Abstract
Objective
To identify the risk factors for and short-term prognosis of intrauterine infection (IUI) in preterm infants.
Methods
We retrospectively collected clinical data regarding preterm infants (28+0–36+6 weeks of gestational age) with IUI from the neonatal intensive care unit of our hospital between June 2017 and June 2022. The pathologic characteristics of the infants were classified using the Redline criteria, and the resulting groups were compared with respect to their clinical characteristics, indicators of infection, complications, and mortality rates.
Results
Three hundred thirty (78.1%) of the 422 enrolled neonates showed signs of histologic IUI; 51.8% showed histologic chorioamnionitis alone, and 48.2% showed both histologic chorioamnionitis and funisitis. The independent risk factors identified for IUI were maternal prenatal invasive manipulation, premature rupture of membranes (>18 hours before labor), and prenatal C-reactive protein concentration. The infants with IUI showed higher incidences of respiratory distress syndrome, bronchopulmonary dysplasia, retinopathy of prematurity, and prolonged mechanical ventilation. Low gestational age and low birth weight of preterm infants were significantly associated with a higher incidence of histologic IUI, more severe inflammation, and a higher incidence of complications.
Conclusions
Histologic IUI is associated with high complication and mortality rates in preterm infants, as is an increasing severity of inflammation.
Keywords
Introduction
Intrauterine infection (IUI), also known as amniotic infection syndrome, is an infection of the amniotic fluid, placenta, and fetus caused by pathogenic microorganisms that invade the amniotic cavity. 1 Preterm birth has remained common in recent years, with a global incidence of approximately 5% to 18%, 2 and IUI is a major cause of preterm birth. Previous studies have shown that the early diagnosis and treatment of IUI can reduce the risk of maternal and neonatal complications. 3
A diagnosis of IUI can be made clinically and/or histologically. However, a clinical diagnosis lacks specificity and can be affected by other factors. 4 Histologic IUI can involve the maternal and/or fetal interstitium. Histologic chorioamnionitis (HCA) is diagnosed if a pathologic examination of the placenta reveals signs of inflammation that are limited to the placenta and fetal membrane. However, if the inflammation is more severe and involves the umbilical cord blood vessel wall, it may lead to funisitis (FV). 1 Histologic IUI is not associated with a set of typical clinical symptoms, which makes early diagnosis and treatment difficult. Notably, it has previously been shown that a diagnosis of IUI made using clinical findings alone excludes 73.3% of patients with histologic IUI. 4 Therefore, histologic examination remains the gold standard means of diagnosing IUI in clinical practice, because it accurately reflects the intrauterine condition of the fetus.5,6
Previous studies7–9 have shown that the complications of prematurity are associated with clinical and histologic IUI, but to varying extents. A meta-analysis conducted by Blondel et al. 7 showed that clinical IUI is associated with periventricular leukomalacia (PVL) and cerebral palsy, whereas histologic IUI is only associated with PVL. However, Pappas et al. 8 showed that histologic IUI is associated with low Bayley III mental development index in patients with cerebral palsy. Recent studies have also identified associations between IUI and adverse neonatal outcomes, but the relationships between the degree of histologic IUI-associated inflammation and adverse neonatal outcomes remain unclear. 9 Furthermore, although both HCA and FV are acute inflammatory lesions in patients with histologic IUI, FV is associated with a higher incidence of neonatal complications than HCA. 10 Therefore, in this study, we compared the clinical data obtained from a control group with no placental pathology, an HCA + FV− group, and an HCA + FV+ group, to identify independent risk factors and characterize the relationships between the degree of histologic IUI-related inflammation and adverse neonatal outcomes. In this way, we aimed to provide a theoretical basis for the development and implementation of effective interventions for patients with IUI to reduce their risks of preterm complications and mortality, and improve their prognosis.
Materials and methods
Patient sources and groups
We performed an observational, retrospective, multi-center cohort study. We collected data from the clinical records of hospitalized preterm infants (28+0 to 36+6 weeks of gestation [WG]) and their mothers who had been diagnosed with IUI between June 2017 and June 2022 in the Department of Neonatology of the First Affiliated Hospital of Chengdu Medical College, Sichuan Provincial Women’s and Children’s Hospital, and Xindu District People’s Hospital. The study was approved by the Medical Ethics Committee of the First Affiliated Hospital of Chengdu Medical College (ethics approval no. KY2022-95) and was conducted according to the principles of the Declaration of Helsinki. We obtained the general verbal consent from the families of the patients for their participation in retrospective studies. Coding was used to refer to the patients instead of using their names. The participants were duly informed that all data gathered during the research would be held in strict confidence and solely used for scientific purposes. The reporting of this study conforms to the STROBE guidelines. 11
Diagnostic criteria
The pathologic characteristics of the placenta, membranes, and cord were described and classified according to the Redline criteria. 6 HCA was diagnosed if the pathologic examination revealed inflammation that was limited to the placenta and fetal membranes, and FV was diagnosed if the inflammation was worse and involved the umbilical cord blood vessel wall. The neonates were placed into three groups on the basis of the results of the pathologic examination of the fetal adnexa: a control group with no placental pathology, an HCA + FV− group, or an HCA + FV+ group.
Clinical IUI was diagnosed if one of the following was present: 12 ① the presence of placental pathology suggestive of HCA and/or FV; ② amniotic fluid culture positive for pathogenic microorganisms; or ③ the presence of diagnostic criteria for clinical IUI. Clinical IUI was defined as the presence of at least two of the following criteria during labor, in the absence of any other known cause: maternal fever (>37.5°C), maternal tachycardia (>100 beats per minute [bpm]), fetal tachycardia (>160 bpm) or bradycardia (<120 bpm), maternal leukocytosis (>15,000 leukocytes/mm3) or a leftward shift in leucocyte maturity, vaginal discharge, purulent or foul-smelling amniotic fluid, and uterine fundal tenderness. The diagnostic criteria for histologic IUI 6 were diffuse infiltration of the chorionic villi, amniotic membrane, and meconium with neutrophils on histopathologic examination. FV was diagnosed if inflammation had progressed and spread to the umbilical cord (umbilical vein, umbilical arteries, and Wharton’s jelly), as part of the fetal inflammatory response syndrome. The classification of the preterm infants based on gestational age (GA) and birth weight (BW) and the diagnostic criteria used for the related complications were obtained from the fifth edition of Practical Neonatology. 13
Inclusion and exclusion criteria
The inclusion criteria were as follows: 1) GA of 28+0 to 36+6 WG, and the availability of 2) complete birth medical records, 3) complete records for the mother, and 4) a complete set of pathologic information regarding the placenta. The exclusion criteria were as follows: 1) incomplete clinical data or abandoned treatment, 2) inherited metabolic disease or severe congenital developmental malformation, and 3) age >24 hours at the time of admission.
Data collection
We recruited preterm infants who were delivered during the period between June 2017 and June 2022. Data for the mothers and neonates were retrospectively obtained from the hospital’s electronic medical record system. The following maternal parameters were recorded: the age at delivery, occurrence of multiple births, mode of delivery, results of a standardized prenatal examination, occurrence of invasive prenatal manipulation, occurrence of premature rupture of the membranes (PROM), presence of prenatal fetal heartbeat and fetal movement, prenatal circulating indicators of infection (white blood cell [WBC] count, neutrophil percentage [N%], platelet [PLT] count, and C-reactive protein [CRP] concentration), and details of placental pathology. We also recorded the following neonatal data: GA; sex; BW; small-for-gestational-age (SGA) status; the Appearance, Pulse, Grimace, Activity, and Respiration (APGAR) score 1, 5, and 10 minutes postnatally; the need for resuscitation (at least ventilation using a mask); the presence of respiratory distress syndrome (RDS) or neonatal pneumonia; the need for intubation and the presence of surfactant at birth; the duration of invasive and non-invasive ventilation; the presence of bronchopulmonary dysplasia (BPD), intracranial hemorrhage (IVH), PVL, culture-confirmed early-onset sepsis (EOS), late-onset sepsis (LOS), patent ductus arteriosus (PDA), retinopathy of prematurity (ROP), or neonatal necrotizing enterocolitis (NEC); death during hospitalization, postnatal (within 24 hours) routine blood data; and other indicators of inflammation.
Pathologic examination of the placenta
The maternal placenta, membranes, and cord tissue were sent for pathologic examination immediately after delivery. The membranes and placenta were sampled at multiple locations in the center of the placenta, the periphery, and the umbilical cord, ensuring that each sample contained meconium, chorionic villi, chorionic villus plates, and amniotic membrane. After fixation in 10% formaldehyde, the samples underwent paraffin embedding, sectioning, and hematoxylin and eosin staining. 6 Two highly qualified pathologists analyzed all the tissue sections.
Statistical analysis
SPSS 26.0 (IBM Corp., Armonk, NY, USA) was used for statistical analysis. Normally distributed data are described using the mean ± standard deviation, and Student’s t-test or ANOVA was used to compare the groups. Non-normally distributed data are described using the median (interquartile range) and comparisons between groups were made using the rank–sum test. Categorical data are described using number (%), and pairwise comparisons between multiple groups were made using the Bonferroni test. Independent risk factors were identified using dichotomous multifactorial logistic regression, and the Hosmer–Lemeshow test was used to test the model’s goodness of fit. Statistical significance was accepted at a two-sided P of <0.05.
Results
Comparison of the general clinical information
We enrolled 422 preterm infants (42.5% girls), with a mean GA of 31.0 ± 3.8 weeks and mean BW of 1,752.9 ± 498.1 g. The full dataset is provided in Figure 1. Notably, 330 (78.1%) of the 422 enrolled neonates showed signs of histologic IUI; 171 (51.8%) showed signs of HCA alone, and 159 (48.2%) showed signs of HCA + FV (Table 1).

Flow diagram for the study.
Comparison of the clinical characteristics of mothers of preterm infants with IUI.

Student’s t-test or the rank sum test were used, as appropriate. aP < 0.05 vs. the control group. bP < 0.05 vs. the HCA + FV− group. IUI, intrauterine infection; HCA, histologic chorioamnionitis; FV, funisitis; IQR, interquartile range; PROM, premature rupture of the membranes; WBC, white blood cell; N% neutrophil percentage; PLT, platelet; CRP, C-reactive protein.
Comparison of the clinical characteristics of mothers of preterm infants with IUI
The proportion of mothers who underwent a standardized prenatal examination was lower (89.4% vs. 93.4%, 81.1% vs. 93.4%; P < 0.05), the proportion who underwent invasive prenatal manipulation was higher (8.1% vs. 5.4%, 10.6% vs. 5.4%; P < 0.05), and the proportion who underwent cesarean section was lower (62.5% vs. 75.0%, 51.5% vs. 75.0%; P < 0.05) for the HCA + FV− and HCA + FV+ histologic IUI groups vs. the control group, respectively. Pairwise comparisons among the three groups showed that the proportion of mothers undergoing standardized prenatal examinations decreased, the proportion undergoing prenatal invasive manipulation increased, and the proportion undergoing cesarean section decreased as the degree of inflammation present on histologic sections increased. However, the maternal age; parity; incidences of multiple birth, intrauterine distress, fecal staining of amniotic fluid, and placenta previa did not differ between each histologic IUI group and the control group (Table 1).
The incidences of PROM (>18 hours before labor) and prenatal fever, the prenatal WBC count, the prenatal CRP concentration, and the incidences of the prenatal administration of antibiotics and steroids were higher in both histologic IUI groups than in the control group (all P < 0.05, Table 1). Pairwise comparisons among the three groups showed that the incidences of PROM (>18 hours before labor) and prenatal fever, the prenatal CRP, and the prenatal incidences of antibiotic and steroid use increased as the degree of inflammation in the patients with histologic IUI increased (all P < 0.05, Table 1).
Comparison of clinical characteristics in preterm infants with IUI
There were significant differences between the IUI groups and the control group with respect to GA and BW (all P < 0.05, Table 2). Pairwise comparisons showed that the lower were the BW and the GA, the more severe was the histologic IUI inflammation (all P < 0.05, Table 2). There were no significant differences in the sex or incidence of SGA between the IUI groups or between each histologic IUI group and the control group (Table 2).
Comparison of the clinical characteristics of the groups of preterm infants.

Student’s t-test or the rank sum test were used, as appropriate. aP < 0.05 vs. the control group. bP < 0.05 vs. the HCA + FV− group. HCA, histologic chorioamnionitis; FV, funisitis; SGA, small-for-gestational-age; GA, gestational age; BW, birth weight; WBC, white blood cell; N% neutrophil percentage; PLT, platelet; PCT, plateletcrit.
The plateletcrit (PCT) of the preterm infants in both of the histologic IUI groups were significantly higher than that of the control group (P < 0.05). Pairwise comparisons showed that the higher the PCT count was, the more severe the histologic IUI inflammation was (Table 2).
Comparisons of the incidences of IUI in preterm infants with various GA and BW
Of the 422 preterm infants (28+0 to 36+6 WG) enrolled in the study, 160 (37.9%), 153 (36.2%), and 109 (25.8%) were in the very preterm (28+0–31+6 WG), moderate preterm (32+0–33+6 WG), and late preterm (34+0–36+6 WG) groups, respectively. Compared with the very preterm group (28+0–31+6 WG), the incidence of IUI was significantly lower in the moderate preterm (32+0–33+6 WG) and late preterm (34+0–36+6 WG) groups (all P < 0.05, Supplementary File 1). Notably, 182 (43.1%) of the preterm infants had very low BW (1,000 to 1,500 g), 201 (47.6%) had low BW (1,500 to 2,500 g), and 39 (9.3%) had normal BW (2,500–4,000 g). The incidence of histologic IUI in the preterm infants was significantly higher in the very low BW group (1,000 to 1,500 g) than in the other groups (P < 0.05, Supplementary File 1).
Results of the multifactorial logistic regression analysis of IUI in the preterm infants
Table 3 shows that invasive prenatal manipulation, PROM >18 hours before labor, and high prenatal CRP concentration were independent risk factors for IUI in the preterm infants (P < 0.05), but standardized prenatal examination, cesarean section, and prenatal fever were not. The goodness-of-fit for the model was good (P = 0.411, Table 3).
Results of the multifactorial logistic regression analysis of IUI in preterm infants.
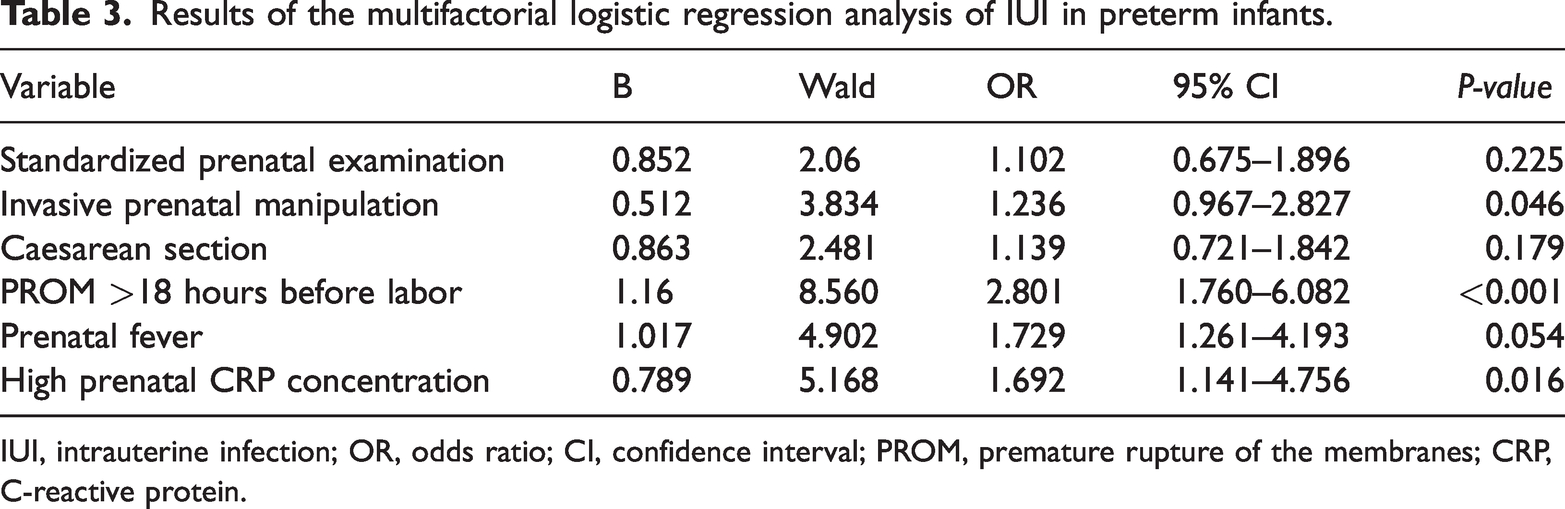
IUI, intrauterine infection; OR, odds ratio; CI, confidence interval; PROM, premature rupture of the membranes; CRP, C-reactive protein.
Comparison of the incidences of short-term complications and mortality for the preterm infants in each IUI group
The incidences of 1-min and 5-min APGAR scores <7, resuscitation at birth, neonatal asphyxia, RDS, neonatal pneumonia, BPD, IVH, ROP, the use of alveolar surface-active substances, and the duration of invasive/non-invasive mechanical ventilation were significantly higher in both of the histologic IUI groups than in the control group (all P < 0.05, Supplementary File 2). Pairwise comparisons showed that the incidences of resuscitation at birth, BPD, and IVH, and the duration of invasive mechanical ventilation all increased with increases in the degree of histologic inflammation (Supplementary File 2).
Comparison of the incidences of short-term complications and mortality of the preterm infants of all gestational ages in the HCA+FV− group
For infants that were HCA + FV−, the incidences of complication and mortality were significantly lower in the moderate preterm (32+0 to 33+6 WG) and late preterm (34+0 to 36+6 WG) groups than in the very preterm group (28+0 to 31+6 WG) (P < 0.05, Table 4). Pairwise comparisons among the three groups showed that, except for the incidences of RDS, BPD, ROP, NEC, EOS, LOS, abnormal blood glucose concentration, and mortality, all the incidences evaluated increased as GA decreased (all P < 0.05, Table 4).
Comparison of the incidences of short-term complications and the mortality of preterm infants according to GA in the HCA + FV− and HCA + FV+ groups.

Student’s t-test or the rank sum test were used, as appropriate. aP < 0.05 vs. the 28+0–31+6 WG group. bP < 0.05 vs. the 32+0–33+6 WG group. HCA, histologic chorioamnionitis; FV, funisitis; APGAR, Appearance, Pulse, Grimace, Activity, and Respiration score; RDS, respiratory distress syndrome; BPD, bronchopulmonary dysplasia; IVH, intracranial hemorrhage: PVL, periventricular leukomalacia; EOS, culture-confirmed early-onset sepsis; LOS, late-onset sepsis: PDA, patent ductus arteriosus; ROP, retinopathy of prematurity; NEC, neonatal necrotizing enterocolitis.
Comparison of the incidences of short-term complications and mortality among preterm infants of various GA who were HCA+FV+
For infants who were HCA + FV+, the incidences of complications and mortality were significantly lower in the moderate preterm (32+0–33+6 WG) and late preterm (34+0–36+6 WG) groups than in the very preterm group (28+0–31+6 WG) (P < 0.05, Table 4). When pairwise comparisons of these groups were performed, except for the incidences of BPD, EOS, LOS, abnormal blood glucose concentration, and mortality, all the incidences evaluated significantly increased as GA decreased (all P < 0.05, Table 4).
Discussion
IUI is one of the most common causes of preterm labor. Previous studies6–9 have identified differing risk factors for IUI, associations between IUI and adverse outcomes in preterm infants, and effects of various levels of inflammation on adverse outcomes in preterm infants with different GAs. This is probably because of differences in the inclusion criteria and diagnostic criteria used for IUI. Therefore, in the present study, we aimed to characterize the relationships among risk factors for IUI, the degree of inflammation in histologic sections, and adverse neonatal outcomes.
PROM occurs in 3% to 8% of pregnant women, 14 and histologic IUI occurs in combination with PROM in 13% to 60% of pregnancies. 15 PROM increases the risk of IUI and is associated with adverse neonatal outcomes.16–18 In the present study, we found that PROM that occurred >18 hours before labor was an independent risk factor for IUI. This is consistent with the findings of previous studies,16,18 and the longer PROM occurs before labor, the higher the incidence of IUI is. 17 Furthermore, most previous studies16–18 have shown that early maternal or neonatal interventions for PROM, such as early delivery or the administration of antibiotics, can reduce the incidences of HCA and neonatal infections. However, early exposure to antibiotics in preterm infants has been shown to be associated with high incidences of complications and mortality.19,20 We also found greater prenatal antibiotic use by mothers in both histologic IUI groups, which is consistent with the findings of previous studies.
We also found that the prenatal WBC counts and CRP concentrations of the mothers in each histologic IUI group were higher than those of the control group, and these also increased as the degree of inflammation on histologic sections increased, suggesting that these parameters may be useful predictors of histologic IUI. Previous studies have also shown9,21 that high prenatal CRP concentrations in mothers suggest a higher risk of IUI. However, the postnatal WBC counts and CRP concentrations of the two histologic IUI groups did not differ. Furthermore, the PCT of the preterm infants in the histologic IUI groups increased as the degree of inflammation on histologic sections increased, which is consistent with the findings of Hartnet et al. 22 However, it is thought that this relationship is affected by the timing of blood sample collection. Stocker et al. 23 found that the CRP concentration between birth and 36 or 48 hours postnatally had no predictive value for neonatal infection. Blood samples were collected within the first 24 hours postnatally in the present study, but preterm infants born to IUI mothers should be closely monitored for changes in the concentrations of markers of infection. 24
HCA affects the development of the fetal lungs 25 and leads to a significantly higher incidence of intrauterine infectious pneumonia. 26 The inflammatory response to IUI leads to the remodeling of the fetal pulmonary vasculature, which increases the volume of the pulmonary circulation and the resistance to pulmonary blood flow, which leads to lower left ventricular output and affects lung development. 25 Sood et al. 27 found that IUI increases the risk of lung disease in preterm infants. We also found that as histologic IUI progressed, the use of prolonged mechanical ventilation, surfactant use, and the incidence of RDS increased in the preterm infants, which implies that the greater the degree of inflammation that is present in histologic IUI, the more severe is the lung damage that occurs. In addition, the presence of histologic IUI increases the risks of ROP and BPD in preterm infants, and these risks increase with the degree of inflammation in patients with histologic IUI, which is consistent with the findings of Pietrasanta et al. 9 Helmo et al. 28 also showed that histologic IUI is significantly associated with the incidence and severity of ROP. However, the mechanism by which histologic IUI leads to ROP remains unclear, and the influence of potential confounding factors, such as extremely premature birth and oxygen therapy, should be evaluated to better understand the development of ROP. 22
Chorioamnionitis in pregnant mothers is closely associated with preterm labor. Lahra et al. 29 identified a negative association between histologic IUI and GA, with an overall prevalence of IUI of 31%, which decreased to 16% after 34 WG. HCA is associated with the low GA and low BW of preterm infants. 30 Similar results were obtained in the present study: the lower the GA of the preterm infants and the more severe the inflammation was in each of the histologic IUI groups, the higher were the incidences of complications and mortality. The incidences of complications and mortality were significantly higher in the extremely preterm infants (28+0 to 31+6 WG). The lower the GA of preterm infants, the more immature their tissues and organs will be, the poorer their immunity will be, 28 and therefore the more likely they will be to develop complications. Previous studies have shown31,32 that early IUI can lead to miscarriage, stillbirth, or congenital malformations. In contrast, most late IUIs are subclinical infections that can induce preterm labor and several neonatal disorders, suggesting that IUI may be more harmful for preterm infants with a low GA. However, in the present study of the incidences of complications and mortality in preterm infants with IUI and differing GAs, we found that the incidences of complications, such as neonatal pneumonia, RDS, IVH, and BPD, increased significantly with increases in the degree of inflammation on histologic sections, suggesting that histologic IUI is harmful to preterm infants of all GAs.
The present study had a few limitations. This was a retrospective study, and therefore we did not follow the preterm infants to identify predictors of their long-term prognosis, such as neurologic, motor, psychologic, or growth-related parameters. We also did not include preterm infants with a GA of <28 weeks, and the sample size was small. Therefore, the findings require validation using a large, multicenter prospective study.
Conclusion
We can conclude that prenatal invasive manipulation, PROM >18 hours before labor, and a high prenatal CRP concentration are risk factors for IUI in preterm infants. In addition, we found that RDS, BPD, ROP, and prolonged invasive/non-invasive mechanical ventilation affect the short-term prognosis of preterm infants with IUI. Finally, we found that the lower the GAs of preterm infants are and the lower their BWs are, the greater the degree of inflammation there is on histologic sections and the higher their risks of complications and mortality are.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241286728 - Supplemental material for Risk factors for and short-term prognosis of intrauterine infection in preterm infants
Supplemental material, sj-pdf-1-imr-10.1177_03000605241286728 for Risk factors for and short-term prognosis of intrauterine infection in preterm infants by Xuemei Zhao, Yue Song and Liqun Lu in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605241286728 - Supplemental material for Risk factors for and short-term prognosis of intrauterine infection in preterm infants
Supplemental material, sj-pdf-2-imr-10.1177_03000605241286728 for Risk factors for and short-term prognosis of intrauterine infection in preterm infants by Xuemei Zhao, Yue Song and Liqun Lu in Journal of International Medical Research
Footnotes
Acknowledgements
We are grateful to the Department of Neonatology, Sichuan Provincial Women’s and Children’s Hospital for providing the clinical data.
Author contributions
XZ, YS, and LL designed the study. XZ, YS, and LL performed the research and analyzed the results. XZ and YS drafted the paper. XZ, YS, and LL edited the manuscript and provided critical comments. All the authors read and approved the final version of the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by a Medical Research Project of Sichuan Province (S21007) and a Chengdu Medical Research Project (2021216).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
