Abstract
Eosinophilic gastroenteritis poses a significant diagnostic challenge, particularly in developing countries, where the awareness of this condition may be limited. Here, the case of a patient in her early 30s, who presented with recurrent episodes of abdominal pain and diarrhea, is reported. Initial standard laboratory investigations revealed normal complete blood counts and elevated total serum immunoglobulin E levels. Upper and lower endoscopic evaluations with systemic biopsies did not reveal any significant abnormalities. However, computed tomography revealed a thickened small intestine wall, halo signs, and mild ascites. Analysis of the ascitic fluid confirmed eosinophilia. These findings prompted a diagnosis of eosinophilic gastroenteritis. The patient responded well to a targeted elimination diet, corticosteroids, and antileukotriene medication. The present case emphasizes the importance of considering eosinophilic gastroenteritis in the differential diagnosis of patients who present with abdominal pain and eosinophilic ascites.
Introduction
Eosinophilic gastroenteritis is a rare idiopathic disease that may affect one or more digestive tract organs.1,2 The prevalence of eosinophilic gastroenteritis in the USA is estimated to range from 8.4 to 28 cases per 100 000 people, 1 with eosinophilic ascites being a manifestation of the serosal subtype.1,2 The prevalence of the serosal eosinophilic gastroenteritis type is the rarest, reported to be approximately 4.5–9% and 13% of all eosinophilic gastroenteritis types in Japan and the USA, respectively. 1 However, in Vietnam, with only rare and isolated reports of the disease, 3 this condition is underrated due to nonspecific manifestations, leading to potential misdiagnosis. Here, a case of eosinophilic gastroenteritis associated with eosinophilic ascites is presented, highlighting the importance of recognizing this condition in Vietnam.
Case report
A female patient in her early 30s presented at hospital with a 1-month history of recurring generalized abdominal pain, nausea, and frequent loose stools. She reported no experience of fever, night sweats, weight loss, arthralgia, skin rash, atopic disease, or food allergies. Upon first admission, she underwent upper and lower gastrointestinal endoscopies without biopsy, which revealed normal findings, and abdominal computed tomography (CT) scans did not show abnormalities (Figure 1(a) and (b)). The patient was initially diagnosed with gastroenteritis, and she was prescribed empiric antibiotics (500 mg ciprofloxacin, intravenously, twice daily for 5 days) and discharged. However, she only experienced a partial response. Four weeks later, the patient experienced similar symptoms that reappeared during 1 week and she returned to hospital. At the second hospital admission, her abdominal examination revealed mild tenderness in the epigastric region but no shifting dullness. The complete blood count showed eosinophils at 0.1 × 109 cells/L (normal range, 4–11 × 109 cells/L), while a peripheral blood smear revealed an absolute eosinophil count of 150 cells/µl. Total serum immunoglobulin (Ig)E was elevated at 370.4 IU/ml (normal range, <100 IU/ml). Serum levels of vitamin B12, thyroid stimulating hormone, free thyroxine, free triiodothyronine, and tryptase were all within normal ranges. Serological tests were negative for hepatitis B and hepatitis C, as well as parasitic infections (Toxocara, Strongyloides, Echinococcus, Taenia solium, Ascaris, Ancylostoma, Schistosoma, Trichinella, Entamoeba, Paragonimus, Angiostrongylus), interferon-gamma, HIV, cytomegalovirus antinuclear antibody, and antineutrophil cytoplasmic antibodies. Three consecutive stool tests for ova and parasites were negative. 2D echocardiography revealed no abnormalities. Abdominal ultrasound showed only mild ascites. Ascitic fluid analysis revealed a low serum albumin gradient with eosinophilia (total cells, 1.785 cells/µl, eosinophils 60%), adenosine deaminase 0.5 IU/L, bacterial culture, and malignant cytology (Figure 2). The second CT scan revealed diffuse concentric parietal thickening of the small bowel wall, diffuse circumferential wall thickening with postcontrast enhancement in multiple small intestine loops within the peritoneal cavity, associated intense peritoneal fat stranding, and the presence of halo signs (Figure 1(c) and (d)). Upper and lower gastrointestinal endoscopies with systemic esophagus, stomach, duodenum, and colon biopsies were unremarkable. After ruling out other causes, the diagnosis of eosinophilic ascites with enteritis was established based on eosinophilia of the ascitic fluid and infiltration in the serosal layers of the small intestines on CT. Serum-specific IgE antibodies were evaluated against common allergens using the radioallergosorbent method, and the results were positive only for house dust mites. The patient was prescribed 20 mg prednisolone, orally, once daily, and 10 mg montelukast, orally, once daily, for 2 weeks. Additionally, the patient was advised to try to identify the food allergen. After 2 weeks, her clinical symptoms improved, and no ascites were observed in a follow-up ultrasound. The prednisolone dose was gradually tapered over 2 weeks. However, when tapering down, the patient experienced symptoms similar to those previously after eating water chestnuts, with mild ascites observed in the ultrasound. As a result, the prednisolone dose was adjusted back up to 20 mg for 2 weeks, and a target elimination diet involving water chestnuts was implemented. The patient responded well and the prednisolone dose was tapered down after 4 months. The serial test results, treatment, and clinical response are summarized in Table 1.

Computed tomography (CT) scan images obtained during the first and second hospital admissions of a female patient in her early 30s who presented with recurring generalized abdominal pain, nausea, and frequent loose stools, showing: (a and b) no abnormalities on abdominal CT scans at first admission (coronal and axial planes, respectively); and (c and d) multiple areas of thickening of the small bowel from the D4 segment wall with halo signs (white arrows) and intense peritoneal fat stranding (white triangle) on abdominal and pelvic CT scans at second admission (coronal and axial planes, respectively), with evident accumulation of perihepatic ascitic fluid (dash line circle).
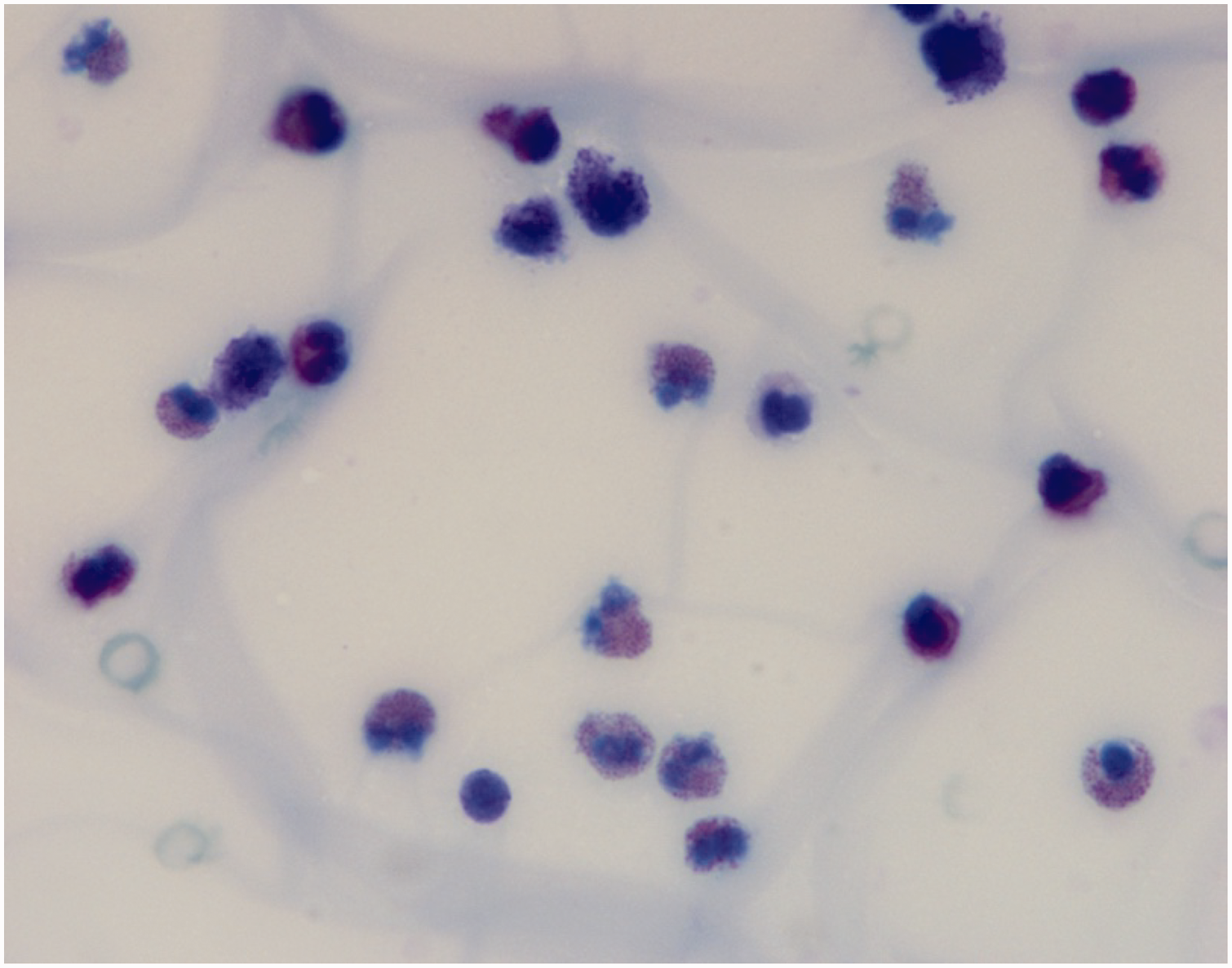
High-power oil immersion view of ascitic fluid from a female patient in her early 30s who presented with recurring generalized abdominal pain, nausea, and frequent loose stools, showing eosinophils containing eosinophilic granules and bilobed nuclei (hematoxylin and eosin stain; original magnification, ×60).
Serial results of eosinophil count, serum IgE levels, imaging findings, and treatment administered during the follow-up period in a female patient in her early 30s who presented with recurring generalized abdominal pain, nausea, and frequent loose stools.

AEC, absolute eosinophil count; CT, computed tomography; IgE, immunoglobulin E; NP, not performed.
Approval by the local ethics committee was not required as no prospective study was performed. The patient provided written informed consent for publication of the data and accompanying images for the present case report, and the manuscript was prepared and revised according to the CARE checklist (2013). 4
Discussion
Eosinophilic gastroenteritis is characterized by eosinophilic infiltration in the gastrointestinal tract, leading to inflammation without apparent cause, such as drug reactions, parasitic infections, or malignancy.1,2,5 Klein et al. 6 classified eosinophilic gastroenteritis into three subtypes–mucosal, muscular, or serosal–based on which intestinal layer exhibited eosinophilic infiltration. Eosinophilic gastroenteritis is strongly associated with atopic disease, peaks between 30 and 50 years of age, and has a slight male predominance. Eosinophilic infiltration of the gastrointestinal tract is primarily driven by T-helper type 2-dependent cytokines, particularly interleukin (IL)-5, and is induced by food allergy. Additionally, eotaxins, such as eotaxin-1 and eotaxin-3, play crucial roles in the migration of eosinophils towards tissues. The activation and degranulation of eosinophils lead to the release of cytotoxic proteins and other mediators, contributing to tissue damage and inflammation in the gastrointestinal tract. 7 The criteria for eosinophilic gastroenteritis include gastrointestinal symptoms, absence of parasitic or extraintestinal manifestations, and gastrointestinal tract biopsy showing eosinophilic infiltration, or characteristic radiological findings with peripheral eosinophilia or eosinophilic ascites.2,5,8 In the present case, eosinophilic infiltration may have solely infiltrated the small intestine from the D4 segment with predominant serosal involvement, showing mural thickening of the bowel loops. Consequently, multiple systemic biopsies from the esophagus, stomach, and D1 and D2 segments exhibited normal findings. The diagnosis relied on imaging findings and the presence of eosinophilia of the ascitic fluid. The differential diagnosis includes eosinophilic gastroenteritis, parasitic and fungal infections, hypereosinophilic syndrome, and less common causes, including eosinophilic pancreatitis, chronic eosinophilic leukemia, myelofibrosis, T-cell lymphoma, Churg-Strauss Syndrome, systemic lupus erythematosus, familial paroxysmal polyserositis, and Ménétrier’s disease.9–11 Particularly in tropical countries, occult parasitic infections should be ruled out before starting corticosteroid therapy to avoid exacerbating occult infections.10,11 Other common causes were thoroughly investigated in the present case before diagnosing eosinophilic gastroenteritis. The delayed diagnosis of this serosal subtype has been linked to factors such as the patient’s sex, lack of atopy history, detection of eosinophilic ascites in very small volumes that can only be collected via ultrasound-guided paracentesis, and nonspecific clinical and imaging findings during initial examinations. Notably, a normal peripheral blood eosinophil count is reported to be observed in only 10% of patients.8,12 Although laparoscopic serosal biopsies are required for a definite diagnosis, diagnostic enteroscopy or laparoscopy was not performed in the present case due to the nonspecific location in the CT scan image and the more invasive nature of these procedures compared with paracentesis. The patient’s excellent response to initial steroid therapy further supported the diagnosis. The treatment of eosinophilic gastroenteritis involves dietary restriction with a targeted elimination diet and steroids, followed by gradual reduction. This approach typically yields an excellent response and a good prognosis. Notably, the serosal subtype shows a better response to steroid therapy without a chronic course. 2 Alternative drugs include mast cell stabilizers, leukotriene receptor antagonists, anti-IgE monoclonal antibodies, anti-tumor necrosis factor, and anti-IL-5 monoclonal antibodies in steroid-resistant patients.1,2,7,8 In addition, dietary restriction may help identify and eliminate potential food triggers contributing to gastrointestinal inflammation. However, the clinical usefulness of allergy testing for guidance is contentious due to the likely combination of IgE- and non-IgE-mediated hypersensitivity, and the limitations of available food allergen testing in Vietnam. 8 The patient in the present case was able to identify and eliminate the trigger food (water chestnut) on her own, rather than relying on serum-specific IgE antibodies.
Despite its rarity in tropical regions, eosinophilic gastroenteritis should be considered in patients with unexplained eosinophilic ascites, even without peripheral blood eosinophilia. Prompt diagnosis and early initiation of appropriate therapy with corticosteroids and montelukast may lead to excellent results.
Summary
Eosinophilic gastroenteritis can manifest as mild ascites even when peripheral eosinophil counts are normal. In low-resource settings where enteroscopy might be unavailable, a less invasive approach involving evaluation of ascitic fluid for eosinophilic enteritis and clinical response monitoring may be more feasible. Comprehensive evaluations are critical to rule out secondary causes, particularly occult parasitic infections prevalent in tropical regions. A targeted elimination diet should be considered to identify specific food allergens among a wide range of foods, as there is a lack of evidence supporting the efficacy of allergen tests.
Footnotes
Acknowledgments
The authors thank the staff of the Department of Gastroenterology, Cho Ray Hospital for approving this case report.
Author contributions
TMH, NTVH, and TTLV were the patient’s physicians in charge; TMH, NTVH, and DLP reviewed the literature and contributed to manuscript drafting; and PTH and DTQ reviewed and criticized the manuscript. All authors approved the final version to be submitted.
Data availability statement
The original clinical datasets generated during the case are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
