Abstract
The popliteal artery segment is particularly challenging for endovascular treatment. Stents used for treating popliteal artery lesions are usually associated with an increased risk of stent fracture and re-occlusion. The Supera stent is designed to withstand mechanical stress, with a low risk of fracture. A man in his mid-70s with left popliteal artery occlusion was treated with a Supera stent implantation. After 5 months, he revisited our hospital complaining of left leg pain. Preprocedural computed tomography angiography showed popliteal artery occlusion associated with a Supera stent fracture. A new Supera stent was implanted at the previous stent fracture site. However, 3 months after the second procedure, computed tomography angiography showed popliteal artery re-occlusion associated with a repeated fracture of the Supera stent. A femoral-popliteal bypass was performed. At the 6-month follow-up, computed tomography angiography showed a patent graft. Our findings indicated that recurrent fractures occurred after additional Supera stenting at the previous Supera stent fracture site. Careful observation is necessary after deploying a new Supera stent for treating a Supera stent fracture because of the possibility of a repeated fracture.
Keywords
Introduction
The popliteal artery, which is situated posterior to the knee joint in the popliteal fossa, branches from the superficial femoral artery into the following three segments: P1 (suprapatellar region), P2 (from the patella to the tibial plateau), and P3 (from the tibial plateau to the anterior tibial artery origin). The popliteal artery then divides into the anterior tibial artery and tibioperoneal trunk.1,2 A popliteal artery lesion is exposed to complex biomechanical forces resulting from repetitive knee joint flexion, which is a particularly challenging segment for endovascular treatment. 3
Stents in the popliteal segment have traditionally been associated with increased risks of stent fracture and re-occlusion. Furthermore, previous studies have shown that stent placement specifically within the P2 and P3 segments of the popliteal artery is associated with a high incidence of stent fractures and correlated with an increased rate of restenosis.4,5 The Supera stent (Abbott Vascular, Santa Clara, CA, United States), which is a self-expanding interwoven nitinol stent, is designed to withstand mechanical stress along the popliteal segment and has been reported to have satisfactory results.6–8 A systematic review conducted by Salamaga et al. 9 specifically evaluated the performance of the Supera stent in patients with isolated popliteal artery disease. They found that the 12-month primary patency rate ranged between 68% and 90%, without any fractures. However, previous reports have shown that Supera stent fracture can occur.10–14 Treatment options for popliteal artery occlusion secondary to Supera stent fracture are not well established. Only a limited number of case reports have described the use of various treatments, which included balloon angioplasty alone, bypass surgery, and additional Supera stenting.10–14 We present a case of repeated Supera stent fracture following stenting for the treatment of popliteal artery occlusion.
Case report
A man in his mid-70s presented to the Emergency Department with gradually worsening numbness (Rutherford category IIa) in his left leg for 2 weeks. He had a history of 18 years of hypertension and 50 years of smoking. During a physical examination, the patient's left foot appeared pale and his dorsalis pedis pulse was not palpable. Although there was sensory loss in the left toes, no motor deficits were observed in the left foot. Preoperative computed tomography angiography (CTA) showed a left popliteal artery occlusion (Figure 1(a)). Cardiovascular assessments, including electrocardiography and echocardiography, showed no major abnormalities. Further investigations also identified no potential embolic sources, such as thoracoabdominal aneurysms. Based on the symptom onset within 2 weeks and the findings of the physical examination, there was a strong suspicion of acute limb ischemia, necessitating prompt revascularization.

Endovascular procedure for the treatment of a popliteal artery occlusion, with implantation of a Supera stent. (a) Preoperative computed tomography angiography. (b) Angiography shows occlusion of the popliteal artery. (c) Arteriogram after mechanical thrombectomy using a Rotarex device and (d) recanalization of the popliteal artery after the deployment of a Supera stent.
Endovascular treatment is the preferred option for managing patients showing Rutherford classes I or IIa. 15 Consequently, the patient received his first endovascular treatment at our institution on the day of his visit. At that time, his left popliteal artery thrombosis (P1 and P2 segments) was treated with mechanical thrombectomy using a 6-French (6 F) Rotarex catheter (Straub Medical, Wangs, Switzerland) and implantation of a 6.5- × 100-mm Supera stent (Figure 1). The selection of a 6.5-mm diameter for the Supera stent was based on preoperative CTA measurements, which showed that the diameters of the healthy proximal and distal segments adjacent to the lesion ranged between 6 and 6.4 mm. After mechanical thrombectomy, residual thrombosis remained in the popliteal artery, and it required additional intervention. Initial attempts with endovascular aspiration thrombectomy were made multiple times, but showed little change in the residual thrombosis. Therefore, surgical embolectomy or thrombolytic treatment was considered. However, open surgery was impossible because of the presence of other patients requiring emergency interventions. Additionally, our hospital’s protocol mandated that thrombolytic treatment with recombinant tissue plasminogen activator, which is the only thrombolytic agent available in Korea, should be performed within the intensive care unit. Supera stenting was performed as an alternative for treating the remaining thrombosis because of a lack of available intensive care unit beds and the unfeasibility of open surgery. After the procedure, the patient’s symptoms improved, and the ankle–brachial index (ABI) was 1. Subsequently, the patient was treated with antithrombotic therapy (cilostazol and aspirin). CTA 1 week after the procedure showed patent popliteal artery flow and no stent fracture.
At a 3-month follow-up, a stent fracture was detected on CTA. Although in-stent restenosis was also observed, the ABI was 0.9. The patient was asymptomatic. He revisited our hospital reporting left leg pain (Rutherford category 3) at 5 months after the stent implantation. CTA showed an occlusion of the distal superficial femoral artery and popliteal artery associated with the stent fracture (Figure 2(a) and (b)). Endovascular revascularization was performed by interventional radiologists. Using the ipsilateral approach, a 7 F introducer was placed in the left common femoral artery under local anesthesia. Femoropopliteal artery angiography showed an occlusion of the distal superficial femoral artery and popliteal artery associated with the stent fracture (Figure 2(c) and (d)). The occluded lesion, including the fractured Supera stent, was passed using a 0.035-inch hydrophilic guidewire (Radifocus M; Terumo Medical Corporation, Tokyo, Japan) supported by a 5 F Rubicon catheter (Boston Scientific, Marlborough, MA, United States). Mechanical thrombectomy of the lesion was performed using a 6 F Rotarex device. After mechanical thrombectomy, a new 6.5 × 150-mm Supera stent was implanted (Figure 2(e)). The decision for using a 6.5-mm diameter for the second Supera stent was based on the need to position it within the previously implanted Supera stent. Sequential predilation of the lesion was performed using an Admiral Xtreme 6 × 40-mm balloon catheter (Medtronic, Minneapolis, MN, United States) before the Supera stent re-implantation. A completion angiogram showed good patency of the stent (Figure 2(f)). The patient’s leg pain improved, and the ABI was 1 after the intervention. After this second intervention, the patient continued antithrombotic therapy using cilostazol and aspirin.

Popliteal artery re-occlusion associated with a fracture of the Supera stent. (a) An axial image of computed tomography angiography shows left popliteal artery occlusion and a Supera stent fracture. (b) A maximum intensity projection image of computed tomography angiography shows popliteal artery thrombosis and a Supera stent fracture (arrows). (c) Anteroposterior radiogram of the Supera stent immediately after implantation (between the white arrows) and a radiographic image of the same stent at 5 months after the procedure (between the black arrows). A radiogram of the Supera stent taken immediately after implantation shows moderate elongation (white arrowhead) of the stent segment within the P2 segment of the popliteal artery. The 5-month follow-up radiogram shows a fracture of the Supera stent (black arrowhead). (d) Anteroposterior projection diagnostic angiogram. (e) Deployment of a new Supera stent for popliteal artery re-occlusion and (f) final angiogram after the implantation of a new Supera stent.
It should be noted that left leg claudication (Rutherford category 3) recurred at 3 months after the second procedure. CTA showed popliteal artery re-occlusion associated with a repeated fracture of the Supera stent (Figure 3(a) and (b)). Radiographs showed stent fracture at the previous fracture site (Figure 3(c) and (d)). A femoral–popliteal bypass was performed using an autologous greater saphenous vein graft by a vascular surgeon. Following the surgery, the patient maintained antithrombotic therapy administration, which comprised cilostazol and aspirin, to prevent thrombotic complications. The patient’s symptoms improved after the operation. At a 6-month follow-up, the patient was asymptomatic and CTA showed a patent graft (Figure 3(e)).
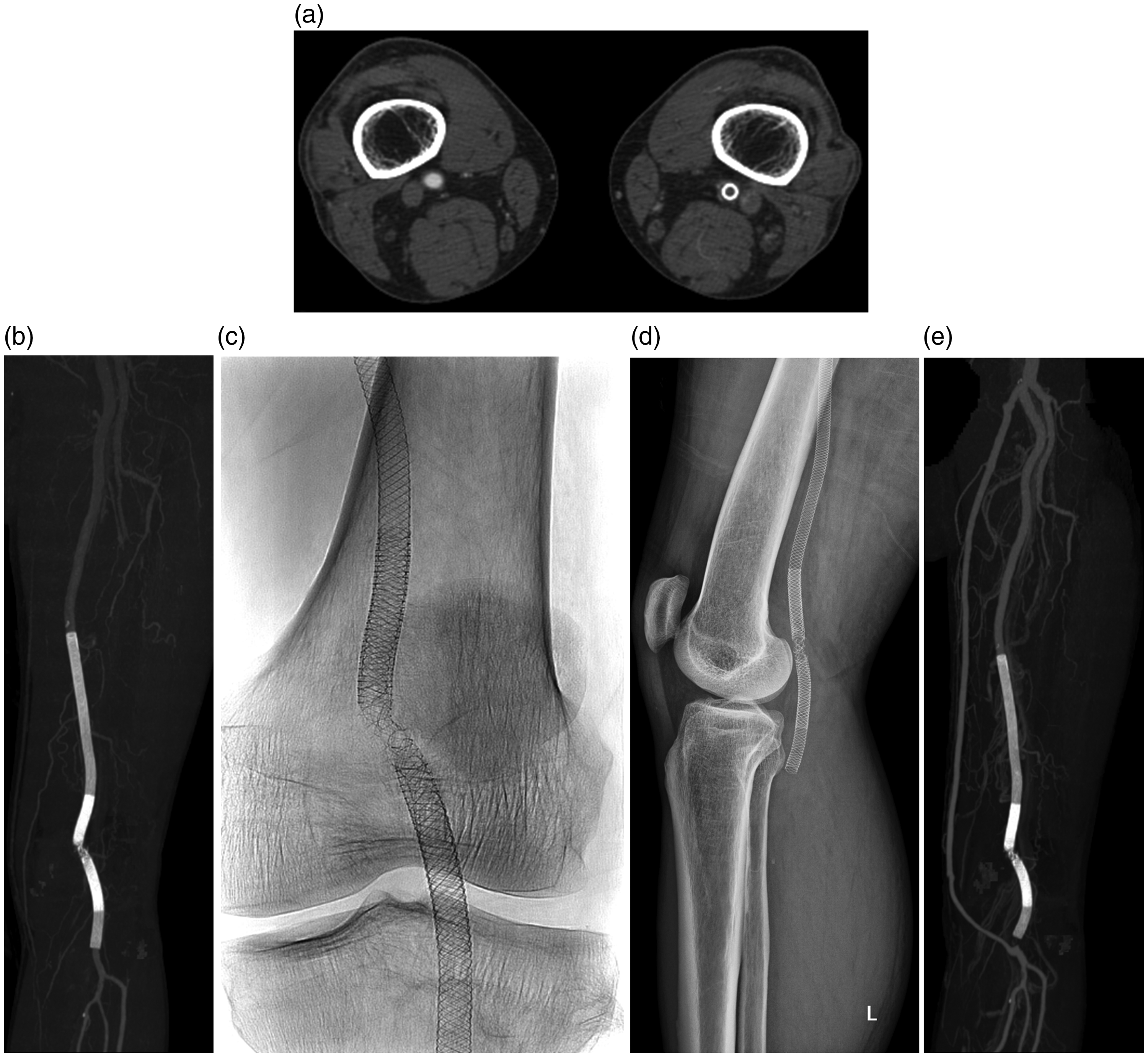
Repeated Supera stent fracture and re-occlusion of the popliteal artery. (a) A computed tomography angiography image shows left popliteal artery occlusion. (b) A repeated fracture of the Supera stent can be seen on a maximum intensity projection image of computed tomography angiography. (c) An anteroposterior radiograph of the left knee shows repeated stent fracture at the previous fracture site. (d) Lateral radiograph of the left knee and (e) computed tomography angiography image after bypass surgery.
The reporting of this study conforms to the CARE guidelines. 16 Informed consent was obtained from the patient for all treatment procedures, including endovascular treatments and surgery. Additionally, we have de-identified all patient information and obtained written consent for the publication of his anonymized data. The necessity for approval from an Ethics Committee or Institutional Review Board was exempted owing to the classification of this study as a case report.
Discussion
Popliteal artery stenting has historically been performed for highly selected cases because of the unacceptable rate of stent fracture. Two-year patency of popliteal artery stenting was reported in 64.2% of cases, with a 4.6% fracture rate before the introduction of the interwoven nitinol stent. 17 However, the Supera stent, which has a design that resists stressors and fractures, had a stent fracture rate of 0.6% (only 1 case of fracture) in the 3-year follow-up in the SUPERB trial. 8
In addition to the single case of fracture reported in the SUPERB trial, 8 a few other cases of Supera stent fractures have been reported.10–14 A recent study, which investigated the 5-year, real-world outcomes of Supera stent implantations in below-the-knee prosthetic bypasses, also reported a case of Supera stent fracture. 18 Various treatment methods for Supera stent fractures have been documented in previously published case studies. Balloon angioplasty alone was performed in one case. 10 Another three cases were treated with bypass surgery.11,13,14 San Norberto et al. 12 performed additional Supera stenting within a fractured Supera stent, as in our case, and they reported good results in a 6-month follow-up imaging examination. However, in our case, fracture of the new Supera stent was observed at 3 months after the second intervention, and bypass surgery was performed.
The exact reason for the initial popliteal artery occlusion in our patient could not be determined. There was no evidence of popliteal artery entrapment syndrome on a preoperative CTA. Possible sources of embolism also could not be found. However, despite the absence of identifiable embolic sources based on the investigations, the possibility of arterial embolism as a cause of the popliteal artery occlusion could not be completely ruled out. The Supera stent fracture was first detected on CTA at 3 months after the first intervention, and in-stent restenosis was also observed. The popliteal artery occlusion was first detected on CTA at 5 months after the Supera stent implantation. In our case, during the first endovascular treatment, a Supera stent that was excessively long for the relatively short lesion of the popliteal artery was used. At that time, the shortest available Supera stent length at our institution was 100 mm, which resulted in the stent involving the P2 and P3 segments of the popliteal artery. Although previous reports did not specifically study Supera stents, they suggested that stent placement specifically within the P2 and P3 segments of the popliteal artery is associated with a high incidence of stent fractures and an increased rate of restenosis.4,5 Therefore, the involvement of the P2 and P3 segments in Supera stenting may have contributed to the fracture of this stent and subsequent popliteal artery re-occlusion in our patient. Moreover, moderate elongation (Figure 2(c)) of the Supera stent segment located in the P2 segment of the popliteal artery was observed on a radiogram immediately after the first Supera stent implantation. Additionally, subsequent examinations showed a stent fracture at this location. The radial force at the elongated segment of the Supera stent was reduced, and the accompanying high mechanical stress may have led to the stent fracture.
At our institution, the protocol for antithrombotic therapy following femoropopliteal artery stenting includes the administration of dual antiplatelet therapy with clopidogrel for 1 month to prevent acute stent occlusion. However, dual antiplatelet therapy with clopidogrel was not administered to this patient for unspecified reasons, which might have contributed to the popliteal artery re-occlusion. Additionally, a possible cause of the repetitive Supera stent fracture in our case could be related to the occupation of our patient as a gardener, which involved excessive and repeated bending of the knees while working. During gardening, the popliteal artery is exposed to strong mechanical stresses associated with considerable torsion during knee flexion. At our institution, we routinely assess patients' occupations and daily activities that might involve frequent knee bending before femoropopliteal artery stenting. Before the initial endovascular treatment, the patient declined to disclose his occupation and reported engaging in minimal activities that required knee bending, which influenced the decision to proceed with the implantation of a Supera stent in the popliteal artery. However, after the detection of a fracture in the previously implanted Supera stent, the patient disclosed during the re-evaluation process that he worked as a gardener, which is a role that required extensive bending of the knees. We informed the patient that a Supera stent fracture was a rare complication and the treatment options for popliteal artery occlusion secondary to Supera stent fracture were not well established. We further explained to the patient that the available treatment methods for popliteal artery occlusion secondary to Supera stent fracture, as reported in previous case reports, might include endovascular treatment options such as balloon angioplasty or additional Supera stenting, as well as bypass surgery.10–14 Consequently, we recommended bypass surgery as the primary treatment option for popliteal artery re-occlusion associated with the Supera stent fracture because of his occupational risks and the potential for repeated Supera stent fractures. However, the patient expressed a strong preference for undergoing an additional endovascular treatment. Therefore, we proceeded with a second endovascular treatment involving additional Supera stenting. Despite advising the patient to minimize knee bending post-treatment, he continued gardening.
A surveillance protocol has been proposed for infrainguinal arterial stents, suggesting that stent fractures can be categorized into the following five types: type I, single strut fracture; type II, multiple strut fractures; type III, fracture with preserved alignment of the stent components; type IV, stent fracture with misalignment of the components; and type V, trans-axial spiral dissection of the stent. 19 In our case, additional Supera stenting was performed on a type V stent fracture. However, in a case reported by San Norberto et al, 12 new Supera stenting was performed on a type III stent fracture. We speculate that this difference in fracture types observed in the pre-existing Supera stent, prior to additional stenting, might have contributed to the negative outcomes in our case. This finding is in contrast with the maintained patency reported in the case reported by San Norberto et al. 12
Conclusion
To the best of our knowledge, few reports have described recurring stent fractures after additional Supera stenting for the treatment of a Supera stent fracture. The Supera stent is designed to withstand mechanical stress and has a low risk of fracture. However, Supera stent fractures may reoccur when the stent is deployed at a previous stent fracture site, such as in our case. In addition, careful observation is necessary after deployment of a new Supera stent for treatment of a Supera stent fracture because of the possibility of a repeated fracture.
Footnotes
Acknowledgements
None.
Author contributions
HOK and HKL contributed to conceptualization and data curation. BCL and CP reviewed the literature and interpreted the imaging findings. HOK supervised the study. BCL and HOK wrote the original draft. CP and HOK reviewed and edited the manuscript.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
