Abstract
Objective
To evaluate the relationship between nutritional scoring systems, nutritional support methods, and the prognosis of severe and critically ill patients infected with the Omicron variant of coronavirus disease 2019 (COVID-19).
Methods
Patients with confirmed Omicron variant severe and critical COVID-19, who were admitted to Chongqing Medical University First Hospital between December 2022 and January 2023, were enrolled into this retrospective study. Clinical data of patients who survived for 28 days were compared with those who died during the same period. Nutritional status was assessed using the 2002 Nutrition Risk Screening (NRS) tool and Prognostic Nutritional Index (PNI). Factors influencing patient mortality were identified by multivariate logistic regression, and the relationship between patient nutrition and mortality as the disease progressed was illustrated using Kaplan–Meier curves. The study was registered on the ChiCTR platform (No. ChiCTR2300067595).
Results
A total of 508 patients were included (349 survivors and 159 non-survivors). Significant differences were found in sex, age, NRS score, PNI score, albumin level, lymphocyte count, chronic comorbidities, mechanical ventilation, neutrophil count, procalcitonin, and platelet count between survivors and non-survivors. Multivariate analysis revealed that high NRS score (OR 3.87, 95% CI, 1.97, 7.63), fourth-level nutritional support (combined enteral and parenteral nutrition; OR 7.89, 95% CI, 1.32, 47.28), chronic comorbidities (OR 4.03, 95% CI, 1.91, 8.51), and mechanical ventilation (OR 6.03, 95% CI, 3, 12.13) were risk factors for mortality (OR > 1). The malnutrition rate among patients with NRS ≥ 3 was 41.93%. The median (interquartile range) PNI score was 38.20 (35.65, 41.25) for survivors versus 32.65 (29.65, 36.58) for non-survivors. The mortality rate was higher in patients with high nutritional risk within 28 days of hospitalization. The descending order for mortality rate in patients receiving different nutritional support was: dual parenteral and enteral nutrition, no nutritional intervention, single enteral nutrition, and single parenteral nutrition.
Conclusion
A high proportion of severe and critically ill patients with COVID-19 experience malnutrition, and various factors are associated with their prognosis. High nutritional risk is significantly related to patient mortality. Early assessment using NRS or PNI is crucial for these patients, and personalized interventions should be implemented to improve overall nutritional status, maintain organ function, and enhance the body’s antiviral defence.
Keywords
Introduction
Coronavirus 2019 (COVID-19) is an acute inflammatory disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), including the Omicron variant, and may manifest as a mild, moderate, severe, or critical disease. Severe and critical cases often involve refractory respiratory failure, shock, and multi-organ failure, posing life-threatening risks. 1 With over a billion infections globally over 3 years, the pandemic has shown seasonal and local transmission patterns. In densely populated China, a large number of severe and critical cases of respiratory and critical care-related illnesses are admitted annually. 2
Previous studies have shown that cytokines (interleukin [IL]-1β, IL-6, tumour necrosis factor [TNF]-α and others) reduce appetite, food motivation and energy intake by acting on the hypothalamus. Specific inflammatory cytokines, such as TNF and IL-10, are involved in the regulation of taste perception.3,4 Thus, severe inflammation may lead to reduced appetite and increased body demand, compromising overall nutritional status. 5 Malnutrition, in turn, exacerbates systemic inflammation, causing multi-organ failure, infections, prolonged hospital stays, or even death. 6 The interplay between systemic inflammation, high metabolic state, and cytokine storm may worsen protein breakdown, deepening malnutrition. This creates a vicious cycle of inflammation, malnutrition, cytokine storm, worsening malnutrition and inflammation, and disease progression or death. 7 Research has shown that malnutrition is prevalent among patients with COVID-19 and is considered a potential risk factor for severe outcomes. 8
Upon hospitalization, The First Affiliated Hospital of Chongqing Medical University employs the 2002 Nutrition Risk Screening (NRS-2002) to assess nutritional risk in patients with COVID-19, 9 and dynamically reassess their condition throughout their stay. Based on these assessments, appropriate nutritional support is provided. However, there is no gold standard for measuring nutritional supplementation, with clinical assessments often relying on albumin levels as a nutritional indicator and lymphocyte counts for immune status. 10 The Prognostic Nutritional Index (PNI), which combines albumin concentration and total lymphocyte count, is recommended for evaluating immune-nutritional status. 11 Through a retrospective analysis of hospitalized patients, the aim of the present study was to explore the relationship between nutrition, nutritional support, and disease prognosis, in order to identify patterns and potentially uncover a path for managing inflammation and nutritional status in patients with COVID-19.
Patients and methods
Ethics and guidelines
This study was conducted in accordance with the principles of the Helsinki Declaration of 1975, as revised in 2013. As this is a retrospective study, an exemption was obtained from the Ethics Committee of The First Affiliated Hospital of Chongqing Medical University. All patient data were de-identified in accordance with guidelines, and written informed consent was not required due to the retrospective study design. The reporting of this study conforms to STROBE guidelines. 12
Study population
Patients with severe and critical COVID-19 (Omicron variant), who were admitted to Chongqing Medical University First Hospital between December 2022 and January 2023, were enrolled into this retrospective study by convenience sampling (due to the retrospective nature of the study). Patients were recruited consecutively based on their admission to the respiratory and critical care units because of COVID-19 infection. The index date for all patients was defined as the day of hospital admission. Consistent criteria were used across all groups. The survival status of all patients over 28 days following hospital admission was tracked through the hospital’s electronic medical record system. Patients discharged before day 28 were contacted via telephone on the 28th day to confirm their survival status. Inclusion criteria were: (1) aged ≥ 18 years; (2) diagnosed with the Omicron variant through genetic sequencing; and (3) classified as severe or critical cases. Exclusion criteria were: (1) aged <18 years; and (2) asymptomatic or mild cases. A case was defined as severe if the patient met one of the following conditions: (1) shortness of breath with a respiratory rate ≥ 30 breaths per min; (2) oxygen saturation ≤93% while breathing ambient air at rest; (3) arterial oxygen partial pressure (PaO2)/fraction of inspired oxygen (FiO2) ≤ 300 mmHg (1 mmHg = 0.133 kPa); or (4) progressive worsening of clinical symptoms, with imaging showing a significant increase in lung lesions by > 50% within 24–48 h. A case was defined as critical if the patient met one of the following conditions: (1) respiratory failure requiring mechanical ventilation; (2) occurrence of shock; or (3) failure of other organ functions that required intensive care unit monitoring and treatment. 13
Data collection
Nutritional risk was assessed using various tools. Upon admission, patients were screened for nutritional risk by trained healthcare professionals (YQ and XPX) using the NRS-2002 tool. 14 The NRS-2002 consisted of three questions: (1) weight loss of 5% or more (none, within 1 month, within 2 months, or within 3 months); (2) body mass index (BMI) categories (BMI < 16, 16–16.9, 17–18.4, >18.4 kg/m2); and (3) age (< 70 years, ≥ 70 years). A score of 3 or more indicated a risk of malnutrition, requiring further assessment based on test results. 14 The PNI has been validated for its sensitivity and is widely used for assessing nutritional risk in patients. 15 During treatment, PNI scores were calculated using the formula: (10 × serum albumin [g/dL]) + (0.005 × lymphocyte count [mm³]). A PNI score >38, 35–38, or <35 indicated normal, moderate, or severe nutritional risk, respectively. Nutritional support was classified into four categories: (1) regular diet; (2) enteral nutrition (EN); (3) parenteral nutrition (PN); and (4) combined EN + PN, following the American Society for Parenteral and Enteral Nutrition clinical guidelines. 16
Clinical data were collected from patients diagnosed with severe and critical COVID-19 (Omicron variant) based on the National Health Commission’s guidelines. 17 Data included admission date, NRS and PNI scores, presence of chronic diseases (diabetes, or respiratory, cardiovascular, liver, and kidney diseases), all medications (including antivirals, corticosteroids, and antibiotics) that were administered during hospitalization (according to treatment protocols), 12 oxygen therapy, nutritional support type, and survival status after 28 days. Blood samples were collected within 24 h of admission for the evaluation of haematochemical parameters, such as haemoglobin (g/L), albumin (g/L), platelets (×109/L), lymphocyte count (×109/L), neutrophil count (×109/L), C-reactive protein (mg/dL), and procalcitonin (mg/dL).
Patient health risk factors that may exacerbate COVID-19 severity, such as smoking, alcohol consumption, irregular sleep, and stress, were retrieved from medical records. 18 Vaccination history was also considered, as it has been shown to reduce symptoms and shorten the illness duration. 19
Statistical analyses
Patients were divided into two groups based on their 28-day survival: survivors and non-survivors. Normally distributed continuous data are presented as mean ± SD, and between-group comparisons were performed using Student’s t-test. Data that were not normally distributed are presented as median (interquartile range), and were compared using Mann–Whitney U-test. Categorical data are presented as n (%) prevalence and were analysed using χ2-test. Covariates, such as age, sex, presence of chronic comorbidities, and type of nutritional support were included in a multivariate logistic regression model. Adjustments were made to account for these covariates, to isolate the effect of nutritional status on patient outcomes. The association between nutritional risk and nutritional support types with patient mortality over time was depicted using Kaplan–Meier survival curves. Statistical analyses were performed using IBM SPSS Statistics software, version 25.0 (IBM Corp., Armonk, NY, USA), and statistical significance was set at P < 0.05.
Results
A total of 511 patients with severe and critical COVID-19 (Omicron variant) were admitted to Chongqing Medical University First Hospital between December 2022 and January 2023. Of these, 508 patients met the inclusion criteria and were included in the study: 349 patients who survived for 28 days following hospital admission (survivors) and 159 patients who died within the 28-day period (non-survivors).
Demographic and clinical characteristics of survivor and non-survivor groups
The median study population age was 74 years (64.00, 84.00), 330 patients (65%) were male, and the male-to-female ratio was 1.85:1. Comparison of the baseline characteristics showed statistically significant between-group differences in sex, age, NRS score, PNI score, serum albumin level, lymphocyte count, neutrophil count, platelets, level of nutritional support, prognostic inflammatory index (PII), oxygen therapy, combined chronic diseases, mechanical ventilation, corticosteroid therapy, procalcitonin, and positive nucleic acid results (all P ≤ 0.001). However, there were no statistically significant differences between survivors and non-survivors (P > 0.05) regarding smoking history or triple inhalation therapy (inhaled corticosteroid + long-acting β2-agonist + long-acting muscarinic antagonist). The demographic and clinical characteristics of the two patient groups are summarized in Table 1.
Demographic and clinical data of patients hospitalised with severe and critical coronavirus 2019, grouped according to 28-day survival.

Data presented as n (%) prevalence or median (interquartile range).
NRS, 2002 Nutrition Risk Screening; PNI, Prognostic Nutritional Index; PII, Prognostic Inflammatory Index; PCT, procalcitonin; ICS, inhaled corticosteroid; LABA, long-acting β2-agonist; LAMA, long-acting muscarinic antagonist.
NS, no statistically significant between-group difference (P > 0.05; χ2-test).
Multifactorial logistic analysis of risk factors associated with mortality in patients with COVID-19
Data regarding sex, age, NRS score, serum albumin level, PNI score, nutritional support programs, smoking history, and other variables (Table 2) were included in a multivariate logistic regression. Results showed that high NRS score, nutritional support levels 3 and 4, comorbid chronic diseases, mechanical ventilation and PII score were risk factors associated with patient death (odds ratios [ORs] > 1; P < 0.05).
Multifactorial logistic regression analysis of risk factors associated with 28-day mortality in patients hospitalised with severe and critical coronavirus 2019.

CI, confidence interval; NRS, 2002 Nutrition Risk Screening; PNI, Prognostic Nutritional Index; PII, Prognostic Inflammatory Index; PCT, procalcitonin.
NS, no statistically significant correlation (P > 0.05).
Subgroup analyses based on NRS risk category
The median NRS score for the study population was 2.32. Subgroup analyses were performed according to NRS risk category: those with an NRS score ≥3 were categorized as high-risk (malnutrition, 213 patients [41.93%]), while those with an NRS < 3 were categorized as low-risk (normal nutrition, 295 patients [58.07%]). Subgroup analyses based on NRS risk category showed that 130 patients in the high-risk group died within 28 days, with a mortality rate of 61.03%, while only 29 patients in the low-risk group (9.83%) died within 28 days of hospitalisation (P < 0.001). In addition, statistically significant differences in sex, age, serum albumin, lymphocytes, PNI score, nutritional support level, and complications (gastrointestinal bleeding, and emerging bacterial and fungal infections) were noted between malnourished and well-nourished patients (P < 0.05; Table 3).
Differences in demographic and clinical data between patients hospitalised with severe and critical coronavirus 2019 categorised as high risk or low risk according to NRS score.
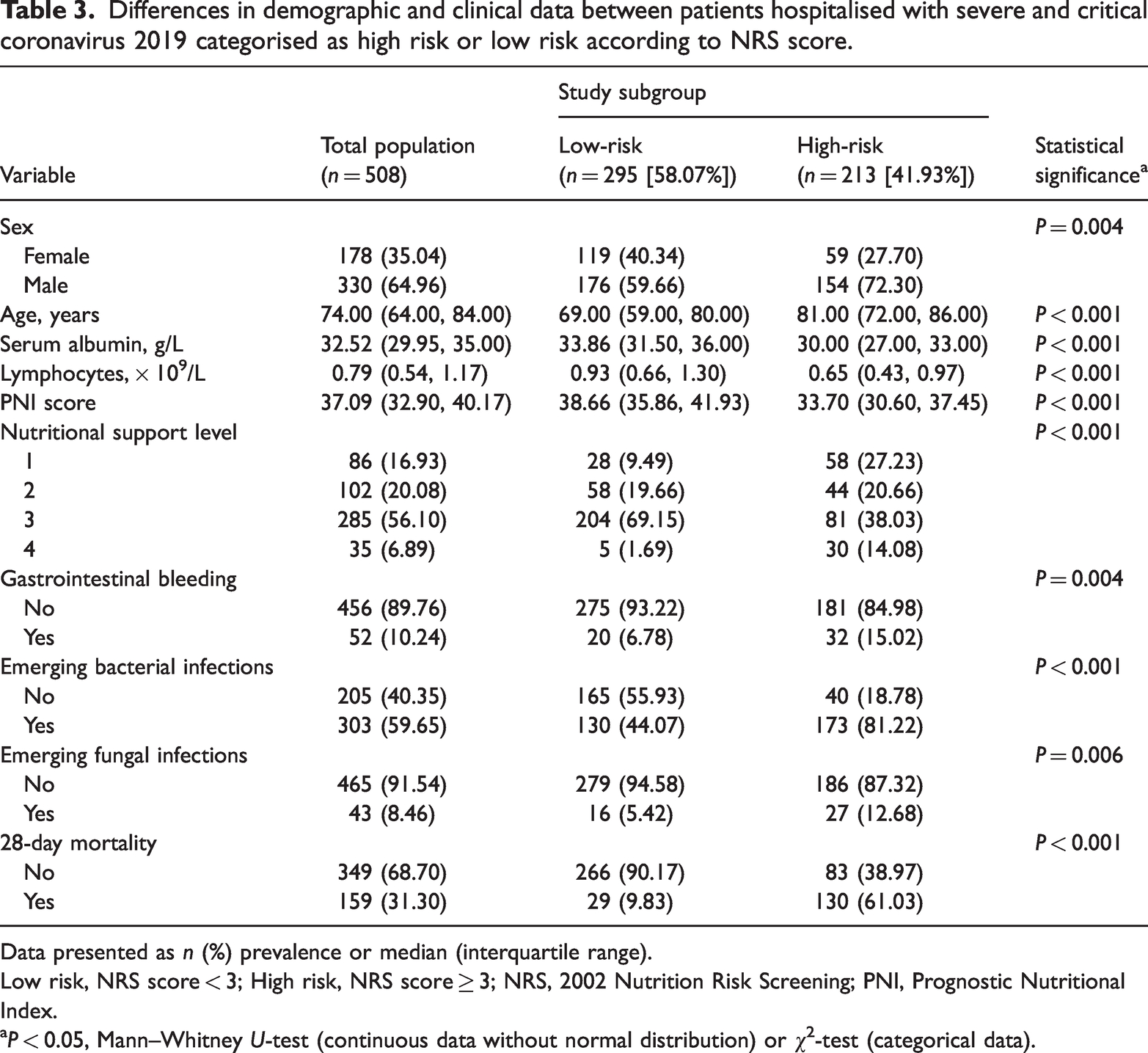
Data presented as n (%) prevalence or median (interquartile range).
Low risk, NRS score < 3; High risk, NRS score ≥ 3; NRS, 2002 Nutrition Risk Screening; PNI, Prognostic Nutritional Index.
P < 0.05, Mann–Whitney U-test (continuous data without normal distribution) or χ2-test (categorical data).
Prognostic analysis based on NRS score category
Risk of NRS score category (<3 versus ≥3) was assessed in patients stratified according to nutritional support level, mechanical ventilation status, and the presence of combined chronic diseases (Figure 1). The ORs indicated that nutritional support level 2 had the strongest association with a high NRS score, followed by levels 4, 1, and 3. Mechanical ventilation was also more strongly associated with high NRS scores versus no mechanical ventilation requirement. Patients with and without chronic diseases had similar odds of being classified in the high NRS group. Subsequent Kaplan–Meier survival curves were used to assess the relationship between median 28-day survival and nutritional support methods or nutritional status (NRS-2002 score) in patients hospitalised with severe and critical COVID-19. The 28-day mortality rates for patients under the four different nutritional support modalities, ranked from highest to lowest, were: those receiving both enteral and parenteral nutrition, those without any nutritional intervention, those on sole enteral nutrition, and those on sole parenteral nutrition (Figure 2). As depicted in Figure 3, the mortality rate was significantly higher in the high-risk malnutrition group (NRS score ≥ 3) compared with the low-risk group (NRS score < 3).
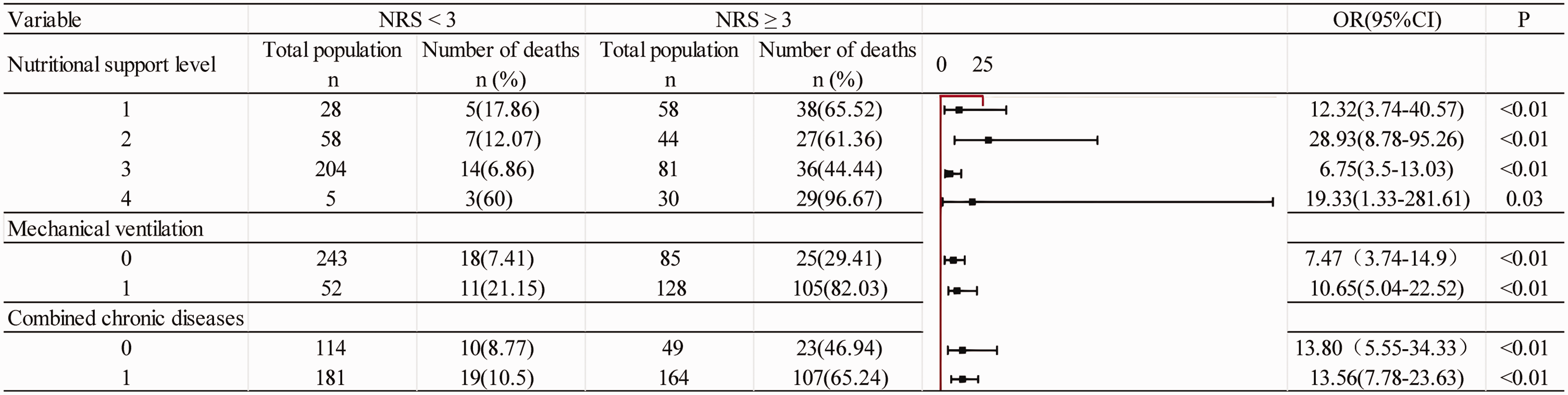
Forest plot showing the association between different subgroups and nutritional risk screening category.

Kaplan–Meier survival curve showing survival rate for 508 patients hospitalised with severe and critical coronavirus 2019 who received different nutritional support programs: (1) regular diet; (2) enteral nutrition; (3) parenteral nutrition; and (4) combined enteral nutrition plus parenteral nutrition, following the American Society for Parenteral and Enteral Nutrition clinical guidelines.
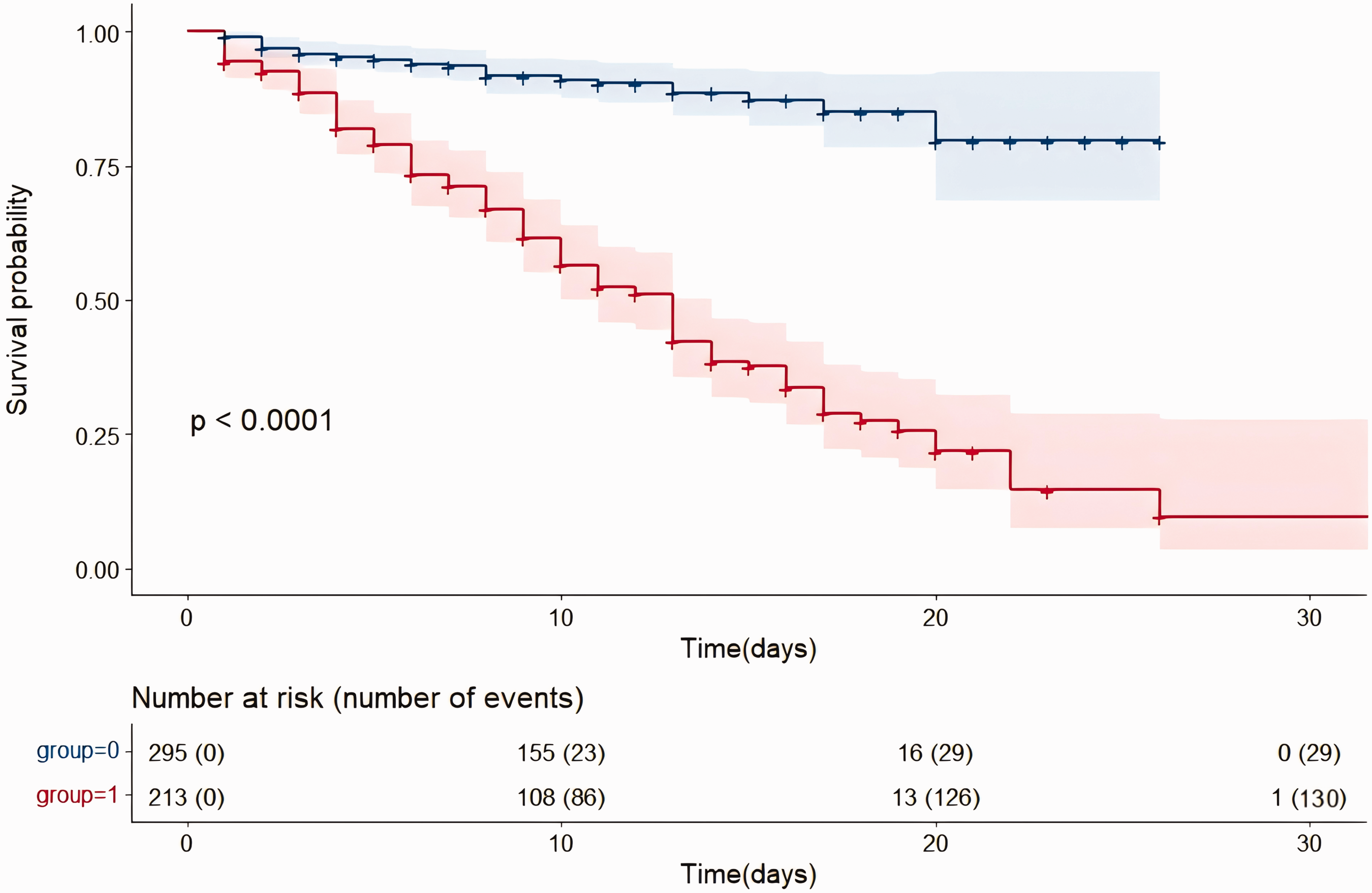
Kaplan–Meier survival curve showing survival rate for 508 patients hospitalised with severe and critical coronavirus 2019 categorised into high-risk (group 1) and low-risk (group 0) based on nutritional status: 2002 Nutrition Risk Screening (NRS) score ≥3 (high risk) or <3 (low risk).
Discussion
In the present study, patients were grouped according to 28-day survival to assess the impact of nutritional status on short-term outcomes, given that nutritional interventions might influence survival within this critical time-frame. A total of 508 critically and severely ill patients with COVID-19 were included, of whom, 349 survived for 28 days and 159 died within that period. Among the 508 patients, 213 had an NRS score of 3 or higher, indicating a malnutrition rate of 41.93%. The PNI analysis showed that survivors had a median index of 38.20 (interquartile range, 35.65, 41.25), considered not to be malnourished, while non-survivors had a median index of 32.65 (29.65, 36.58), indicating malnutrition. Four nutritional support methods were compared: regular diet (86 cases), enteral nutrition (102 cases), parenteral nutrition (285 cases), and a combination of both (35 cases). Through multivariate logistic regression, factors such as high NRS score, fourth level nutritional support (EN+PN), co-existing chronic diseases, PII, use of mechanical ventilation, and longer hospital stays were associated with mortality. Malnourished patients had a 3.87 times higher risk of death than well-nourished patients, highlighting the significant impact of malnutrition on adverse outcomes.
The present findings align with existing studies globally that report malnutrition rates of 49.11%–52.7% in patients with COVID-19, emphasizing the crucial role of malnutrition in poor outcomes, including death,20,21 and reinforce previous research, suggesting that these findings are applicable to all populations, regardless of ethnicity. Malnutrition is often linked to more severe illness, which in turn exacerbates malnutrition, ultimately increasing the risk of death due to factors including low protein levels, inflammation, respiratory failure, and the depletion of the patient’s body reserves. These interactions contribute to the detrimental effects of malnutrition on patient outcomes. 22
The NRS scale, a widely recognized tool in the Chinese healthcare sector, 23 was employed in the present study to evaluate the initial nutritional status of 508 patients with COVID-19, of whom, 41.93% were found to be malnourished (NRS score ≥ 3). Internationally, the NRS is acknowledged as an effective tool for identifying malnutrition, with a score of 3 or above being strongly associated with a higher risk of mortality in critically and severely ill patients. 24 This early assessment highlights the importance of monitoring nutritional status in such patients.
The PNI was employed to evaluate the immune-nutritional status of critically and severely ill patients with COVID-19 at Chongqing Medical University First Hospital. The present results showed that patients who survived for the 28-day period had a median PNI score of 38.20 (35.65, 41.25), indicating no nutritional risk, while patients who did not survive during the 28-day period had a median score of 32.65 (29.65, 36.58), reflecting malnutrition. These PNI scores were consistent with a study in Hanoi, Vietnam, which also reported a mean score of 42.9 for nutritional risk in patients with COVID-19. 25 In the present study, PNI scores were lower in non-survivors than 28-day survivors, in line with findings from previous research in the USA. 26 PNI has been demonstrated to exhibit high predictive power for adverse outcomes, such as death, as it is linked to a patient’s nutritional status and immune system response, based on blood lymphocyte counts and albumin levels. 27 In critical and severe COVID-19 cases, low albumin levels are common, due to inflammation-induced inhibition of protein synthesis, decreased intake, increased capillary permeability, reduced liver synthesis, and ongoing losses from the kidneys or gastrointestinal tract. 28 Additionally, severe infections, such as COVID-19, are characterized by lymphopenia, impairing the immune system, 29 and a correlation has been shown between more severe lymphopenia and increased mortality risk. 30 When NRS data are insufficient (due to unavailable weight or BMI data), the PNI may serve as a substitute indicator, reflecting the nutritional status of patients with COVID-19 through lymphocyte and albumin values.
Previous studies have shown that severe complications and death in critically and severely ill patients with COVID-19 are multi-factorial, involving conditions such as acute respiratory distress syndrome (ARDS), acute multi-organ failure, and septic shock. 31 The present research highlights a strong association between malnutrition and mortality in patients with severe COVID-19, which aligns with existing studies that have found a higher mortality rate and risk of severe complications in critically ill patients with malnutrition, including those with COVID-19. 24 Factors such as gastrointestinal symptoms resulting from severe illness, drug-induced side effects, and increased metabolic demands during infection contribute to endocrine disruption and acute inflammatory responses, exacerbating malnutrition. 32 However, severe malnutrition, by causing a lack of energy for the body, may trigger pathophysiological responses that accelerate disease progression, further increasing the risk of death. 33 This underscores the importance of addressing malnutrition in the management of severe COVID-19 cases.
Four levels of nutritional support were compared among the 508 patients in the present study: 86 patients received a regular diet (level 1 support), 102 patients received enteral nutrition (level 2 support), 285 patients received parenteral nutrition (level 3 support), and 35 patients received combined enteral and parenteral nutrition (level 4 support). Kaplan–Meier survival analysis showed that patients with higher nutritional risk had worse prognoses, while those with lower nutritional risk had better prognoses. The 28-day in-hospital mortality rates for the different nutritional patterns, from high to low, were: patients with combined enteral and parenteral nutrition support, patients without nutritional intervention (regular diet), patients with single enteral nutrition, and patients with single parenteral nutrition. This result may be related to the relatively severe condition of the patients; autonomous eating and simple enteral or parenteral nutrition may not meet the energy demands of the disease, or the patient may no longer able to eat autonomously. A review of the literature revealed that existing studies have confirmed that critically ill patients have poor feeding abilities, increased energy expenditure, and may require parenteral or combined enteral-parenteral nutrition. 34 Nevertheless, critically ill patients often have a higher risk of death due to malnutrition and severe inflammatory storms leading to respiratory failure. 35 Therefore, personalized nutritional patterns are a matter that warrants clinical consideration for such patients.
Given the ongoing sporadic spread of COVID-19 globally, small-scale transmission and prevalence of COVID-19 will persist for years. The present study included a moderate number of patients in Chongqing, effectively illustrating the effectiveness and feasibility of using the NRS scale to assess the nutritional risk of patients with COVID-19, and providing evidence-based guidance for clinical decision-making. However, these results may not entirely reflect the nutritional status of the broader population of Chinese patients, and the results are limited in terms of regional representation. A select group of chronic comorbidities (diabetes, and cardiovascular, respiratory, liver, and kidney diseases) were included, based on their direct impact on COVID-19 prognosis. While the Charlson Comorbidity Index was not used in the present study, we acknowledge its value and will consider using it in future research. Additionally, some patient data could not be collected, such as the weight of bedridden patients, which made the calculation of BMI impossible. Therefore, the result of a 41.93% prevalence of malnutrition in the present study population may not reflect the true rate, and the authors unanimously agree that the actual figure might be higher. Finally, as this was a retrospective study, the implementation of scientific and effective interventions was not possible for the adverse prognoses predicted by malnutrition in severely and critically ill patients with COVID-19.
Conclusion
Based on the research presented above, it is evident that critically and severely ill patients with COVID-19 are at a higher risk of developing malnutrition. This condition is associated with severe complications and mortality. Nutrition risk assessment scales (NRS/PNI) are effective in identifying malnutrition risk in COVID-19 patients, allowing for early intervention in clinical care. Importantly, for patients with a higher nutritional risk, a combination of different nutritional support methods is recommended, considering the interplay between disease progression and malnutrition. Early implementation of nutritional interventions, along with personalized nutritional support strategies and dynamic assessments, are crucial to meet patients’ nutritional needs and minimize related complications.
Footnotes
Acknowledgments
We would like to express our heartfelt gratitude to all patients who provided their medical records and to the nursing staff involved in the study. We are also grateful to all involved in the study for their contributions, ensuring that the research adheres to all legal and ethical standards.
Author contributions
LJ and LY contributed to the conception and design of the study. DT and LJ assisted with data collection. LJ and SYG contributed to data analysis and interpretation. SYG drafted the manuscript. Both LJ and SYG rigorously revised the manuscript. All authors approved the final version and take responsibility for the integrity and accuracy of the research work.
Data availability statement
Datasets generated and/or analysed in the present study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by ChongQing Science and Health Joint Project (2020FYYX109) and ChongQing Key Speciality Construction Project for Clinical Nursing (0203 [2023]47 No. 202336).
