Abstract
Malignant mesothelioma is a rare highly invasive tumour originating from the mesothelial cells of the pleura, peritoneum and pericardium. Malignant pleural mesothelioma (MPM) is the most common type in all malignant mesothelioma. The onset of MPM is associated with exposure to asbestos and it can have an incubation period of up to 40 years. The incidence of MPM has been increasing worldwide in recent years, so more attention has been focused on its diagnosis, treatment and prognosis. Activating mutations, amplifications and fusions/rearrangements of the anaplastic lymphoma kinase receptor tyrosine kinase (ALK) gene are commonly seen in patients with non-small cell lung cancer. However, it is rare in MPM. This current case report describes a female patient with advanced MPM with an ALK gene fusion mutation. In this particular case, treatment with crizotinib demonstrated some initial efficacy, which suggests that this might be a promising strategy for patients with advanced MPM with an ALK gene mutation. This required further research and evaluation in the future.
Keywords
Introduction
Malignant pleural mesothelioma (MPM) is a type of pleural mesothelium originating from the pleura. 1 The exact pathogenesis behind the development of the disease remains unclear. The main risk factors for MPM are asbestos exposure, 2 radiation history and genetic factors; and preventive measures to reduce the use of asbestos have reduced the incidence rate of MPM to some extent.3,4 MPM is generally in the late stage at first diagnosis; and treatment is difficult and the curative effect is poor. The median survival time of patients with MPM is approximately 1 year and the 5-year survival rate is approximately 10%. 5 Patients are rarely cured of MPM. Due to the late diagnosis, most patients miss the opportunity for surgical treatment and there is no universal standardized treatment plans. 6 During the past 20 years, platinum and pemetrexed chemotherapy has been the only approved first-line chemotherapy treatment regimen. 7 The addition of bevacizumab to the platinum-pemetrexed regimen was shown to be beneficial in the French phase III MAPS trial. 8 The addition of bevacizumab to pemetrexed plus cisplatin significantly improved overall survival with an increased rate of adverse events. 8 With the progress of immunotherapy, immune checkpoint inhibitors have shown outstanding efficacy in MPM.4,9,10 The combination of nivolumab and ipilimumab has become a viable first-line option in patients with MPM; especially in those patients with non-epithelioid histologies.11,12 To date, although surgical resection, chemotherapy and immunotherapy have made clear progress,3,13 the prognosis and survival of patients with MPM remains poor. 14 The common genes associated with MPM are the BRCA1 associated deubiquitinase 1 (BAP1) gene, cyclin dependent kinase inhibitor 2A (CDKN2A) gene and NF2, moesin-ezrin-radixin like (MERLIN) tumour suppressor (NF2) gene.14–16 However, the clinical response of MPM patients to targeted therapy remains to be explored. The anaplastic lymphoma kinase receptor tyrosine kinase (ALK) gene rearrangement is extremely rare in MPM and there are few reports. 17 This current case report describes the therapeutic effect of crizotinib in a patient with MPM with a positive ALK mutation, which provides a basis for exploring targeted therapy for MPM patients with rare gene mutations.
Case report
In March 2022, a female patient in her early 70s with no history of smoking, family history of disease and asbestos exposure was admitted to Department of Internal Medicine, The Affiliated Cancer Hospital of Zhengzhou University & Henan Cancer Hospital, Zhengzhou, Henan Province, China for more than 20 days because of a lung mass with pleural nodules. Computed tomography (CT) showed masses in the lower lobe of the left lung, left pleural and interlobar pleural thickening with multiple nodules, and left pleural effusion (Figure 1). Thyroid ultrasound showed 13 × 5 mm lymph nodes on the left clavicle. Head enhanced magnetic resonance imaging showed multiple demyelinating changes of the white matter in the brain. Emission CT showed active bone metabolism in the left 7th rib and active bone metabolism in the 8th and 11th thoracic vertebrae and right acetabulum. The imaging department considered this to indicate bone degeneration. In order to determine the pathological type, a bronchoscopic biopsy was performed with the informed consent of the patient. The results showed that a malignant tumour was considered as follows: (i) adenocarcinoma; (ii) malignant mesothelioma (Figure 2(a)). Immunohistochemistry of the tumour sample was positive for cytokeratin (CK), vimentin, CK5/6, CK7, cluster of differentiation 68 (CD68), calretinin (CR), Wilms tumour protein-1 (WT-1), 25% positive for Ki-67; but negative for P40, thyroid transcription factor-1 (TTF-1), napsin A, cluster of differentiation 56 (CD56), synaptophysin (SyN), podoplanin (D2-40) and paired box 8 (Pax-8) . Negative results for TTF-1 (Figure 2(b)) and napsin A fluorescence in situ hybridization (FISH) staining suggested that a lung adenocarcinoma was unlikely. At the same time, the specific markers of MPM were positive for CR (Figure 2(c)) and positive for CK5/6 (Figure 2(d)) FISH staining, suggesting the pathological diagnosis of MPM was correct.
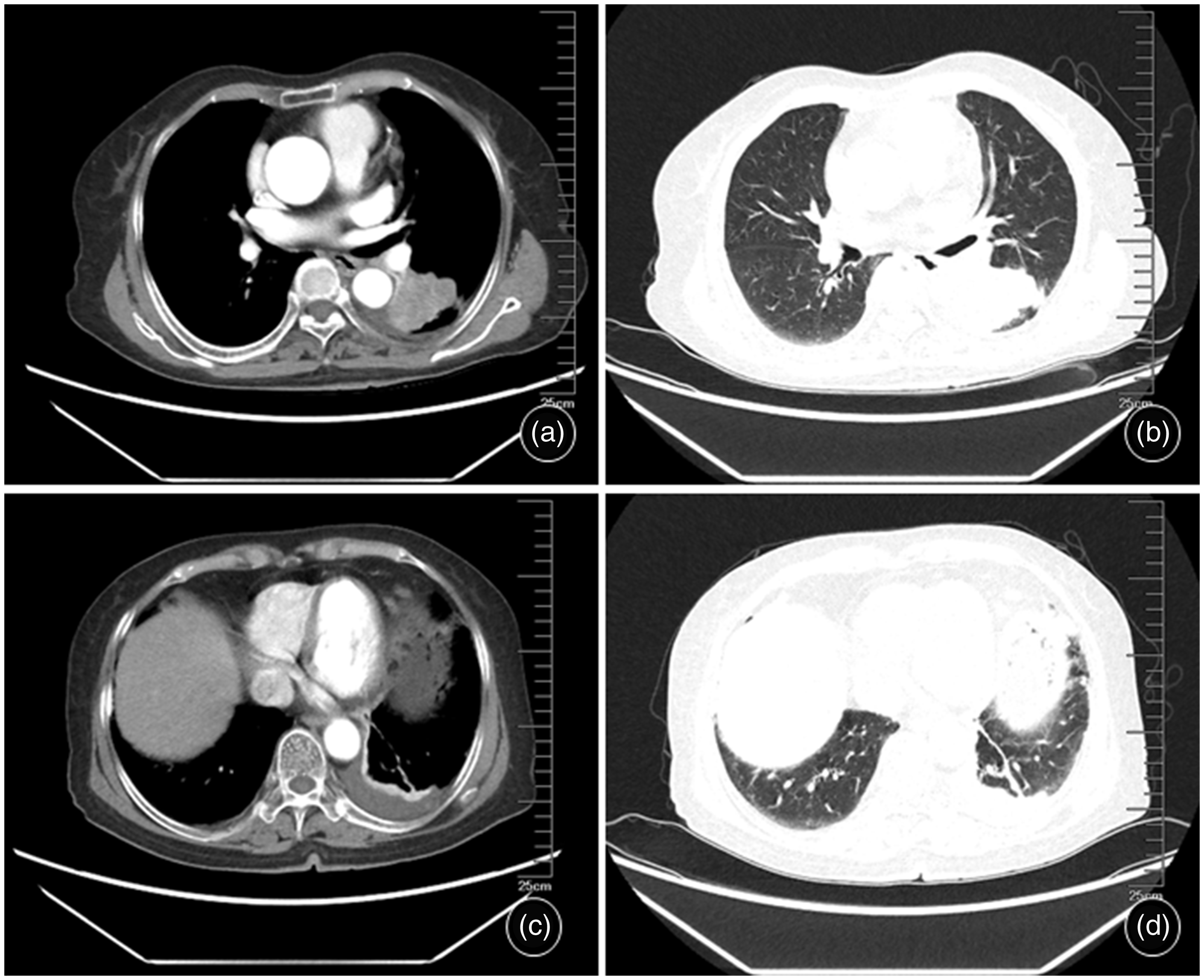
Chest computed tomography (CT) images of a female patient in her early 70s with no history of smoking, family history of disease and asbestos exposure who was admitted because of a lung mass with pleural nodules. CT imaging before treatment showed the following: (a) mediastinal window: masses in the lower lobe of the left lung, left pleural and interlobar pleural thickening with multiple nodules, and left pleural effusion; (b) lung window corresponding to A; (c) mediastinal window: left pleural thickening with left pleural effusion and (d) lung window corresponding to C.
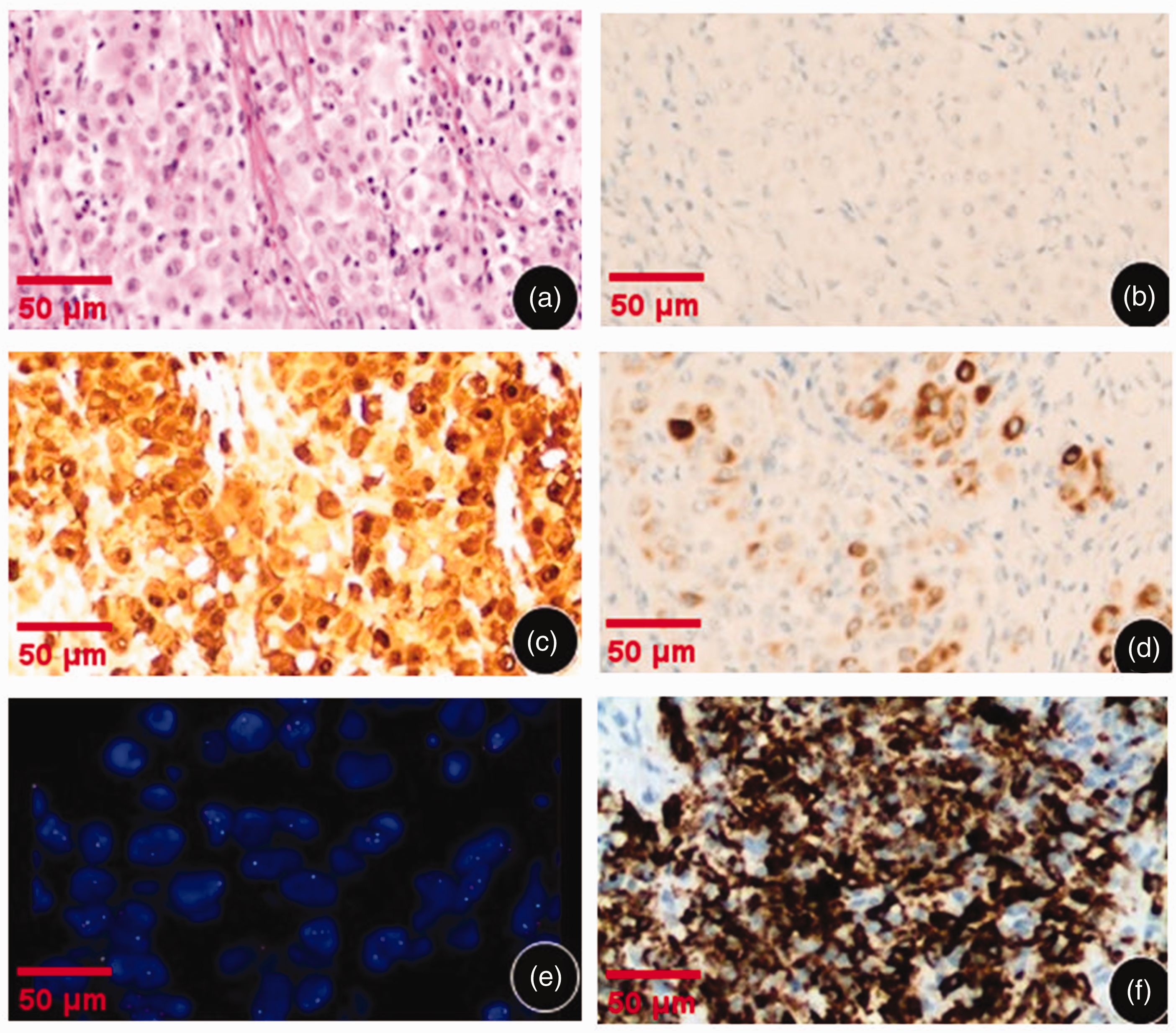
Pathological analyses of the resected specimen taken from a female patient in her early 70s with no history of smoking, family history of disease and asbestos exposure who was admitted because of a lung mass with pleural nodules: (a) tumour puncture biopsy section (haematoxylin & eosin); (b) fluorescence in situ hybridization (FISH) staining showed that the sample was negative for thyroid transcription factor-1; (c) FISH staining showed that the sample was positive for calretinin; (d) FISH staining showed that the sample was positive for cytokeratin 5/6; (e) FISH staining showed that the sample was positive for the cyclin dependent kinase inhibitor 2A (CDKN2A) gene deletion and (f) immunohistochemical staining showed strong positive staining of the anaplastic lymphoma kinase receptor tyrosine kinase (ALK) gene fusion protein. Scale bar 50 µm.
The pathological characteristics and treatment of lung adenocarcinoma and MPM are completely different, so the differential diagnosis is extremely important. FISH staining demonstrated that the specimen was positive for the CDKN2A gene deletion (Figure 2(e)). The CDKN2A gene is a common variant gene of MPM and this deletion confirmed that the pathological diagnosis was MPM rather than lung adenocarcinoma. At the same time, in order to further clarify the pathology, next generation sequencing (NGS) was used to detect 26 lung cancer-related genes in the biopsy specimen. The results suggested that there was an EMAP like 4 (EML4)-ALK-subtype 3-(E6a: A20) gene rearrangement, which results from the fusion of exon 6 of the EML4 gene and exon 20 of the ALK gene (Figure 2(f)). ALK gene rearrangement is extremely rare in MPM and there have only been five previous reports.17–21 Taking into account the pathological findings, the patient was diagnosed as advanced MPM with a EML4-ALK fusion mutation (T4N2M1, stage IV).
The Multiple Disciplinary Team (MDT) discussed the fact that there is a drug, crizotinib, that specifically targets the ALK mutation. Some reports suggest that the use of crizotinib in other lung tumours with the ALK mutation has achieved remission. 6 After a comprehensive evaluation, the current patient was administered 250 mg crizotinib oral treatment twice a day for 150 days. The patient provided consent for this treatment.
During the course of treatment, the patient had mild skin itching and gastrointestinal reactions including nausea and vomiting, but these were tolerable. After treatment with crizotinib for 3 months later, a chest enhanced CT scan showed that the left lower lobe mass and the left pleural thickening with multiple nodules were smaller than before (Figure 3). Her clinical symptoms of abdominal distension and chest tightness were also alleviated. The curative effect was evaluated as a partial response (PR). Two months later, after 5 months crizotinib treatment, the patient went to The Xihua First Hospital, Zhoukou, Henan Province, China for chest CT examinations, which found the following: the density of soft tissue in the dorsal segment of the lower lobe of the left lung had increased slightly, the pleural nodule in the left part of the lobe had increased slightly and there was pleural effusion on the left side and a little pleural effusion on the right side (Figure 3). The curative effect was evaluated as PR maintenance.

Chest computed tomography (CT) images of a female patient in her early 70s with no history of smoking, family history of disease and asbestos exposure who was admitted because of a lung mass with pleural nodules during the course of her treatment. The first image shows the chest CT scans before a diagnosis of malignant pleural mesothelioma (MPM) (same as Figure 1). The second image shows the chest CT scans of the patient after 3 months of treatment with 250 mg crizotinib oral treatment twice a day. These scans showed that the pulmonary masses and pleural thickening were significantly reduced. The third image shows the chest CT scans after 5 months of treatment with 250 mg crizotinib oral treatment twice a day. These follow-up CT scans showed that the lung mass and pleural thickening were slightly enlarged.
All procedures performed in this study were undertaken in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). The patient provided written informed consent to participate in this study. Written informed consent was also obtained from the patient for the publication of any potentially identifiable images or data included in this article. The reporting of this study conforms to CARE guidelines. 22
Discussion
In recent years, targeted tumour therapy has become the focus of considerable research. Gene rearrangement involving the ALK gene has been found in many cancers.23–27 However, there is still a lack of data on MPM with ALK gene mutations. 18 Only five cases have been reported previously and the use of crizotinib is reported here for the first time in this current case.17–21 In 2007, the ALK gene was found to be a potential target for lung cancer therapy. 28 The ALK gene is one of the important genes associated with lung adenocarcinoma, which is usually presented in the form of EML4-ALK fusion gene mutation.29,30,31 The EML4-ALK fusion gene represents one of the newest molecular targets in non-small cell lung cancer (NSCLC). The EML4 and ALK genes are located on p21 and p23 of human chromosome 2, respectively.32,33 The inverted fusion of the two gene fragments can make the tissue express a new fusion protein EML4-ALK, which has carcinogenic activity in vivo and in vitro. 34 ALK positivity is becoming increasingly common in NSCLC and other solid tumours.35–38
Clinically, there are many types of MPM; and their morphology and structure are very complex. 39 It is difficult to distinguish between and diagnose NSCLC and MPM of epithelial type. 40 The commonly used differential method is immunohistochemical analysis. The main markers supporting the diagnosis of MPM include calretinin, CK5/6, WT1, mesothelin and D2-40. 41 The main markers supporting the diagnosis of lung adenocarcinoma include TTF-1, napsin A, carcinoembryonic antigen, BerEP4 and claudin 4. 42 Usually, at least two MPM markers and two lung adenocarcinoma markers should be used for a differential diagnosis. The immunohistochemical staining of this patient was TTF-1 (–), napsin A (–), CK5/6 (+), WT-1 (+) and D2-40 (–), so it was impossible to distinguish MPM from lung adenocarcinoma. As a consequence, FISH staining was undertaken and it demonstrated that the specimen was positive for the CDKN2A gene deletion, which identified it as MPM. When the diagnosis of MPM is highly suspected clinically, it should be evaluated as soon as possible by an MDT with experience in the diagnosis and treatment of MPM. This allows for an early diagnosis and timely treatment. This current patient was diagnosed as MPM with an ALK mutation after pathological biopsy and NGS. The common MPM mutation is the deletion of the CDKN2A gene and this was observed in the current patient. The CDKN2A deletion mutation is found in almost 100% of patients with sarcoma MPM and it is usually associated with a high degree of malignancy and a poor prognosis.43,44
After MDT evaluation combined with the current patient's age and physical factors, the ALK targeting drug crizotinib was chosen as the first-line treatment. After 3 months of treatment, chest CT imaging showed that the left lung mass, pneumonia, pleural thickening with multiple nodules and pleural effusion were significantly reduced. Her clinical symptoms of abdominal distension and chest tightness were also alleviated. After 5 months of treatment, chest CT imaging showed that the lung mass and pleural thickening were slightly increased and her response was categorized as PR maintenance. Therefore, for clinically rare MPM with an ALK mutation, targeted therapy with crizotinib can be used as an effective treatment. Research findings have found a close correlation between ALK and the activation of the MEK5/ERK5 pathway,45,46 so whether crizotinib is related to this pathway still needs to be confirmed through further research. The number of patients with MPM that has this rare ALK gene mutation is gradually increasing, but there are currently no relevant treatment guidelines. The progression-free survival of the current patient was 5 months, which was not long, but it was consistent with the characteristics of a highly malignant tumour and the poor prognosis of MPM. It also contrasts with the long survival time of NSCLC with an ALK mutation. 47 Referring to the targeted therapy of NSCLC with an ALK mutation, the clinical team treating the current case suggested and implemented a new treatment scheme according to the idea of treating different diseases with the same treatment; and this approach achieved good results. However, only one case was reported in this study and more cases are needed in the future to verify the efficacy and mechanism of crizotinib in the treatment of MPM with an ALK gene mutation.
In conclusion, this current case report of a patient diagnosed with MPM with an ALK mutation indicates that crizotinib treatment is a potential new option for patients with such mutations. In the future, further studies should be conducted on the effectiveness and safety of crizotinib for MPM with an ALK mutation.
Footnotes
Author contributions
Yuhua Zhao and Limeng Yu were involved in the study conception and writing of the article. Ruilin Wang, Wen Feng and Yingxi Wu provided pathological data. Lili Wang, Haiyang Chen and Zhen He provided imaging data. Yufeng Wu read and revised the entire manuscript. All authors were involved in revising, reading and approving the submitted version of the manuscript.
Data availability statement
The original contributions presented in the study are included in the article/supplementary material, and further inquiries can be directed to the corresponding author.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This work was supported by a project cosponsored by Key Research and Development Projects of Henan Province in 2023 - Key technologies of novel precision immunotherapy for refractory malignant tumors (No. 231111313300), Henan Medical Key Laboratory of Refractory lung cancer (No. [2020]27), Key project of medical science and technology in Henan Province (SBGJ202101009), Leading Talent Cultivation Project of Henan Health Science and Technology Innovation Talents (YXKC2020009), Henan Refractory Lung Cancer Drug Treatment Engineering Technology Research Center (No. [2020]4), Natural Science Foundation of Henan (No. 242300420094).
