Abstract
Phosphaturic mesenchymal tumors (PMTs) are extremely rare mesenchymal tumors of soft tissue and bone that cause tumor-induced osteomalacia (TIO). Some of these tumors are completely asymptomatic and may grow undetected unless they become large enough to cause pain or discomfort. This type of tumor is crucial to diagnose in patients being treated for phosphate metabolism disorders and are a rare reason why patients seek medical help owing to pain. Here, we report the details of a patient with progressive bone pain caused by a PMT originating in the left femur.
Keywords
Introduction
Phosphaturic mesenchymal tumors (PMTs) are rare tumors of uncertain origin that can cause paraneoplastic syndrome-derived osteomalacia. The clinical manifestations of PMTs are fractures, bone pain, hyperphosphaturia, hypophosphatemia, and inappropriately low serum calcitriol levels.1–2 The main cause of these symptoms is the secretion of phosphatase by the tumor, which leads to excessive excretion of phosphate from the kidneys. 3 Because PMTs are generally small in size and may be hidden, they are difficult to find. 4 The main treatment methods are surgery and radiofrequency ablation (RFA). 3 We report a rare case of a patient with a PMT that caused progressive bone pain for 3 years.
Case presentation
The reporting of this study conforms to the CARE guidelines. 5
The patient was a 75-year-old woman. Three years prior to presentation at our hospital, she developed multiple body aches without obvious causes, especially in her joints, chest, and lower back. She was treated at other hospitals for this pain. A flow chart for assessing patients with progressive bone pain when a diagnosis of PMT is suspected is presented in Figure 1. Serum electrolyte measurement indicated low blood phosphorus, and she was diagnosed with hypophosphatemia and given a phosphorus supplement. Her pain subsequently worsened, and she presented to another hospital for treatment. Positron emission tomography-computed tomography (PET-CT) indicated the possibility of moderate neoplastic lesions in the left femur. She then presented to our hospital for surgery. She had a history of hypertension and long-term oral antihypertensive drug use with good blood pressure control. She also had a history of hepatitis B and was receiving long-term oral entecavir antiviral therapy. There was no history of tuberculosis, coronary heart disease, diabetes, or other diseases. Electrolyte measurement revealed a low blood phosphorus level (0.28 mmol/L; normal value: 0.85–1.51 mmol/L) and a markedly high parathyroid hormone level (142 ng/L; normal value: 15–65 ng/L). Superficial B-ultrasonography revealed a slightly low echogenicity mass measuring 24 × 13 mm on the anterior surface of the left mid-femur, with clear boundaries (Figure 2). Magnetic resonance imaging (MRI) revealed a patchy abnormal signal in the left mid-femur, and T1-weighted imaging (T1WI) revealed a slightly higher-intensity signal that was continuously enhanced in enhanced scans (Figure 3). To further confirm the diagnosis, the tumor was removed from the left mid-femur under general anesthesia. Intraoperatively, the tumor was grayish-brown with clear boundaries. Hematoxylin and eosin (HE) staining of the resected specimen revealed short fusiform and oval cell composition, no obvious nuclear division, and an obvious smoke-like matrix (Figure 4). Postoperative re-examination of the patient’s blood phosphorus level indicated a marked increase compared with preoperative levels, and the level reached the normal range on the 5th postoperative day (Figure 5). Similarly, parathyroid hormone levels decreased markedly and were nearly normal 6 days after surgery (Figure 6). There were no signs of recurrence or metastasis 10 months after surgery.
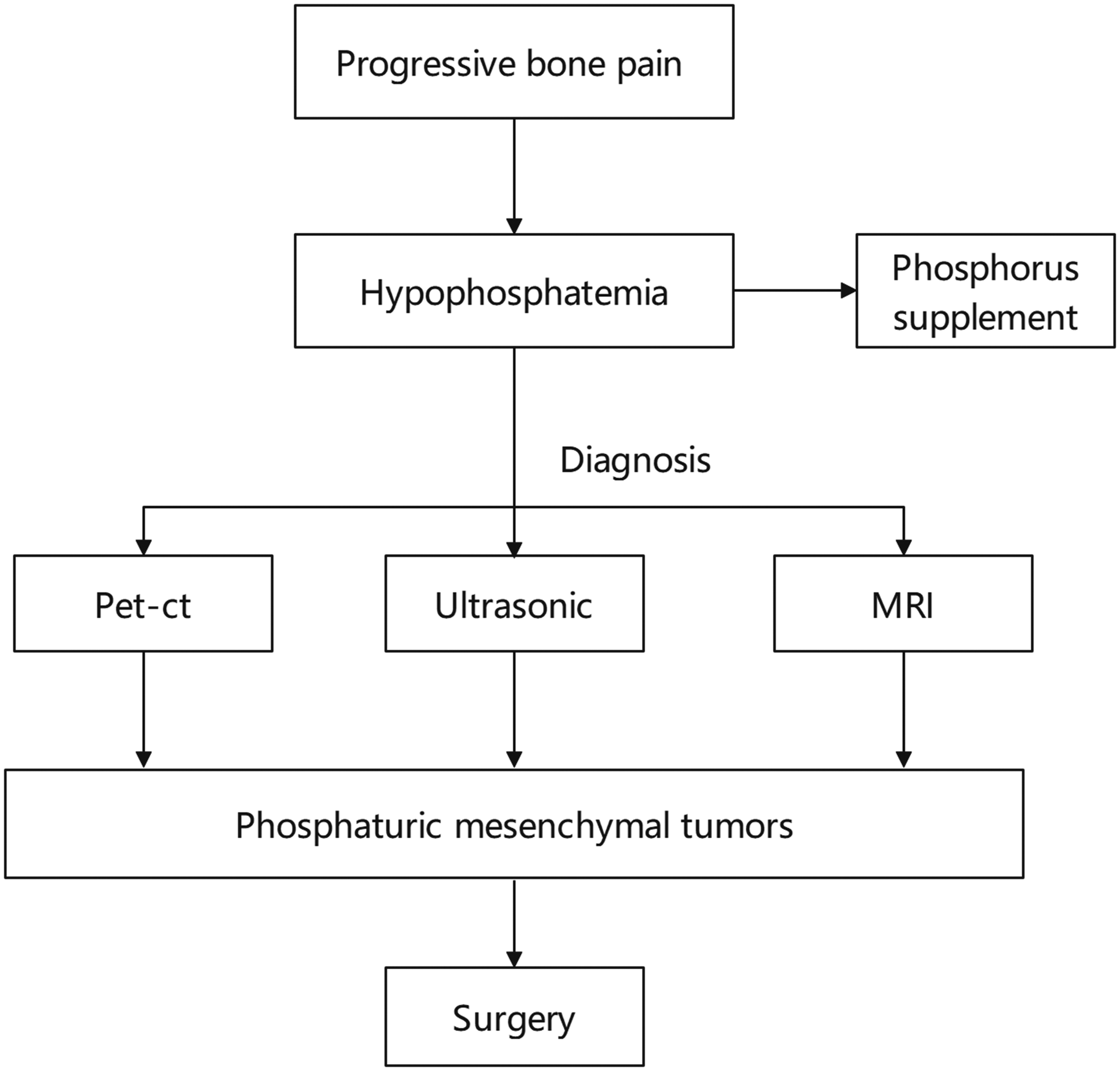
Flow chart for assessing patients with progressive bone pain when a diagnosis of PMT is suspected. PMT, phosphaturic mesenchymal tumor; PET-CT, positron emission tomography-computed tomography; MRI, magnetic resonance imaging.

Superficial B-ultrasonography showing a slightly low echogenicity mass (red arrow) measuring 24 × 13 mm on the anterior surface of the left mid-femur, with clear boundaries.
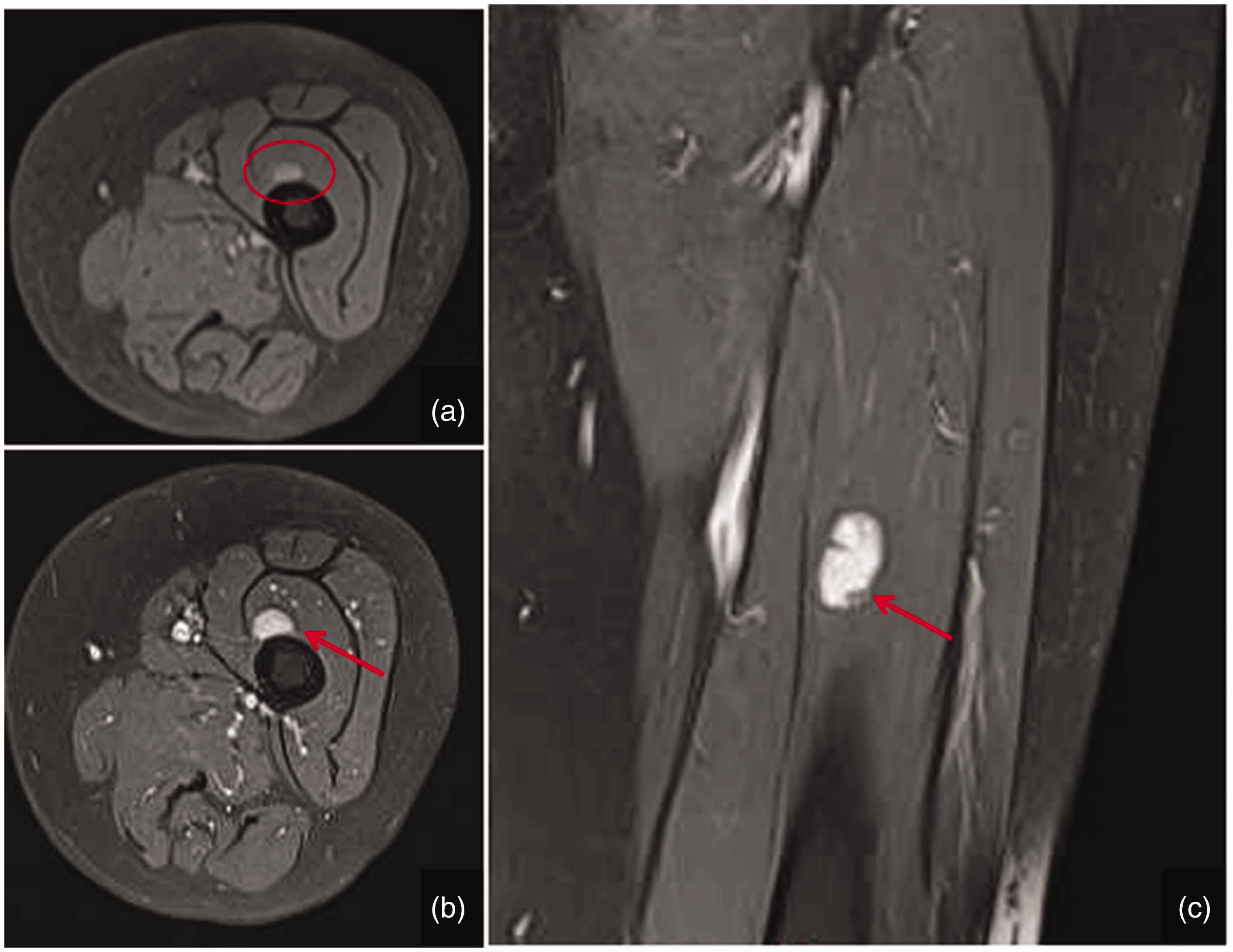
MRI showing a patchy abnormal signal in the left mid-femur (a, red circle), and T1WI showing a slightly increased signal, which was continuously enhanced on enhanced scans (b, c, red arrow in each panel).

HE staining of the resected PMT from the left mid-femur revealed a short fusiform and oval cell composition, no obvious nuclear division, and an obvious smoke-like matrix (area contained within the red circle) (×50). HE, hematoxylin and eosin; PMT, phosphaturic mesenchymal tumor.

Postoperative laboratory examination revealed a markedly increased blood phosphorus concentration compared with preoperative levels, and the level reached the normal range on the 5th postoperative day.
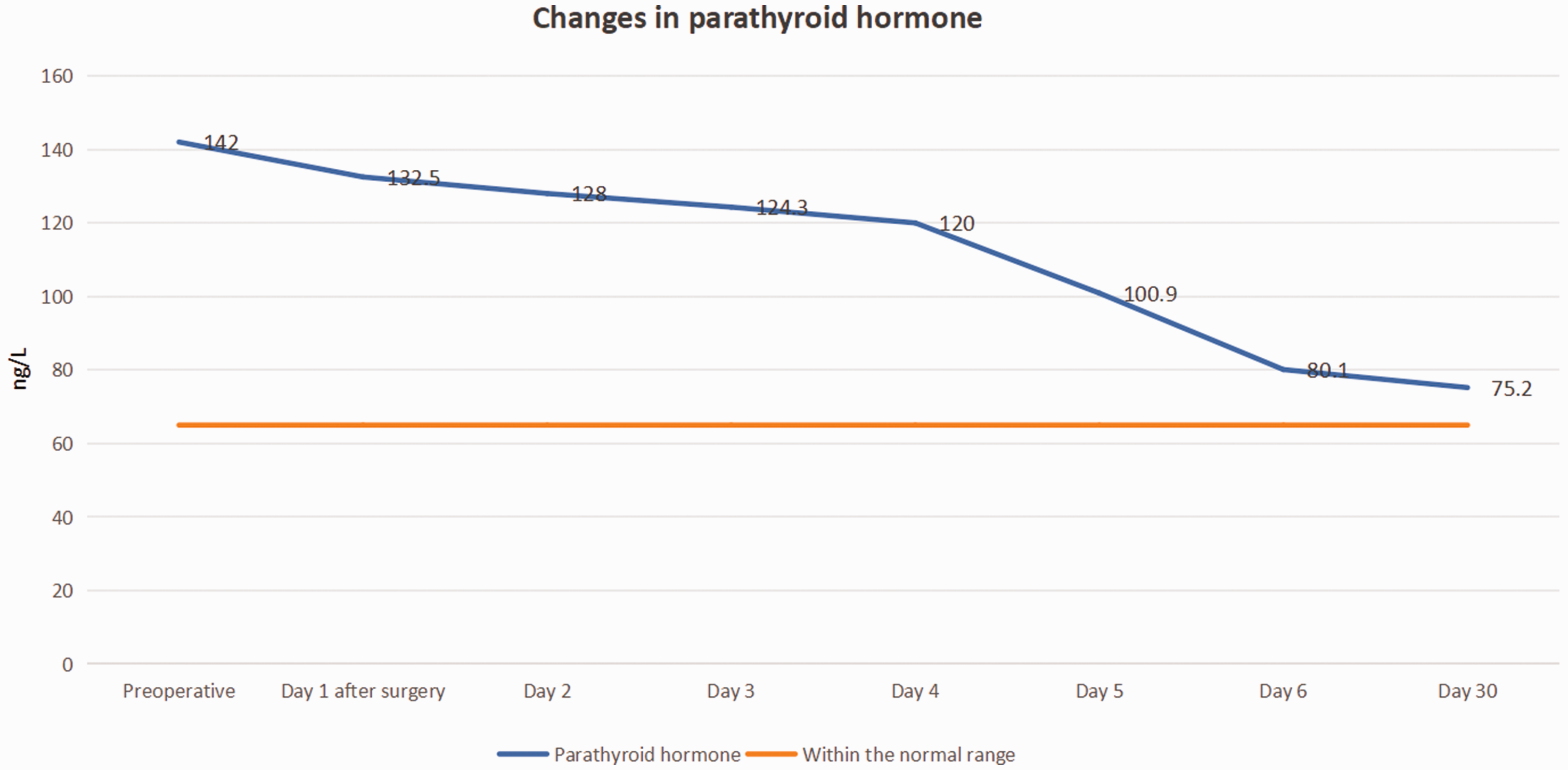
Postoperatively, parathyroid hormone levels decreased markedly compared with preoperative levels and were nearly within the normal range 6 days after surgery.
Discussion
Tumor-induced osteomalacia (TIO) is a rare paraneoplastic syndrome. TIO is caused by the secretion of fibroblast growth factor 23 (FGF23) by the primary tumor. FGF23 has phosphaturic activity and can inhibit the conversion of 25-hydroxyvitamin D to calcitriol and the absorption of phosphate by the renal tubules. This results in increased urine phosphate levels and causes osteomalacia and hypophosphatemia. 3 Many types of tumors cause osteomalacia, such as mesenchymal tumors, prostate tumors, colorectal adenocarcinomas, clear cell carcinomas of the kidney, small cell carcinomas of the lung, B-cell lymphoma, and neurofibromatosis.6–9 TIO-associated mesenchymal tumors represent various benign and malignant tumors and are not uniformly named. 10 In 1987, Weidner and Santa Cruz 11 officially named mesenchymal tumors associated with osteomalacia as PMTs and divided them into four subtypes—mixed connective tissue, osteoblastomatoid, nonossifying fibromatoid, and ossifying fibromatoid subtypes—among which the mixed connective tissue subtype is most common. 12 In 2013, and for the first time, the World Health Organization (WHO) included PMTs as a new type of tumor in the classification of soft tissue tumors. In the past several decades, PMTs have been identified in numerous locations, including the spine, 13 femoral head, 14 knee, 15 and foot. 16
Common symptoms of PMTs comprise fatigue, bone pain, repeat pathological fractures, and muscle weakness. 1 Because of the nonspecific symptoms and the occult nature of these tumors, they are difficult to detect and easy to misdiagnose. Because the lesions are hidden, the disease progresses slowly over a long period resulting in osteomalacia and a series of clinical symptoms. The clinical characteristics are generally, adult onset and more common in men; 17 progressive systemic bone pain, with affected individuals often unable to pinpoint the specific location; and severe skeletal malformations and activity disorders, which are most common in weight-bearing portions of the skeleton, such as the lumbar spine and near the joints of the lower extremities. Our patient developed bone pain 3 years prior to presentation at our hospital and had been treated in other hospitals several times. Laboratory test results revealed low blood phosphorus and high urine phosphorus. After repeat administration of standard doses of a phosphorus supplement, the patient’s symptoms were relieved temporarily, but the bone pain worsened after the withdrawal of the phosphorus supplement.
Because the primary PMT has no typical imaging characteristics and is often hidden, the diagnosis is made mainly on the basis of laboratory test results, such as a low blood phosphorus, high urine phosphorus, high blood alkaline phosphatase, and high secreted FGF23, in addition to comprehensive imaging analysis findings. The possibility of a PMT should be considered in patients with progressive bone pain accompanied by low blood phosphorus levels or high urine phosphorus levels. Superficial B-ultrasonography, limb X-rays, CT, and MRI may detect the presence of some PMTs. PET-CT is the most effective examination.
Notably, PMTs may develop in very hidden locations in the body. Correia et al. 17 reported the case of a 70-year-old man with generalized bone pain, multiple pathological fractures, and persistent hypophosphatemia who was diagnosed with neoplastic osteomalacia 4 years after the onset of symptoms. The tumor was identified in the forefoot using PET-CT and was successfully treated surgically. However, not all lesions can be detected by PET-CT. Then et al. 18 reported the case of a 50-year-old woman who presented with diffuse musculoskeletal pain and multiple fractures. Cancer osteomalacia caused by neoplastic FGF23 secretion was suspected; however, PET-CT did not detect any tumors. The tumor was identified in the cerebellum by brain MRI. Our patient experienced progressive bone pain for 3 years, and no tumor was identified in routine examinations. Finally, PET-CT was performed at another hospital, and a PMT was identified in the left mid-thigh. This case report highlights the importance of recognizing these rare tumors, as early diagnosis can prevent long-term sequelae.
Currently, it is believed that with a clear diagnosis and complete surgical resection, the symptoms of PMTs can be markedly alleviated. However, the biological behavior of PMTs is borderline, characterized by local aggressive growth, often with unclear boundaries with surrounding tissues and the presence of satellite nodules around the tumor; therefore, PMTs are difficult to completely resect surgically. 19 The clinical biological behavior of PMTs is generally benign, but some patients may experience relapse and metastasis, which may lead to death.19,20 Although PMTs are rare, there is still a possibility of recurrence and metastasis in the late stage. It is estimated that <5% of patients experience recurrence, which usually spreads to the lungs. 3 Therefore, long-term follow-up is essential. In our patient, the tumor was eventually resected, and her symptoms were relieved immediately after surgery. Ten months after surgery, there were no signs of recurrence or metastasis, and her bone pain was completely relieved.
In addition to surgery and RFA, some drugs are used in the treatment of TIO. Burosumab is a human monoclonal antibody that antagonizes FGF23 and can normalize phosphatase levels. This drug was approved by the United States Food and Drug Administration for use in the treatment of TIO in 2020. 3 Normalization of serum phosphate levels can reduce the systemic skeletal burden of PMTs, improve fracture healing, and enhance a patient’s quality of life. 3
Conclusions
We reported a rare case of TIO caused by a PMT. Combined with findings in previous studies, we summarized three points. First, PET-CT may be considered for patients with progressive bone pain accompanied by a low blood phosphorus level. Second, high urine phosphorus and markedly low blood phosphorus are typical in patients with PMTs, and a smoky-like matrix is the specific pathological manifestation of PMTs. Third, surgical treatment is appropriate for PMTs, and the prognosis is generally good. Clinicians should increase their awareness of this disease and consider its possibility in patients with bone pain.
Footnotes
Author contributions
Authors YJF and JJL drafted the manuscript. WXG performed the surgery. ZLY assisted with collection of the clinical data and made critical revisions for important intellectual content. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Data availability statement
We declare that the materials described in this manuscript, including all relevant raw data, will be freely available to any scientist wishing to use them for non-commercial purposes without breaching participant confidentiality.
Ethical review
This study was reviewed and approved by the Lishui Municipal Central Hospital Institutional Review Board. Written informed consent was obtained from the patient for the publication of this case report.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
