Abstract
Objective
This study evaluated the 50% effective dose (ED50) and 95% effective dose (ED95) of butorphanol tartrate in patients undergoing painless gastroscopy.
Methods
Patients who underwent painless gastroscopy at Binzhou Medical University Hospital were divided into the youth, middle-aged, and older groups. The ED50 and ED95 required for successful sedation using butorphanol tartrate were measured using the Dixon up-and-down method in patients in the different age groups. Patients in each group were administered intravenous butorphanol 5 minutes before gastroscopy. Each patient was administered 2 mg/kg propofol. The ED50 and ED95 of butorphanol were calculated using probit analysis.
Results
In total, 95 patients were included. The ED50s of butorphanol in the youth, middle-aged, and older groups were 7.384, 6.657, and 6.364 μg/kg, respectively. The ED95s of butorphanol doses in these groups were 9.108, 8.419, and 7.348 μg/kg, respectively.
Conclusions
The ED50 and ED95 varied among the age groups, indicating that the effective dose decreases with age.
Introduction
With rapid developments in medicine, patient comfort during treatment has become an increasingly important area of focus. Modern gastroscopy has become popular because it alleviates patient anxiety, minimizes discomfort during the procedure, and increases the success rate and efficiency of disease diagnosis.1,2 Propofol is commonly used during painless gastroscopy because of its rapid onset, ease of arousal, and controllability. However, the analgesic effects of propofol are limited, and its administration alone necessitates large doses and causes various side effects. 3 Consequently, propofol is often used in combination with other analgesic drugs in clinical practice. 4
Butorphanol is a mixed opioid receptor agonist and antagonist that provides effective visceral analgesia, alleviates respiratory depression, and effectively reduces the incidence and severity of propofol injection pain. 5 Several studies have demonstrated the efficacy of the butorphanol–propofol combination for gastroscopy, reporting its rapid onset, minimal adverse effects, and high levels of safety and reliability.6,7 The 50% effective dose (ED50) and 95% effective dose (ED95) are widely used in clinical trials to guide drug use.8,9 However, the optimal dosage of butorphanol in different age groups remains unclear. This study used a sequential method to explore the use of butorphanol tartrate in patients of different ages receiving painless gastroscopy (ED50 and ED95) to provide references for clinical treatment, increase comfort, and create a safer medical environment.
Materials and methods
Study design
This prospective, non-randomized, non-controlled trial was approved by the Ethics Committee of Binzhou Medical University Hospital (grant No. KYLL-2022-170,2022-11-22) and registered with the Chinese Clinical Trial Registry (registration no. ChiCTR2200066304). This study conformed to the STROBE guidelines 10 and followed the CONSORT statement. Patients scheduled to undergo gastroscopy at Binzhou Medical University Hospital were eligible for the study. Participants were then grouped by age with no sex restrictions. Our study was conducted in accordance with the Declaration of Helsinki of 1975 as revised in 2013. Before the examination, all patients underwent a preoperative evaluation at the anesthesia clinic and signed an informed consent form. All patient detailed were de-identified.
Inclusion and exclusion criteria
The inclusion criteria were as follows: age ≥18 years with no sex restrictions; American Society of Anesthesiologists (ASA) classification of I or II; body mass index (BMI) of 18 to 25 kg/m2; no history of analgesic drug abuse, serious cardiopulmonary disease, or asthma; and no use of sedatives within 24 hours before the examination. The exclusion criteria were as follows: pregnancy or lactation in women; obvious difficulties in airway and esophageal cancer; combined dysfunction of important organs; and cognitive impairment or mental disorders.
Anesthesia procedure
According to the World Health Organization age classification, the study participants were divided into the following three groups: youth (18–44 years, Group A), middle-aged (45–59 years, Group B), and older (≥60 years, Group C). Upon entering the gastroscopy room, the patients received oxygen
Adequate contingency measures were implemented throughout the examination to mitigate potential complications. In the event of hypotension (blood pressure <20% of the baseline value recorded on admission), intravenous ephedrine or phenylephrine was used as a corrective measure. In cases of respiratory depression (SpO2 < 90%) despite an unobstructed airway, positive pressure ventilation
Outcomes
Main outcome measures
The primary outcomes were ED50 and ED95 in patients of different ages undergoing painless gastroscopy with butorphanol tartrate. The secondary outcome measures included SBP, DBP, HR, and SpO2 measured 1 minute before injection (T1), immediately after the completion of butorphanol administration (T2), immediately after the completion of propofol administration (T3), immediately after gastroscope placement (T4), immediately after endoscope withdrawal (T5). The time of awakening and pain scored on a visual analog scale (VAS) score 10 minutes after awakening were also recorded. Adverse effects including pain associated with propofol injection, hypotension, hypertension, bradycardia, tachycardia, respiratory depression (SpO2 < 90%), dizziness, drowsiness, nausea, vomiting, and agitation during resuscitation were documented. The Ramsay Sedation Scale was used to assess the degree of agitation during resuscitation. The time to awakening was defined as the time from the completion of gastroscopy to the time when the patient was able to open his or her eyes and cooperate in the anesthesia recovery room.
Statistical analyses
SPSS statistical software (version 27.0, IBM Corp., Armonk, NY, USA) was used for data analysis. Normally distributed data were expressed as the mean ± standard deviation, and the Kruskal–Wallis test was performed for non-normally distributed data. Repeated-measures analysis of variance was used to compare the time points. The probit model was employed
13
to calculate the ED50 and ED95 as well as the corresponding 95% confidence intervals (CIs) for butorphanol tartrate combined with propofol to inhibit the response to gastroscope insertion.
14
Sequential plots and dose–effect curves were plotted using Microsoft Excel software (Microsoft, Redmond, WA, USA), and
Results
In total, 95 patients were enrolled in this study, including 30 participants in group A, 35 participants in group B, and 30 participants in group C. As presented in Table 1, there was no significant difference in the distribution of BMI, sex, or ASA classification across the three groups. However, the age distribution significantly differed among the groups (
Comparison of participants’ profiles.

Data are presented as the mean ± standard deviation or number of patients.
BMI, body mass index; ASA, American Society of Anesthesiologists; Group A, patients aged 18–44 years; group B, patients aged 45–59 years; group C, patients aged ≥60 years.
A sequential plot of the response to gastroscope insertion following butorphanol administration using the improved sequential method is presented in Figure 1. The ED50 and ED95 of butorphanol were determined for each patient during gastroscopy. The ED50 and ED95 were 7.384 (95% CI = 6.23–8.523) and 9.108 μg/kg (95% CI = 8.213–24.807), respectively, in group A, 6.657 (95% CI = 5.898–7.573) and 8.419 μg/kg (95% CI = 7.532–16.903), respectively, in group B, and 6.364 (95% CI = 5.954–6.828) and 7.348 μg/kg (95% CI = 6.864–9.923), respectively, in group C. The dose–effect curve of butorphanol regarding inhibition of the response to gastroscope insertion is presented in Figure 2.

Sequential plot of the response to gastroscope insertion.
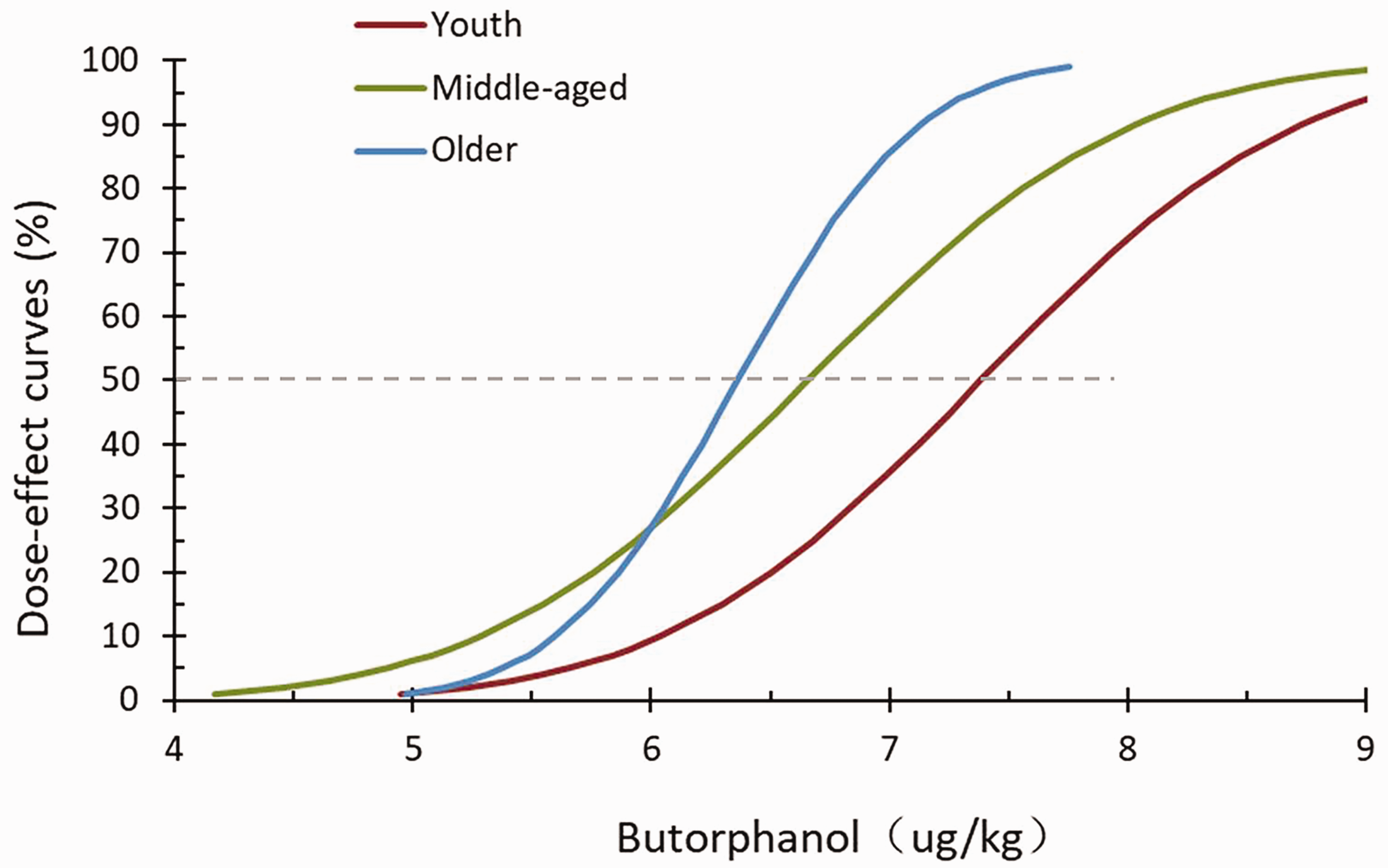
Dose–effect curve of the inhibitory effect of butorphanol on the response to gastroscope insertion.
An ingroup comparison illustrated that SBP, DBP, and SpO2 were significantly lower (all
Group comparisons of the vital signs of participants.

Data are presented as the mean ± SD or number of patients. a:
SBP, systolic blood pressure; DBP, diastolic blood pressure; HR, heart rate; T1, 1 minute before injection; T2, immediately after the completion of butorphanol administration; T3, immediately after the completion of propofol administration; T4, immediately after gastroscope placement; T5, immediately after endoscope withdrawal; Group A, patients aged 18–44 years; group B, patients aged 45–59 years; group C, patients aged ≥60 years.
Comparison of postoperative conditions among the three groups.

Data are presented as the mean ± standard deviation.
VAS: visual analog scale; Group A, patients aged 18–44 years; group B, patients aged 45–59 years; group C, patients aged ≥60 years.
Comparison of adverse reaction occurrences among the three groups (%).

Data are presented as n (%).
Group A, patients aged 18–44 years; group B, patients aged 45–59 years; group C, patients aged ≥60 years.
Discussion
Previous studies indicated that potent opioids such as fentanyl and sufentanil can effectively reduce the amount of propofol required during anesthesia.15,16 However, potent opioids are classified as dangerous anesthetics, and their management in outpatient settings is cumbersome and complicated. By contrast, butorphanol is a second-class psychotropic drug with a relatively simple management system and certain advantages. Butorphanol is a mixed opioid agonist with an affinity of 1:4:25 for the opioid receptors (μ:δ:κ).
17
The pain-relieving effect is achieved by stimulating the κ receptor, and it has a significant advantage in relieving visceral pain. It also increases the frequency of the spontaneous opening of the pyloric sphincter, making endoscopic examination easier.17,18 Butorphanol partially antagonizes the μ receptor, thus reducing the incidence of postoperative adverse reactions such as nausea and vomiting.
19
The onset time of butorphanol after intravenous injection was approximately 1 minute, reaching a peak at 10 to 15 minutes, with rapid elimination after peak efficacy. Compared with other sedatives and hypnotics, the incidence of side effects such as respiratory suppression is low, and a mild analgesic effect can be maintained for an extended period after the sedative effect disappears.
20
Butorphanol is widely used in painless gastroscopy because of its rapid onset of action, minimal side effects, and good pain relief. However, the sedation caused by butorphanol is also associated with adverse reactions, such as dizziness and drowsiness, during the recovery period, prolonging the patient’s recovery time. The incidence of adverse reactions is related to the dose, and the occurrence of adverse reactions can be significantly reduced by using an appropriate dose.
21
Propofol is widely used in gastroscopy because of its rapid onset, strong efficacy, and short recovery time. However, it is limited by pain during injection, instable respiration, and blood flow dynamics. Relevant studies demonstrated that the intravenous infusion of propofol at a dose ranging from 2 to 2.5 mg/kg can result in substantial reductions in SBP and DBP, with the reductions reaching 25% to 40%.
22
Moreover, the suppressive effect of the drug on the respiratory and circulatory systems progressively increases with increasing doses,
23
thus necessitating the concurrent use of other pharmacological agents. By combining several synergistic drugs, the therapeutic dose of each drug is reduced, and the effect is enhanced, both retaining the individual advantages of each drug and weakening their side effects.
19
Agarwal
Shun
The findings of the study revealed that the ED50 and ED95 of butorphanol in group A were 7.384 and 9.108 μg/kg, respectively. Meanwhile, these values were 6.657 and 8.419 μg/kg, respectively, in group B and 6.364 and 7.348 μg/kg, respectively, in group C. These results suggest that the ED50 and ED95 of the propofol–butorphanol combination for suppressing the response to gastroscope insertion tended to decrease with increasing age. During gastroscopy, all patients displayed comparatively stable hemodynamics. An anesthesiologist conducted follow-up by telephone 24 hours after the procedure, and no adverse symptoms were reported. This examination of the effective concentration of butorphanol in different age groups could lead to a more accurate treatment dose, reducing the respiratory and circulatory suppression caused by excessive propofol use and adverse reactions such as dizziness and drowsiness caused by excessive analgesic use. This study has significant implications for clinical guidance and patient safety.
This study had several limitations. First, the study was limited to gastroscopic examinations, and it did not extend to colonoscopies. Further research is required to determine whether the program can be applied to colonoscopy and whether the effective dose of propofol differs. Second, the study only included patients with ASA I or II and excluded high-risk patients (ASA III–IV). Consequently, the results and conclusions might not be generalizable to high-risk populations. Third, the study consisted of a small sample size from a single center. Finally, patients were not followed-up 24 hours later, and it was unclear whether there was any long-term effect.
When propofol was combined with butorphanol tartrate, the corresponding ED50 and ED95 for inhibiting the response to gastroscope insertion varied among different age groups. Therefore, the required dose might gradually decrease with age.
Footnotes
Acknowledgments
The authors would like to acknowledge the contribution of the Department of Anaesthesia and Endoscopy Room.
Author contributions
Yaorui Sun: Conceptualization; Investigation; Methodology; Writing – original draft.
Shujiao Liu: Formal analysis; Investigation; Writing – review & editing.
Wenli Si: Investigation; Formal analysis; Writing – review & editing.
Quanyi Zhang: Investigation; Methodology; Writing – review & editing.
Fei Yu: Investigation; Writing – review & editing.
Ming Su: Methodology; Writing – review & editing.
Chao Sun: Conceptualization; Formal analysis; Supervision; Writing – review & editing.
Data availability statement
The data of this study will be shared on reasonable request to the corresponding author.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
