larger and more heterogeneous patient population. However, this trial can serve as an important reference for investigating the advantages of levosimendan in addition to rhBNP for patients with HF who have diuretic resistance and low EF.
Future recommendations
Multicenter, randomized, controlled clinical trials with larger patient groups and longer follow-up durations are warranted to confirm our findings. Such investigations can help clarify the efficacy and safety of this therapeutic approach and improve patient management in this patient population.
Conclusion
Levosimendan in addition to rhBNP may benefit diuretic resistance in patients with HF and low EF by increasing urine volume, reducing body weight, and improving dyspnea. Additionally, the combination of levosimenden with rhBNP appears safe in this patient population.
Footnotes
Author contributions
All authors have contributed equally to the conception of the study and the study design, the literature review, data acquisition and analysis, as well as manuscript writing. All authors have read the manuscript and given their final approval for its submission.
Data availability and sharing
All data used in this research are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
