Abstract
Tumor-induced osteomalacia (TIO) is a rare paraneoplastic syndrome. We herein report a rare case of TIO in a 58-year-old Chinese man who presented with a large lump in the right palm. Clinical, biochemical, and radiological assessments were performed. Laboratory examination showed severe hypophosphatemia, phosphaturia, an elevated serum alkaline phosphatase level, and an elevated serum fibroblast growth factor 23 (FGF-23) level. Dual-energy X-ray absorptiometry showed low bone mineral density. Magnetic resonance imaging revealed an irregular mass located in the right palm and abnormal findings in several metacarpal bones. During the operation, the surgeons found that the tumor had penetrated the surrounding muscles. The tumor had unique characteristics of local tissue invasion. The patient’s symptoms fully resolved and his serum phosphorus level normalized, although his serum FGF-23 level remained slightly high in the postoperative phase. Our findings suggest that in some patients with TIO, the serum phosphorus level might return to the normal range despite a relatively high postoperative serum FGF-23 level. These patients should be kept under close observation and regularly surveyed for any evidence of a residual tumor.
Keywords
Introduction
Tumor-induced osteomalacia (TIO), also termed oncogenic osteomalacia, is a rare paraneoplastic syndrome with an insidious presentation. 1 More than 400 cases have been reported in the English-language literature worldwide since the first case was reported by Robert McCance in 1947.2,3 We herein report a rare case of TIO in which the tumor was located in the palm and showed unique characteristics of local tissue invasion. Notably, the patient’s serum phosphorus level returned to the normal range, although he had a relatively high serum fibroblast growth factor 23 (FGF-23) level in the postoperative phase. All procedures were performed according to the Declaration of Helsinki and approved by the Ethics Committee of the Second Affiliated Hospital and Yuying Children’s Hospital of Wenzhou Medical University.
Case presentation
A 58-year-old Chinese man presented with a 4-year history of fatigue and disabling bone pain that was most prominent in the knees. He was admitted to our hospital in 2015. During the prior 4 years, his symptoms had been progressively worsening. He had been unable to climb stairs for the past 7 months. He had also recently been unable to get up from a supine position. The patient had no history of trauma or any familial inherited bone disorders. He had been diagnosed with type 2 diabetes 2 years previously and was undergoing treatment with metformin with a well-controlled glucose level. On physical examination, his height was 165 cm; it had previously been recorded at 167 cm. He had a waddling gait. Palpation revealed tenderness in the bilateral hypochondriac regions. Muscle strength was normal in all four limbs, although tendon hyper-reflexia was noted. A lump measuring 8 × 6 cm was found in the right palm (Figure 1). The left lower limb was 2 cm shorter than the right limb, and the left lower extremity had an external rotational deformity. The remaining systemic examination findings were unremarkable.

Irregular lump located in the thenar eminence of the right palm
Laboratory findings revealed severe hypophosphatemia, phosphaturia, an elevated serum alkaline phosphatase level, and an elevated serum intact FGF-23 level as measured by enzyme-linked immunosorbent assay (Kainos Laboratories, Tokyo, Japan). The percent tubular reabsorption of phosphate was 77% (reference range, 85% to 95%), and the maximal tubular renal phosphate reabsorption normalized for the glomerular filtration rate was 0.32 (reference range, 0.89 to 1.34). Serum protein electrophoresis findings were normal. The patient’s laboratory data are summarized in Table 1.
Biochemical parameters before and after surgical resection

ALP, alkaline phosphatase; FGF-23, fibroblast growth factor 23; Tmp/GFR, maximal tubular renal phosphate reabsorption normalized for the glomerular filtration rate; PTH, parathyroid hormone; β-CTX, carboxy-terminal telopeptide of type 1 collagen; OC, osteocalcin; P1NP, amino-terminal prepeptide of type 1 procollagen; NA, not available
A pelvic radiograph revealed a fracture of the left femoral neck. Dual-energy X-ray absorptiometry (Lunar Prodigy; GE Medical Systems, Chicago, IL, USA) revealed low bone mineral density at L1–L4 (0.957 g/cm2; T-score, −2.0; Z-score, −1.5), the femoral neck (0.601 g/cm2; T-score, −3.6; Z-score, −2.3), and total hip (0.774 g/cm2; T-score, −2.9; Z-score, −2.1). Whole-body 99mTc-methylene diphosphonate bone scintigraphy showed focal increased uptake in multiple bones and joints (Figure 2). Magnetic resonance imaging revealed an irregular mass located in the right palm (7.2 × 6.2 × 3.3 cm in maximum diameter). The lesion had low signal intensity on T1-weighted images and a heterogeneously isointense bright signal on T2-weighted images. The tumor was broadly attached to the flexor digital tendons. Abnormal signals were present in several metacarpal bones (I/II/V), exhibiting low signal intensity on T1-weighted images and a bright signal on T2-weighted images (Figure 3).
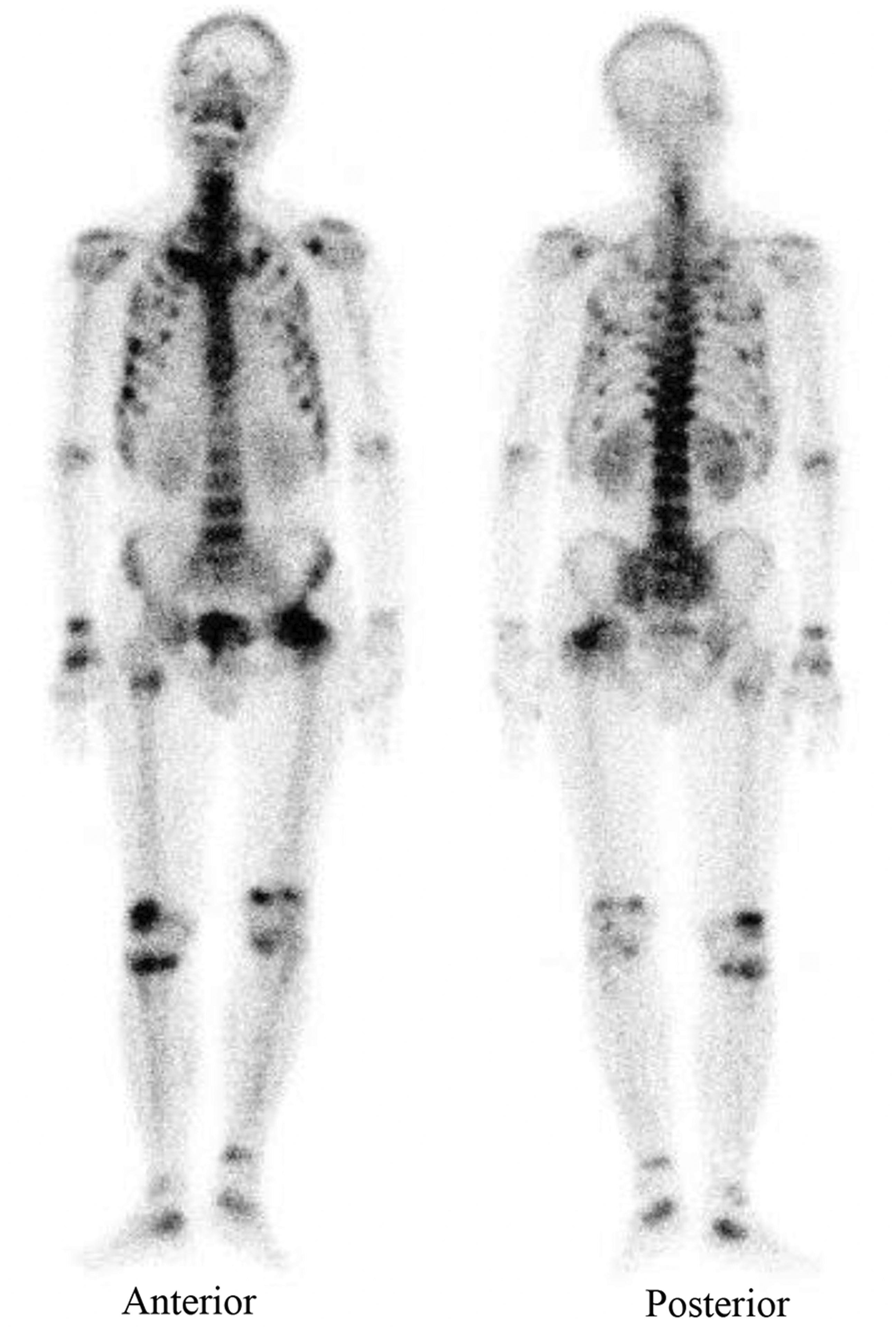
Whole-body 99mTc-methylene diphosphonate bone scintigraphy showed diffusely increased uptake in the left supramaxilla, proximal right clavicle, left scapula, right elbow right wrist, ribs, spinal column, left hip, proximal right femur, knees, ankles, and calcanei

Magnetic resonance imaging revealed an irregular mass. The tumor was generously enhanced by gadolinium. (a, d) Coronal position. (b, e) Sagittal position. (c, f, g) Horizontal position. (a–c) T2-weighted images with fat suppression technique. (d–f) T1-weighted images enhanced by gadolinium. (g) T1-weighted images enhanced by gadolinium with fat suppression technique
The patient underwent surgical resection. During the operation, the surgeons observed that the tumor had penetrated the mesothenar and interosseous muscles. They excised both the tumor and related muscles. Histopathologic examination indicated that the tumor was an epithelioid vascular leiomyoma, categorized as a phosphaturic mesenchymal tumor, mixed connective tissue variant. Immunohistochemistry of the tumor showed positive staining for vimentin, CD68, CD34, smooth muscle actin, and caldesmon (Figure 4).

The tumor was composed of polygonal epithelioid cells with richly eosinophilic cytoplasm. The cell nucleus was an elliptical or fat spindle shape, and binucleate cells were rare. (a) Hematoxylin–eosin staining. (b) Positivity of the tumor cells for vimentin. (c) Positivity of the tumor cells for CD68. (d) Positivity of the tumor cells for CD34. (e) Positivity of the tumor cells for smooth muscle actin. (f) Positivity of the tumor cells for caldesmon. (a–f) Original magnification, ×40
One week after removal of the tumor, the patient’s serum phosphorus level was 0.95 mmol/L and his serum FGF-23 level had decreased dramatically. Significant amelioration of his symptoms was noted 2 weeks later, and he could walk without assistance. Three months later, he was asymptomatic and his serum phosphorus level was maintained within the reference range (Table 1). Dual-energy X-ray absorptiometry showed that his bone mineral density had increased by 66.98% at the lumbar spine and by 19.13% at the hip. At the time of this writing (40 months postoperatively), he was still asymptomatic, had no evidence of tumor recurrence, and was still undergoing follow-up.
Discussion
The differential diagnoses for patients with renal phosphate wasting-induced hypophosphatemia accompanied by an elevated serum FGF-23 level include acquired conditions, such as TIO, and genetic conditions, such as X-linked hypophosphatemia, autosomal dominant hypophosphatemic rickets, and autosomal recessive hypophosphatemic rickets.1,4 According to our patient’s age at onset, negative family history, recovery after tumor removal, and histopathologic findings, we were able to rule out all three of the above-mentioned genetic disorders and confirm the diagnosis of TIO.
TIO is an acquired renal phosphate wasting disease. The main cause of TIO is the overproduction of phosphaturic FGF-23, an important humoral hormone in the regulation of phosphate metabolism, by the tumor.5–7
Locating these tumors is challenging because most of them are small and present in an insidious fashion. TIO is difficult to diagnose and is easily missed even after the initial onset of symptoms.1,4 In a study by Xia et al., 7 44% (17/39) of the tumors could be located by physical examination; the majority of these tumors had to be identified by several different modalities such as F-18 fluorodeoxyglucose positron emission tomography/computed tomography, 111indium-octreotide scintigraphy, 68Ga-DOTATATE positron emission tomography/computed tomography, 201thallium and 99technetium methoxy-isobutyl-isonitrile scintigraphy, and selective venous sampling from head to toe.1,4,7,8 However, the tumors cannot be identified in some patients. In their single-center experience, Xia et al. 7 investigated the distribution of tumors and found that 56% (22/39) of cases were located in the lower extremities, 5% (2/39) were located in the upper extremities, and the rest were located elsewhere. In the cohort examined by Sun et al. 9 (40 patients with TIO involving extremity tumors), only 4 cases were located in the upper extremities; among these, most were within the soft tissue of the forearm, wrist, and arm and within the bone tissue of the ulna. Finding such a large lump in the palm is rare. To date, only Weiss et al. 10 has reported a case in which the tumor was located in the palm with histopathology analysis indicating that it was giant cell variant of soft tissue chondroma.
Resection of the tumor with wide margins is the preferred treatment option. 11 In the current case, magnetic resonance imaging showed that the tumor was broadly attached to the flexor digital tendons, and abnormal signals were detected in several metacarpal bones. Moreover, during the operation, the surgeons found that the tumor had penetrated several muscles. Sun et al. 9 found that two of the three tumors involving both bone and soft tissue were malignant. Therefore, it could be argued that close postoperative monitoring is necessary in our patient.
Our patient’s symptoms completely resolved after surgical resection and his serum phosphorus level returned to the reference range, although his serum FGF-23 level remained slightly high in the postoperative phase. In most cases, a high serum FGF-23 level after surgical resection indicates the presence of tumor remnants that were not resected. Most of these patients still exhibit hypophosphatemia after resection.9,12 In several reported cases, the serum phosphorus level remained within normal limits along with a relatively high serum FGF-23 level.8,12–15 Intriguingly, in most of these cases, the serum phosphorus level was ≤1.0 mmol/L.8,12–14 In the current case, the serum phosphorus level was normal 1 week after the surgical resection and was 1.52 mmol/L at 3 months postoperatively. The restored serum phosphorus level in our case was higher than that in all other reported cases.8,12–14 Nawrot-Wawrzyniak et al. 14 stated that the serum FGF-23 level can be elevated while not affecting phosphate homeostasis in some patients. Such patients might have a relatively higher serum phosphorus storage capability than other patients, or they might differ in their threshold for phosphatonins, such as FGF-23, to trigger hypophosphatemia. In the present case, we are inclined to believe that the slightly elevated serum FGF-23 level after surgical resection was related to residual tumor tissue due to the unique extensive nature of the tumor.
Footnotes
Acknowledgment
We thank the patient for his cooperation.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research was supported by Zhejiang Provincial Natural Science Foundation of China under Grant No. LY15H070003. The funding source had no involvement in the conduct of the research, the writing of the report, or the decision to submit the article for publication.
