Abstract
Objectives
Central nervous system complications of acute pancreatitis (AP) can result in cerebral edema (CE). We assessed the risk of serious outcomes and health care features associated with CE in patients hospitalized with AP.
Methods
We conducted a retrospective cohort study using the National Inpatient Sample database. Patients were divided into the AP-CE group that developed CE and the AP-only group that did not. Outcome data were analyzed using Stata software.
Results
Among 543,464 patients hospitalized with AP, 220 (0.04%) developed CE. In multivariate analysis, primary outcomes included increased length of hospital stay (adjusted odds ratio [aOR] 10.1; 95% confidence interval [CI] 0.50–19.70), hospital charges (aOR USD 208,713; 95% CI 27,095–390,330), and risk of death (aOR 17.17; 95% CI 5.88–50.07) in the AP-CE group. Secondary outcomes showed patients with AP-CE had a significantly increased risk of serious complications, particularly cardiac arrest (aOR 64.24; 95% CI 24.27–170.02), and higher hospital resource utilization.
Conclusion
Patients with AP who develop CE face worse outcomes, including increased mortality risk, prolonged hospital stay, and greater resource utilization. Timely identification and effective management of CE in AP may reduce mortality and ease the health care burden associated with this neurological complication.
Keywords
Introduction
Acute pancreatitis (AP) is a serious health concern that can lead to fatal complications. AP is emerging as a primary cause of gastrointestinal-related hospital admissions in the United States. The current incidence of AP is approximately 40 cases per 100,000 individuals annually, contributing to over 130,000 new cases each year, with an upward trend.1,2 The spectrum of complications stemming from AP is broad, encompassing severe conditions such as pancreatic necrosis, infection, and progression to chronic pancreatitis. Managing AP complications requires a multifaceted approach, primarily involving supportive care, including fluid replenishment and effective pain management. Among the array of serious complications associated with AP, involvement of the central nervous system (CNS) in the form of cerebral edema (CE) is a less frequently observed but critical issue that can potentially escalate to pancreatic encephalopathy.3,4 Notably, pancreatic encephalopathy, often detected in the early stages of AP, is linked to a high mortality rate of 57%, highlighting the severe implications of this complication. 4 This neurological condition manifests through a spectrum of symptoms, including disorientation, agitation, and delayed response times, indicating the potentially profound systemic impact of AP.
The expanding understanding of AP underscores the importance of addressing its numerous complications, particularly identifying risk factors and comorbidities that may facilitate the progression to AP with multisystem organ failure—most notably CE. Although individual case studies have contributed to our understanding of how patients with AP may present with CE, a conspicuous gap remains in large-scale research concerning the prevalence of CE within the context of AP. 5 By delineating the risk factors and comorbidities predisposing patients with AP to CE, important strides can be made toward enhancing early detection and developing effective interventions. Given that early-stage management of AP has been shown to reduce mortality rates, 6 a deeper understanding and timely identification of patients with AP and CE could be crucial in refining clinical management strategies, thereby improving health outcomes for those affected. To begin to fill this knowledge gap, our aim was to explore the relationship between AP and CE. Therefore, we performed a retrospective cohort study using data from an established national database to determine patient and clinical factors associated with progression to CE in patients with AP. We evaluated the mortality risk, hospital length of stay, and overall hospitalization cost among patients with this severe complication. We also characterized the risk of serious comorbid complications and health resource utilization associated with this rare but severe occurrence.
Methods
This was a retrospective cohort study of patients who developed AP between January 2019 and December 2020 and were included in the National Inpatient Sample (NIS) database. The NIS database includes broad coverage of inpatient hospital stays throughout the United States and provides a solid foundation for a nationwide examination of outcomes in patients with AP.
Patients were identified with specific International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) codes for AP and CE. We excluded ICD-10 codes for traumatic CE and other specific causes unrelated to pancreatitis. After this exclusion, we included ICD-10 codes for CE, leaving a very well-defined cohort of strictly CE cases. Notably, there are no specific ICD-10 codes for CE directly caused by pancreatitis. Similarly, there are no specific codes for pancreatic encephalopathy. Therefore, we excluded ICD-10 codes for encephalopathy from causes unrelated to pancreatitis to ensure a focus on the relevant patient population. Patients were stratified into two groups and compared: those with AP and CE (AP-CE), and those with AP who did not develop CE (AP-only). The primary outcomes were length of hospital stay, length of intensive care unit (ICU) admission, total inpatient costs in USD, and in-hospital mortality rates. Secondary outcomes included comorbid conditions and health care resource use, including acute kidney injury, acute respiratory failure, cardiac arrest, sepsis, length of stay in the ICU, and need for mechanical ventilation.
Ethical considerations
This study was conducted using de-identified data from the National Inpatient Sample (NIS) database. Use of these data does not require full Institutional Review Board (IRB) approval. Therefore, ethical approval and informed consent were not required for this study. We conducted this study with strict adherence to the Helsinki Declaration of 1975, as revised in 2013, ensuring compliance with ethical research principles. 7 We followed the Equator Network guidelines and completed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist to ensure transparency and accuracy in the reporting of observational studies. 8
Statistical analysis
We performed both univariate and multivariate analyses to clarify the relationship between AP and CE. Categorical data are presented as number and percentage, and continuous variables are described as mean and standard deviation. To compare categorical variables between groups, we used either a Pearson chi-square test or Fisher exact test, as appropriate. All continuous variables described as mean ± standard deviation conformed to a normal distribution; this assumption was verified before the analysis. Continuous variables were analyzed with the Student’s t-test. A p-value of <0.05 was the threshold for statistical significance, and all tests were two-sided. Odds ratios (ORs) were calculated with univariate modeling and adjusted analyses yielded odds ratios (aOR) with 95% confidence intervals (CI) for binary outcomes as well as aORs for the mean differences of continuous outcomes. Multivariate analyses were adjusted for age, sex, White race and ethnicity, median income in the patient’s zip code, Charlson comorbidity index, urban versus rural hospital location, hospital region, hospital teaching status, hospital size according to number of beds, type 2 diabetes mellitus (DM), hypertension (HTN), chronic kidney disease, and end-stage renal disease. All analyses were done using Stata 17 software (StataCorp LLC, College Station, TX, USA).
Results
Patient characteristics
Within the NIS database during the study period, 543,464 patients were hospitalized with a diagnosis of AP. Of these, 220 (0.04%) were diagnosed with AP-CE during hospitalization and 543,244 (99%) were diagnosed with AP-only. Although a slightly higher proportion of patients with AP-CE were female (54.6%) and a slightly higher proportion in the AP-only group were male (54.2%), sex distribution did not significantly differ between groups. Both groups had similarly ages, predominantly between 46 and 64 years old, with a mean age of 51.2 years for those with AP-CE and 50.9 years for patients with AP and no CE (Table 1).
Demographic, clinical, and hospital characteristics of patients with acute pancreatitis.

Data are shown as mean ± standard deviation (SD) for continuous variables and as n (percentage) for categorical variables. Statistical comparisons include p-values for differences between groups.
Median household income in USD national quartiles for patients’ zip codes.
AP, acute pancreatitis; CE, cerebral edema.
For the entire cohort, most patients were White, had private insurance or Medicare, were discharged to home, and were treated in an urban regional hospital. Interestingly, a significant difference was observed in the Charlson comorbidity index between groups; patients without CE were more likely to have a comorbidity index of 0, but patients with AP-CE predominantly had scores ≥3 (p < 0.001). Regarding comorbidities in patients without CE, 45.2% had hypertension, 24.9% had type 2 DM, 27.0% had hyperlipidemia, and 35.5% had fluid and electrolyte disorders. In contrast, patients with AP-CE had higher rates of hyperlipidemia (29.6%) and notably higher rates of fluid and electrolyte disorders (61.4%), but they had lower rates of type 2 DM (15.9%) and HTN (34.1%). Comorbidities were not significantly different between groups.
Primary outcomes
After adjusting for confounding variables, multivariate linear regression analysis showed a significantly longer mean hospital stay for patients with AP-CE compared with those who did not have CE, with a difference of 10.1 days (95% CI, 0.50–19.70; p = 0.03). Evaluation of hospital charges showed that adjusted hospitalization expenses were significantly higher for patients with AP-CE than for those without CE, indicating increased resource utilization with CE (aOR, USD 208,713; 95% CI, 27,095–390,330; p = 0.024). Both univariate and multivariate analyses showed that patients with AP-CE had much higher odds of mortality than patients without CE (OR 43.86; 95% CI, 20.92–91.93; p < 0.001 and aOR, 17.17; 95% CI, 5.88–50.07, respectively; p < 0.001) (Table 2).
Risk of primary outcomes in patients with acute pancreatitis who developed cerebral edema.

Reference was patients with acute pancreatitis who did not develop cerebral edema.
aOR, adjusted odds ratio; CI, confidence interval.
Secondary outcomes
Patients with AP-CE were at higher risk for a range of complications than patients who did not develop CE. This included the risk of acute kidney injury (aOR, 3.21; 95% CI, 1.33–7.76; p = 0.009), acute respiratory failure (aOR, 7.96; 95% CI 3.50–18.08; p < 0.001); cardiac arrest (aOR, 64.24; 95% CI, 24.27–170.02; p < 0.001), and sepsis (aOR, 7.98; 95% CI, 3.08–20.66; p < 0.001). Similarly, the mean ICU stay was longer for patients with AP-CE (aOR 10.78; 95% CI, 4.62–25.16; p < 0.001), and patients with AP-CE had a higher risk of requiring mechanical ventilation (aOR, 12.58; 95% CI, 5.36–29.52; p < 0.001) (Table 3). Primary and secondary outcomes are illustrated in the form of forest plots in Figure 1.
Risk of secondary complications in patients with acute pancreatitis who developed cerebral edema.

Reference was patients with acute pancreatitis who did not develop cerebral edema.
aOR, adjusted odds ratio; CI, confidence interval; ICU, intensive care unit.
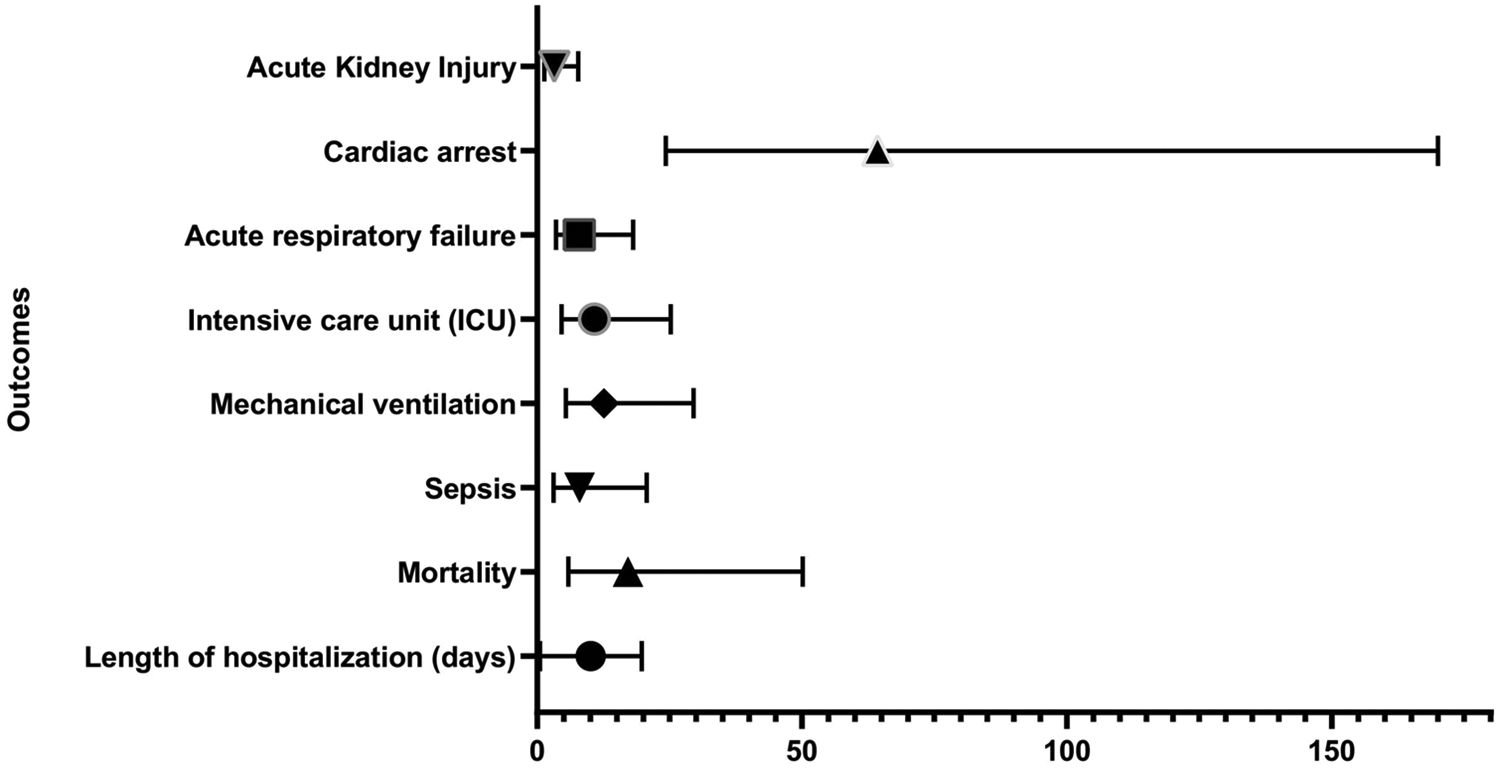
Forest plot illustrating primary and secondary outcomes in patients with acute pancreatitis-related cerebral edema (AP-CE). The plot shows odds ratios and confidence intervals for key outcomes and complications, highlighting significant differences between patients with AP-CE and those without.
Discussion
In this study, a very small proportion (0.04%) of patients hospitalized for AP developed a serious complication of CE. However, despite the low prevalence of CE within the context of AP, those patients who developed CE appeared to be at significantly higher risk of death, longer hospital stay, and several serious complications, including kidney injury, respiratory failure, sepsis, and cardiac arrest in particular. Despite the varied demographics in the entire cohort, patients across both categories were predominantly White and had private insurance or Medicare, indicating that these demographic factors might not significantly sway the likelihood of developing CE within the context of AP. Also, the tendency for patients with AP and CE to be discharged home might reflect more accessible health care resources for this population, and patients who developed CE unsurprisingly required more hospital resources.
In the realm of severe AP complications, CNS involvement is rare but profoundly serious, affecting approximately 10% of patients and potentially leading to diffuse CE and pancreatic encephalopathy.3,9 The pathogenesis of CNS involvement is complex, and the activation of phospholipase A (PLA) is often implicated in this process, specifically PLA2, which plays a critical role in increasing vascular permeability and triggering cerebral capillary thrombosis, subsequently contributing to CE. 10 By compromising the structural integrity of brain cell membranes, the activity of PLA2 exacerbates vascular permeability, promoting the resultant edema. This adverse sequence is aggravated by the excessive release of cytokines during AP, culminating in tissue injury. 11 Furthermore, the production and activity of platelet-activating factor, which enhances platelet aggregation and release, amplify the risk of cerebral capillary thrombosis and subsequent hypoxemia. This intricate cascade of events disrupts cerebral cell metabolism, thereby worsening CE. Studies have highlighted that patients with severe AP manifestations display elevated levels of pro-inflammatory cytokines, including tumor necrosis factor-alpha, interleukin-1 beta (IL-1β), IL-6, and IL-8, which significantly intensify the disease state. 5
In the early phases of severe AP, diffuse CE frequently emerges as part of a multiorgan failure syndrome, leading to pancreatic encephalopathy. As the disease progresses, pancreatic encephalopathy, characterized by confusion and psychomotor agitation, among other symptoms, becomes more evident. Autopsy and magnetic resonance imaging studies have confirmed the presence of diffuse demyelination, CE, neuronal loss, and hemorrhagic cerebrovascular incidents in this condition, underlining the critical nature of this complication. 3 The alarming 50% mortality rate associated with these complications underscores the urgent need for precise identification of causative factors.9,12 Recent findings also suggest that disturbances in consciousness, such as confusion, delirium, and convulsions, can manifest in all forms of AP, irrespective of the cause. 13
Analyzing comorbid conditions within our patient cohort revealed that individuals without CE typically had fewer concurrent health issues, as indicated by a lower Charlson comorbidity index. Conversely, those with CE faced a more complex health profile, hinting at a more challenging clinical trajectory and potentially impacting therapeutic outcomes. Our study indicates that complications such as respiratory insufficiency and hypoxemia, secondary to serious AP, can induce abnormal cerebral metabolism, potentially leading to CE. 14 We found that patients with AP-CE experienced higher rates of acute kidney injury and especially cardiac arrest, along with increased odds of sepsis, highlighting a more severe systemic inflammatory response. In particular, the aOR for cardiac arrest in this group was noted to be strikingly high at 64.24 in patients with AP-CE and vastly exceeded the risks associated with other complications. Studies show that severe pancreatitis can result in myocardial injury, even without obstructive coronary artery disease being a risk factor. 15 Although acute coronary syndrome (ACS) may be mimicked in these instances, actual ST segment elevation myocardial infarction (STEMI)-like presentations in AP are exceedingly uncommon. Electrocardiogram (ECG) changes, specifically in the T-wave and ST segment, occur in approximately 25% of AP cases. 16 The infrequency of STEMI presentation poses a substantial clinical challenge: treating these ECG changes as ACS could lead to unnecessary administration of thrombolytics or invasive cardiac procedures without the presence of coronary disease. Conversely, interpreting these changes as pancreatic rather than cardiac in origin risks overlooking genuine ACS, with potentially fatal outcomes.
Previous studies have underscored the higher prevalence of acute kidney injury (AKI) in patients with AP and its associated poor outcomes, but these studies were constrained by a small sample size and retrospective nature.17,18 However, Devani et al. conducted a retrospective observational analysis using the NIS database, revealing an AKI prevalence of 7.9% among 3,466,493 individuals hospitalized for AP. Notably, the mortality rate in patients with AKI was significantly elevated (8.8% in the AKI group compared with 0.7% in those without AKI; p < 0.01). 19 Furthermore, patients with CE had a significantly longer hospital stay and ICU admission, necessitating mechanical intubation and involving a high incidence of acute respiratory failure. The data indicated that the odds of experiencing acute respiratory failure in this group were nearly eightfold (aOR, 7.96) compared with those who did not have CE, necessitating a more intensive level of care, as evidenced by the prolonged duration of ICU stays (aOR, 10.78) and the increased likelihood of requiring mechanical ventilation (aOR, 12.58). Gajendran et al. estimated that approximately 5.4% of patients with AP were concurrently diagnosed with acute respiratory failure (ARF); among these, the mortality rate was notably high at 26.5%. The authors identified critical predictors for the development of ARF in the context of AP, namely, sepsis, pleural effusion, pneumonia, and cardiogenic shock. 20 This situation points to the severe and complex nature of this condition, emphasizing the increased health care burden presented by this complication. Additionally, the observed higher mortality rate in patients with AP-CE accentuates the critical need for prompt recognition and proactive management of CE in individuals with AP. Also, our findings related to resource utilization, particularly the significantly higher costs associated with patients who develop CE, offer new perspectives on the health care system burden. Previous studies have rarely focused on this aspect, making our analysis critical for health care policy makers and hospital administrators to design more cost-effective interventions.
Preventing CE in the context of AP requires early identification and management of high-risk patients. Timely recognition of severe AP cases, particularly those with systemic inflammatory responses and multi-organ involvement, is critical to mitigating the development of CE. Proactive monitoring of neurological status, along with the management of risk factors such as electrolyte imbalances, hyperlipidemia, and hypoxemia, may help prevent CE. Ensuring that patients with AP receive adequate fluid resuscitation, as well as early intervention for acute respiratory failure and sepsis, can reduce the overall systemic burden and potentially lower the risk of neurological complications like CE.
Our study has several limitations that should be acknowledged. First, as a retrospective analysis, we relied on the accuracy of ICD-10 coding, which may introduce misclassification or undercoding biases. The absence of corresponding laboratory test data and critical clinical details—such as patient symptoms, measured systolic function levels, prescribed medications, and functional capacity—in the NIS database further constrained our analysis. Additionally, the NIS records hospitalizations per admission rather than per individual, potentially leading to duplicate records if a patient is admitted multiple times. This method also limits our ability to analyze secondary diagnoses in relation to specific admissions. Moreover, although we focused on AP and CE, some cases of CE may be secondary to other neurological conditions. The observational nature of this study precludes establishing causality; our findings must therefore be interpreted with caution. Detailed insights into specific treatment strategies could not be confirmed; prospective validation is needed to build on our findings and improve their generalizability.
Conclusion
The increased hospitalization costs for patients with AP-CE mirror their greater resource utilization necessitated by longer hospital stays, intensive treatments, and management of intricate complications. This financial impact underlines the urgent need for effective preventive and management strategies within health care systems to mitigate the economic strain owing to AP-CE. Our findings corroborate as well as extend the current understanding of AP and its rare, yet severe, complication of CE, underscoring the importance of heightened awareness and early intervention to curtail morbidity, mortality, and health care costs. By enriching the knowledge base surrounding AP and CE, our study findings suggest a need for increased clinical vigilance and strategic therapeutic approaches to manage this complex complication. The insights obtained pave the way for further research into the etiological mechanisms involved and the development of targeted treatments, with the potential to improve patient outcomes and reduce health care resource demands owing to AP and its neurological sequelae.
Footnotes
Acknowledgements
The authors would like to extend special thanks to Karla D. Passalacqua for her invaluable contributions and insights. We also thank our colleagues and staff at their respective institutions for their support during this study.
Authors’ contributions
Conception and design: MS, AJC, SJ, AK; Analysis and interpretation of the data: SJ, MS, GT, VM, MAK, MSF; Drafting of the article: AJC, MS, MSF, MAK, VM, GT; Critical revision of the article: AJC, MS, MAK, MSF, SJ, AK.
Data availability
The data that support the findings of this study are available from the National Inpatient Sample (NIS) database. Restrictions apply to the availability of these data, which were used under license for this study. Data are available from the authors upon reasonable request and with permission from the NIS database. For further inquiries or access to specific data points, readers are encouraged to contact the corresponding author.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
