Abstract
Aim
To determine the primary diseases and prognoses of patients with highly elevated levels of B-type natriuretic peptide (BNP; >1000 pg/ml), with or without heart failure.
Methods
Medical records and echocardiograms of patients with BNP levels that fell within one of three predetermined categories (>1000 pg/ml, 200–1000 pg/ml and <200 pg/ml) were retrospectively reviewed.
Results
There were no significant between-group differences in duration of hospitalization. Patients with BNP levels >1000 pg/ml (n = 103) or 200–1000 pg/ml (n = 100) had significantly worse 3-year survival than those with BNP levels <200 pg/ml (n = 100). The majority of patients (64/103) in the BNP >1000 pg/ml group had heart failure. The main cause of death in patients with other causes of BNP levels >1000 pg/ml (39/103) was community acquired pneumonia.
Conclusion
A BNP level >1000 pg/ml has clinical importance in primary care medicine and hospital settings.
Introduction
The 32-amino acid polypeptide, B-type natriuretic peptide (BNP), is used in the diagnosis and evaluation of heart failure and is predictive of patient prognosis. 1 BNP levels differ in patients with clinically similar presentations, and can be higher in individuals without heart failure compared with other conditions, however.2,3 In addition, the clinical characteristics of patients with elevated BNP levels are heterogeneous. BNP may not be an independent prognostic marker of heart failure, since increased BNP levels can be caused by other conditions, including renal failure. 4
Studies have indicated that BNP is an independent predictor of death in patients with heart failure, regardless of their history of cardiovascular disorders,5,6 but the threshold prognostic level of BNP and the length of follow-up vary between studies.7,8 The initial diagnostic BNP level of 40–50 pg/ml for heart failure was increased to 100 pg/ml, 7 but elderly women with hypertension-associated renal impairment (without heart failure) may have BNP levels >200 pg/ml. BNP is secreted along with a biologically inactive 76-amino acid N-terminal fragment (NT-proBNP). We chose to evaluate BNP instead of NT-pro-BNP because NT-pro-BNP levels are more dependent on kidney dysfunction than BNP. 9
The objective of this study was to determine the primary diseases and prognoses of patients with highly elevated BNP levels (>1000 pg/ml), with or without heart failure.
Patients and methods
Study population
This retrospective cohort study included residents of Himi City, Toyama Prefecture, Japan, who visited Kanazawa Medical University Himi Municipal Hospital between January 2010 and December 2011. Inclusion criteria were: sufficient medical records; BNP levels in one of three categories (highly elevated [>1000 pg/ml], moderately elevated [200–1000 pg/ml] and normal or slightly elevated [<200 pg/ml]; availability of laboratory data; provision of written informed consent. Patients were followed-up for 3 years.
The study was approved by the ethics board of the Kanazawa Medical University Himi Municipal Hospital, Himi, Japan.
Data collection
Baseline data included standardized interviews, blood tests, blood-pressure (BP) measurements, laboratory data, electrocardiograms and echocardiograms. Echocardiograms were analysed for left ventricular ejection fraction (LVEF; Deveraux formula modified for the American Society of Echocardiography guidelines). Heart failure with preserved ejection fraction or right ventricular hypertension were not excluded as complications or major causes of death. Heart failure was diagnosed using specific symptoms such as dysponea and fatigue, and by history and physical examination. Systolic BP was defined as the mean of two lowest of three standardized BP measurements. 1
BNP quantification
Plasma BNP was prospectively quantified using blood samples collected at the patient’s initial hospital visit. In brief, samples were taken using a human BNP kit (MI02 Shionogi; Shionogi & Co. Ltd., Osaka, Japan) as described. 10
Statistical analyses
Data were expressed as mean ± SD and compared using one-way analysis of variance (ANOVA). Linear regression analysis was used to evaluate the association between plasma BNP levels and other variables. Logistic regression analyses were used to test the associations between laboratory parameters and clinical diagnosis in each BNP group (adjusting for covariates). P-values < 0.05 were considered statistically significant. All analyses were performed using StatMate version 4.0 (GraphPad Software, La Jolla, CA, USA) for Windows®.
Results
The study included 303 patients (122 male/181 female; mean age 78.4 ± 12.4 years; age range 57–96 years. A PRISMA flowchart of the study is shown in Figure 1. Patients were stratified according to BNP levels: >1000 pg/ml (n = 103); 200–1000 pg/ml (n = 100); and <200 pg/ml (n = 100). Demographic and clinical characteristics of the patients stratified according to BNP level are shown in Table 1. Patients with BNP levels <200 pg/ml were significantly younger than those with BNP levels >1000 pg/ml (P < 0.001). There were no other between-group differences in age or sex distribution.
PRISMA flowchart of the study. Demographic and clinical characteristics of patients included in a study to determine the cause of elevated B-natriuretic peptide (BNP) levels, stratified by circulating BNP level. Data presented as mean ± SD or n (%) of patients. One-way analysis of variance; Kruskal–Wallis test. NS, not statistically significant (P ≥ 0.05); LVEF; left ventricular ejection fraction, FS; fractional shortening, LVDd; left ventricular diastolic dimension, LVDs; left ventricular systolic dimension, AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase; γ-GTP, γ-glutamyltransferase; LDH, lactate dehydrogenase; BUN, blood urea nitrogen; eGFR, estimated glomerular filtration rate; HDL, high-density lipoprotein cholesterol; LDL, low-density lipoprotein cholesterol.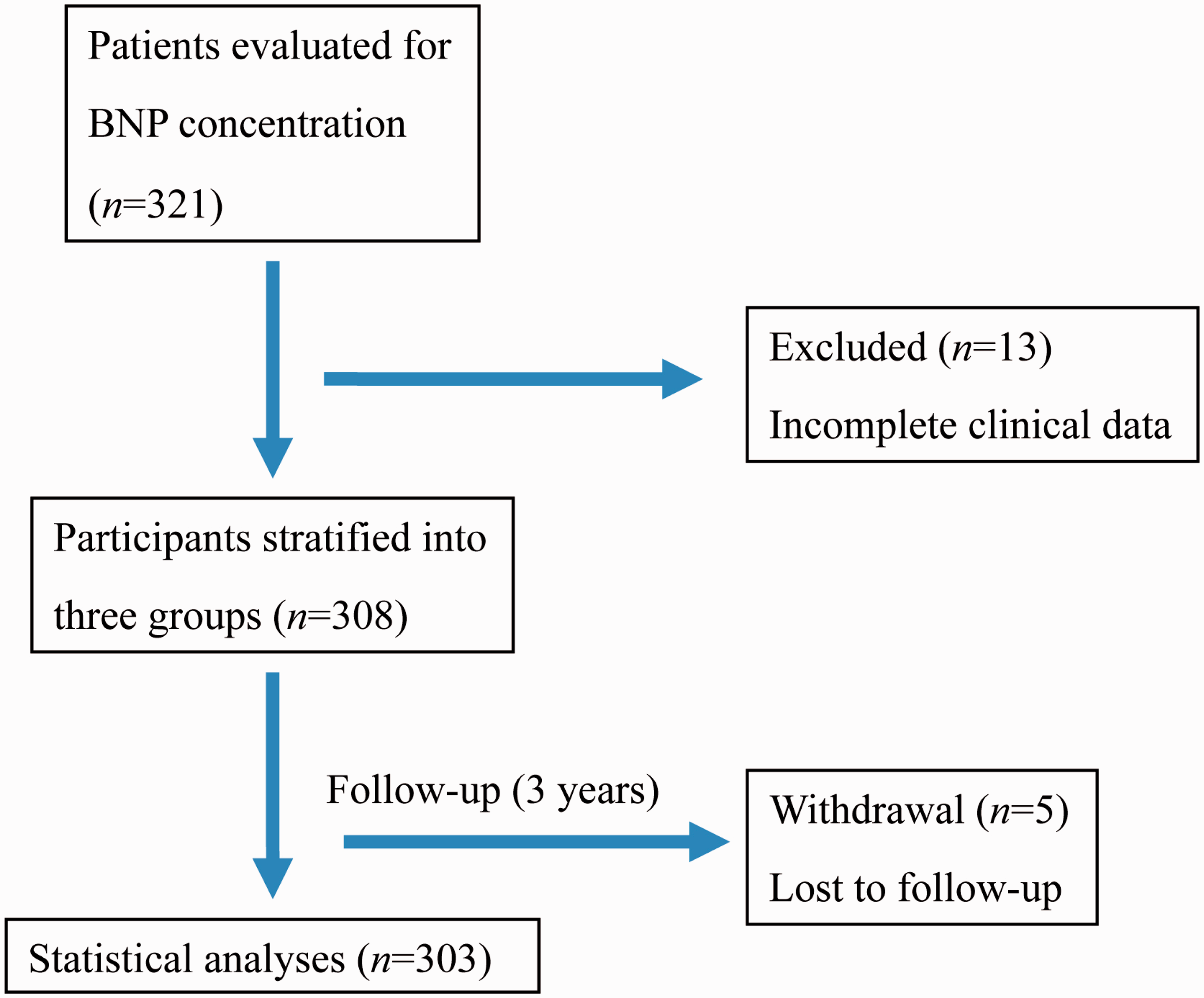

Echocardiography parameters differed significantly between groups (LVEF, P = 0.01; fractional shortening, P < 0.001; left ventricular diastolic dimension, P = 0.01; left ventricular systolic dimension, P < 0.001; Table 1). Creatine kinase and lactate dehydrogenase levels were significantly higher in patients with BNP >1000 pg/ml compared with both other groups (P < 0.05 for each comparison; Table 1). Patients with BNP levels >1000 pg/ml had significantly lower estimated glomerular filtration rate (eGFR; P < 0.001), serum albumin (P < 0.001), total cholesterol (P < 0.001), triglycerides (P = 0.01), red blood cell count (P = 0.002), haemoglobin (P = 0.004), haematocrit (P = 0.024) and platelet count (P < 0.001) than both other groups (Table 1). There were no significant between-group differences in C-reactive protein (CRP), white blood cell count, duration of hospital stay, and sodium or chloride levels. Patients with BNP levels >1000 pg/ml had significantly lower 3-year survival rates than those in both other groups (P < 0.001).
Demographic, clinical and prognostic characteristics of patients with circulating B-natriuretic peptide levels >1000 pg/ml, stratified by the presence or absence of heart failure.

Data presented as mean ± SD or n (%) of patients.
One-way analysis of variance
NS, not statistically significant (P ≥ 0.05); LVEF; left ventricular ejection fraction, FS; fractional shortening, LVDd; left ventricular diastolic dimension, LVDs; left ventricular systolic dimension, AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase; γ-GTP, γ-glutamyltransferase; LDH, lactate dehydrogenase; BUN, blood urea nitrogen; eGFR, estimated glomerular filtration rate; HDL, high density lipoprotein cholesterol; LDL, low density lipoprotein cholesterol.
Logistic regression analysis of laboratory parameters in patients with circulating B-natriuretic peptide levels >1000 pg/ml, stratified by death versus survival during 3 years’ follow-up.
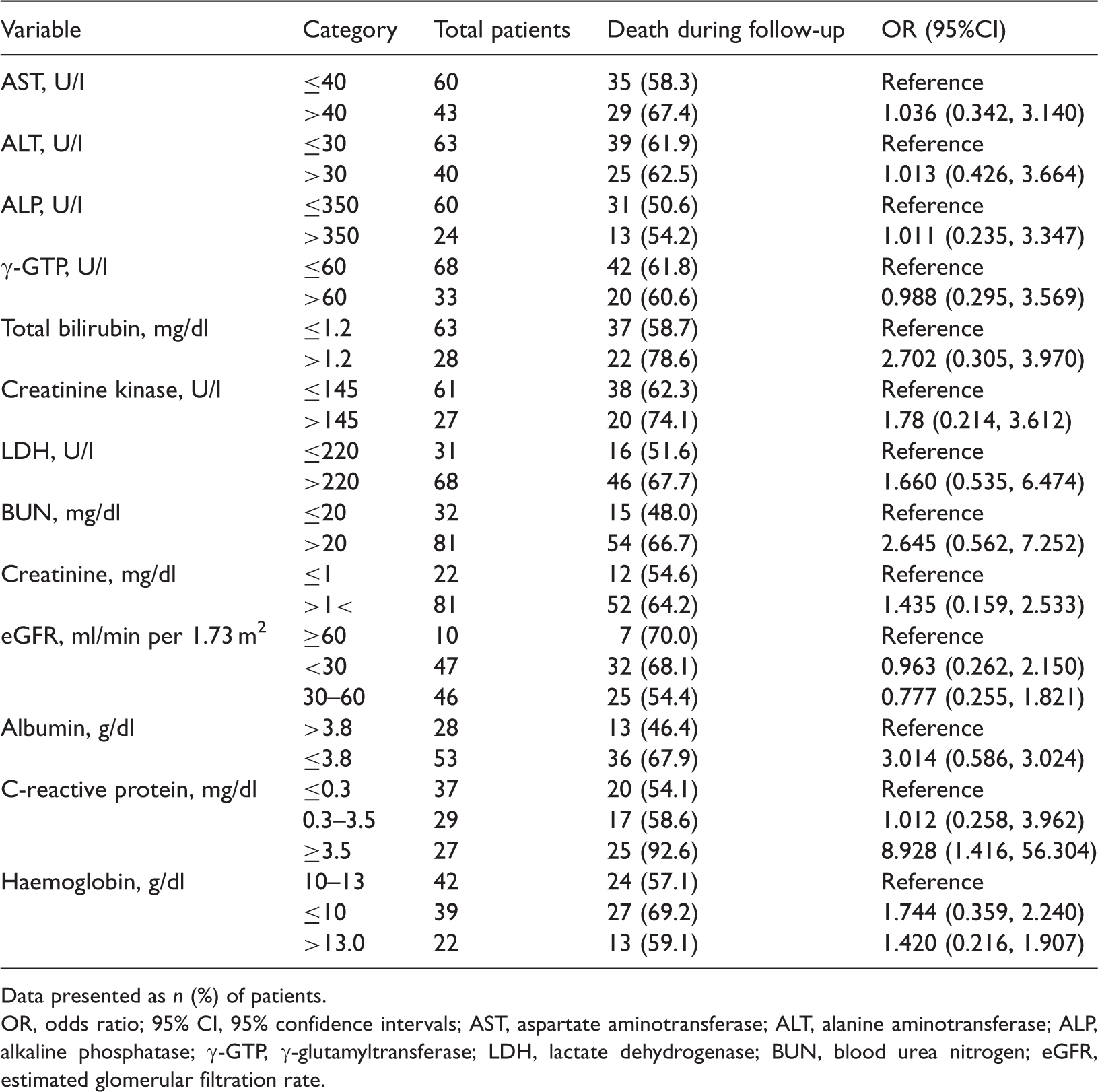
Data presented as n (%) of patients.
OR, odds ratio; 95% CI, 95% confidence intervals; AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase; γ-GTP, γ-glutamyltransferase; LDH, lactate dehydrogenase; BUN, blood urea nitrogen; eGFR, estimated glomerular filtration rate.
Cause of death during 3 years’ follow-up of patients stratified by circulating B-natriuretic peptide level.

Data presented as n (%) of patients.
P < 0.05 versus both other groups; one-way analysis of variance.
Discussion
The circulating BNP concentration has been shown to have prognostic value in community acquired pneumonia,11,12 with its accuracy being comparable with that of the pneumonia severity index. 12 Hypoalbuminaemia on admission and the persistence or development of hypoalbuminaemia during hospitalization have been associated with poor prognosis in patients with heart failure or pneumonia.13,14 BNP and albumin were negatively correlated with major adverse cardiac events in those with congestive heart failure, 7 and it is possible that heart failure negatively influences causes of death such as pneumonia and lethal arrhythmia.
Patients with BNP >1000 pg/ml in the present study had higher creatinine kinase and lactate dehydrogenase levels as well as lower eGFR and platelet counts than those in the two other groups, despite there being no between-group differences in C-reactive protein and white blood-cell count. This suggests that patients with BNP >1000 pg/ml may have myocardial or skeletal muscle damage and renal dysfunction without systemic inflammation. BNP levels have a strong inverse association with eGFR in patients with or without heart failure, 15 and elevated BNP may be caused by renal dysfunction. BNP is correlated with platelet volume, which is in turn negatively correlated with platelet count. 16 Further studies are required to fully elucidate the relationships between BNP and laboratory parameters.
It is known that BNP is a simple and powerful predictor of mortality in patients with community acquired pneumonia.12,17,18 The mechanism of BNP secretion may be related to the development of pulmonary arterial hypertension due to right heart dysfunction, and endothelial inflammation and proliferation in the pulmonary artery.18,19
This study has several limitations inherent to its retrospective design. The risk of bias in the measurements should be low for prognostic factors because BNP was appropriately defined and measured. In addition, we were aware of the important confounders (age, sex and renal failure). 9 The findings of this study require confirmation in prospective studies with serial assessment of other laboratory data. Limitations include the fact that patient diagnoses were established by unselected physicians. Secondly, although we excluded participants without follow-up records, the contribution of such patients cannot be quantified. Thirdly, since BNP levels were measured only once in each patient, it was not possible to analyse post-treatment BNP. Finally, it is not possible to extrapolate our findings to populations of other ethnicities, since their BNP levels may be different from our study population.
In conclusion, we have confirmed the clinical importance of BNP >1000 pg/ml in primary care medicine and hospital settings. Further studies are required to test the external validity, utility, generalizability and cost effectiveness of BNP quantification in different acute care environments representing racially and socio-economically diverse patient populations.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
