Abstract
We herein report two cases of post-treatment maturation of medulloblastoma (MB). We also conducted a literature review to summarize the clinical and pathologic features of MB maturation. From January 1992 to February 2017, 52 patients with MB underwent surgical resection followed by radiotherapy and chemotherapy. Tumor cell maturation was identified in two patients who underwent a second surgery. We conducted a systematic search of PubMed and identified six such cases. In both of our patients, the pathologic type was MB with extensive nodularity (MBEN). Both patients underwent radiotherapy and chemotherapy. The tumor differentiated to gangliocytoma in both patients. In the overall analysis that also included the six cases identified in the literature, the pathologic types were classic MB (n=1), desmoplastic/nodular MB (n=2), MBEN (n=3), and unclassified MB (n=2). MB differentiated into the following types: gangliocytoma (n=2), ganglioglioma (n=1), melanocyte (n=1), neuronal differentiation (n=2), and classic MB (n=1). Desmoplastic/nodular MB and MBEN can differentiate into less malignant cells types after radiotherapy and chemotherapy. Maturation of MB may be affected radiotherapy and chemotherapy.
Introduction
Medulloblastoma (MB) is the most common primary malignant brain tumor in children. 1 Under the current World Health Organization (WHO) classification, MB is categorized into four distinct variants: classic MB (CMB), desmoplastic/nodular MB (DMB), MB with extensive nodularity (MBEN), and large cell/anaplastic MB. 2 Treatment options include surgical resection, chemotherapy, and radiotherapy. Patient survival is affected by several factors, including the disease stage upon diagnosis, the extent of surgical removal, adjuvant therapy, and the pathologic type. We herein report two cases of pediatric MB with post-treatment maturation. We also conducted an extensive search of the literature to identify similar cases and herein discuss the pathologic maturation of MB, chemotherapy, radiotherapy, and prognosis. These cases are being reported to provide surgeons and oncologists with an understanding of the treatment of MB in clinical practice.
Materials and methods
We retrospectively reviewed the medical records of all patients who received treatment for MB in our institution from January 1992 to February 2017. We also searched the published literature in PubMed (January 1990–June 2017) for reports of the result of MB maturation. The search keywords were “medulloblastoma,” “differentiation,” “maturation,” “gangliocytoma,” “ganglioglioma,” and “neuron.” Publications meeting all of the following criteria were included in the final review: patient age of ≤14 years; pathologic diagnosis of MB using a surgically resected specimen; and, if a second pathological diagnosis was not available, elimination of MB with recurrence or distant metastasis. The following clinical data were collected: age at symptom onset, sex, treatment prior to diagnosis, specific diagnosis, sequence of treatments performed, time of MB maturation, result of MB maturation, follow-up, and outcome. The references of all identified articles were also manually searched for additional studies.
No ethics committee review was required for this study. Verbal informed consent was obtained from the patients’ parents.
Results
Case identification
From January 1992 to February 2017, 52 patients with MB underwent treatment at our department. Post-treatment maturation was identified in two patients (Tables 1 and 2).
Clinical data of patients with medulloblastoma with multiple differentiation during comprehensive therapy in the literature (n=6) and in the current report (n=2).

MB: medulloblastoma; DMB: desmoplastic/nodular medulloblastoma; SHH: sonic hedgehog subgroup; CMB: classic medulloblastoma; MBEN: medulloblastoma with extensive nodularity; N/NM: none or not mentioned; F: female; M: male.
Comprehensive therapy of patients in the literature (n=6) and in the current report (n=2).

NM: not mentioned.
Case 1
A 13-month-old girl presented with a 3-day history of vomiting and somnolence. Her clinical examination findings were normal. Laboratory investigation showed electrolyte imbalance. Computed tomography of the brain showed a 5-cm heterogeneous lesion in the posterior fossa. The fourth ventricle was not visible, and the third and lateral ventricles were dilated. External ventricular drainage was performed to control the patient’s symptoms. Magnetic resonance imaging (MRI) revealed a 5-cm lesion in the right cerebellar hemisphere (Figure 1(a)). Subtotal cerebellectomy was performed because of the high vascularity of the mass. Pathologic examination showed an MBEN (Figure 1(c)) with the following immunohistochemical features: synaptophysin (++), neurofilament (−), neuron-specific enolase (++), CD57 (+), glial fibrillary acidic protein (+), NeuN (++), vimentin (+), and Ki67 (+) (85%). She was treated first with chemotherapy using the protocol described by Rutkowski et al. 3 The chemotherapy drugs were cyclophosphamide (800 mg/m2/d), cisplatin (75 mg/m2/d), and vincristine (1.5 mg/m2/d). The patient developed common post-chemotherapy responses such as alopecia and myelosuppression. She underwent secondary radiotherapy (craniospinal irradiation at a dose of 36 Gy in 20 fractions followed by posterior fossa boost at 16 Gy in 11 fractions). Thirty months after the first surgery, a repeat MRI scan showed a 3-cm residual mass in the posterior fossa (Figure 1(b)). The patient underwent microsurgical tumor resection. Pathologic examination showed gangliocytoma cells in scattered clusters on a background of neurogliocytes (Figure 1(d)). At the last follow-up 6 years later, the patient was healthy.
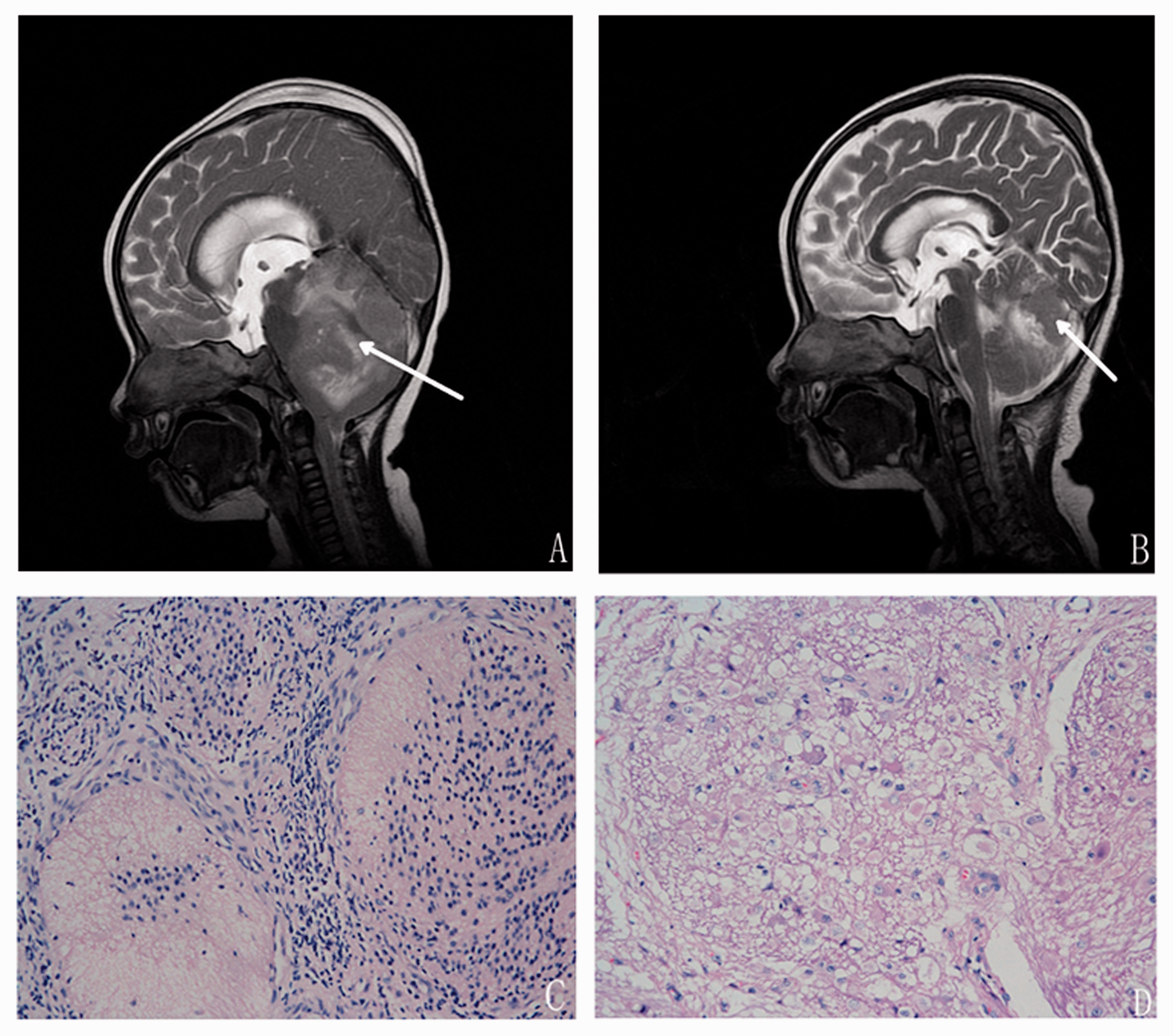
Imaging and pathological findings of Patient 7. (a) Preoperative imaging. T2-weighted magnetic resonance imaging (MRI) shows a mixed-signal mass, approximately 5 cm in size, in the right cerebellar hemisphere. (b) Imaging obtained after partial resection and chemotherapy and radiotherapy. T2-weighted MRI shows a hypointense mass, approximately 2 cm in size, in the original location. (c) First pathological examination. Medulloblastoma with extensive nodularity contains a nodular area composed of small cells (hematoxylin and eosin, 200×). (d) Pathological examination after comprehensive therapy. Gangliocytoma cells are present in scattered clusters on a background of neurogliocytes (hematoxylin and eosin, 200×).
Case 2
A 2-year-old girl presented with a 20-day history of headache and a 4-day history of vomiting and somnolence. Physical examination revealed an ataxic gait. Laboratory examination showed no abnormalities. MRI revealed a 4.5-cm mass with enhanced signals in the left cerebellar hemisphere (Figure 2(a)). Pathologic examination after subtotal tumor resection revealed an MBEN (Figure 2(c)). She then underwent systemic chemotherapy with cyclophosphamide (800 mg/m2/d), cisplatin (75 mg/m2/d), and vincristine (1.5 mg/m2/d) according to the protocol described by Rutkowski et al. 3 Repeat MRI 6 months later revealed a relapsing lesion in the cerebellar hemisphere (Figure 2(b)), but the patient had no neurologic deficits. Pathologic examination of the resected tumor revealed morphological features characteristic of gangliocytoma (Figure 2(d)). The patient underwent craniospinal radiation (36 Gy in 18 fractions) followed by boost to the posterior fossa (19.8 Gy in 11 fractions). The patient was alive and well upon the last visit 3 years later.
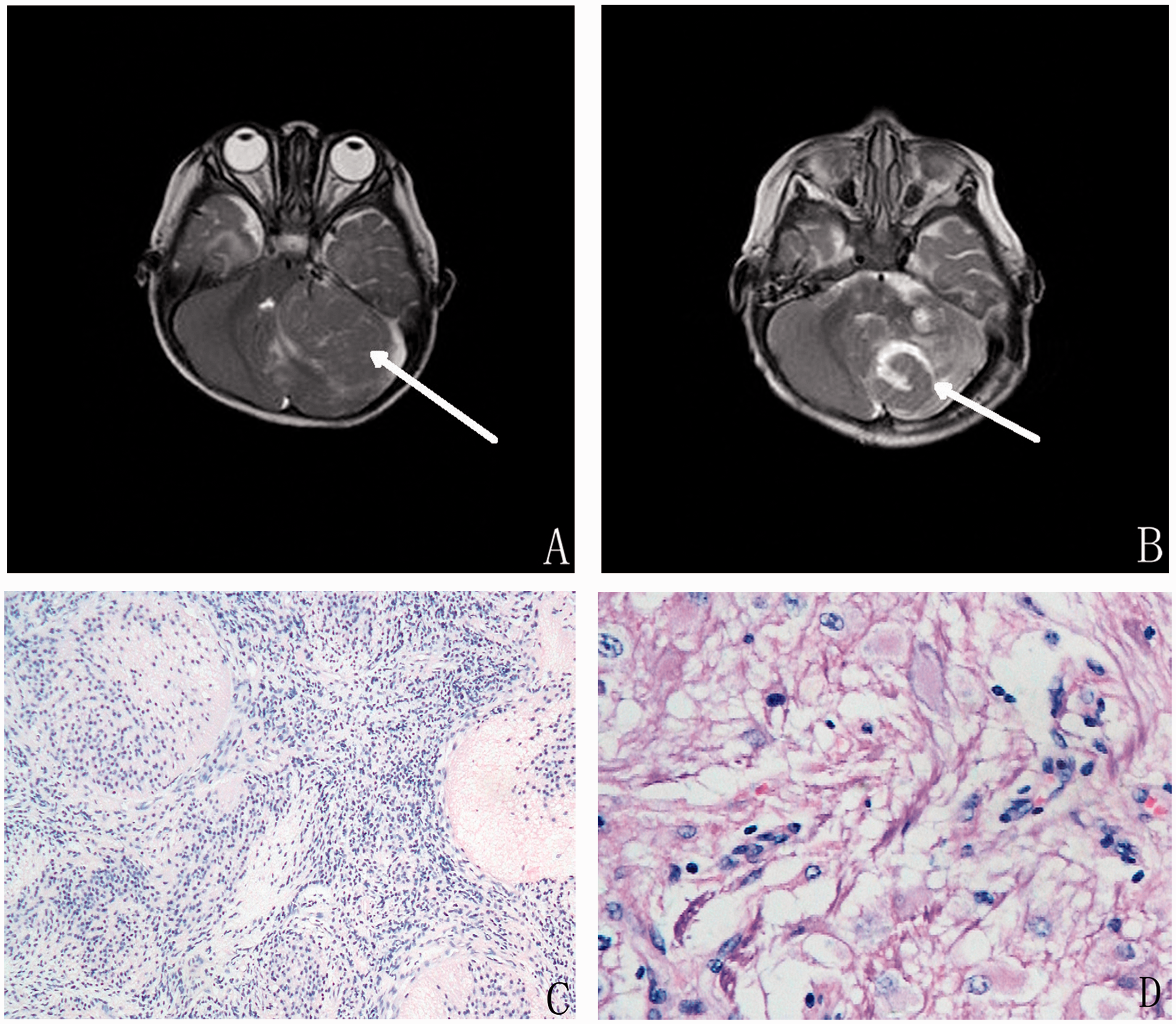
Imaging and pathological findings of Patient 8. (a) Preoperative imaging. T2-weighted magnetic resonance imaging (MRI) shows a hypointense mass, approximately 4.5 cm in size, in the left cerebellar hemisphere. (b) Imaging obtained after subtotal resection and chemotherapy and radiotherapy. T2-weighted MRI shows a new hypointense mass, approximately 1 cm in size, in the original location. (c) First pathological examination. Medulloblastoma with extensive nodularity contains a nodular area composed of small cells in continuity with the fibrillary region (hematoxylin and eosin, 100×). (d) Pathological examination after comprehensive therapy. Mature ganglion cells are seen in clusters on an astrocytic background (hematoxylin and eosin, 400×).
Literature search
We identified five publications that reported six cases of histologically confirmed MB with post-treatment maturation (4 boys, 2 girls) (Tables 1 and 2).4–8 The patients’ mean age was 7.8 years (range, 3 months to 11 years). The MB subtypes were DMB (n=2), MBEN (n=1), CMB (n=1), and unclassified (n=2). Maturation occurred after combined radiotherapy and chemotherapy in five patients and after chemotherapy alone in the remaining patient. MB differentiated into the following types: gangliocytoma (n=2), ganglioglioma (n=1), melanocyte (n=1), neuronal differentiation (n=2), and CMB (n=1). The duration from pathologic verification of the initial MB to maturation ranged from 3 months to 11 years. Two patients died, and one survived for >11 years.
Discussion
The annual incidence of childhood central nervous system tumors is estimated at 5 per 100,000 children. 9 MB is the most common malignant central nervous system tumor in childhood and is 10 times more likely to be diagnosed in children than in adults. 9 MB is accompanied by headache, ataxia, high intracranial pressure, brain stem dysfunction, or nerve root or spinal cord compression caused by a space-occupying lesion. In general, MB is highly invasive and prone to metastasis. Maturation of MB has been reported, but only as scattered case reports. We have herein presented two cases of MB maturation at our institution and described six additional cases identified in a literature search.
Under the WHO classification, MB is categorized into four types: CMB, DMB, MBEN, and large cell/anaplastic MB. 2 DMB contains nodular, reticulin-free zones characterized by neuronal maturation. The reticulin-free zones have a reduced nuclear:cytoplasmic ratio and contain fibrillary matrix cells with a neuronal appearance. These nodules are surrounded by dense, mitotically active cells that produce a dense intercellular reticulin-positive network of fibers. MBEN accounts for <3% of all cases of MB and is often seen in children aged ≤3 years. 1 MBEN has a relatively good prognosis. Compared with DMB, MBEN has an expanded lobular architecture: the reticulin-free zone is typically elongated and rich in neuropil-like tissue. The reticulin-free zone also contains small cells that resemble the cells of central neurocytoma. The internodular component is reduced in some areas. In a study of 82 patients, Garre et al. 10 showed that MBEN was present in 15% (12/82) and that DMB was present in 12% (10/82); additionally, MBEN was associated with Gorlin syndrome in 5 of 12 patients. Their study showed an association of both Gorlin syndrome-associated MBEN and sporadic DMB with mutations in the PTCH1, SMOH, and SUFUH genes.11–13 Their study also indicated that MBEN and DMB have the potential for maturation. In both of our cases, the tumors that eventually matured were MBEN upon the first surgery. In the overall analysis that also included the six cases found in the literature search, DMB and MBEN accounted for 62.5% (5/8) of all cases and showed a high trend toward differentiation and maturation. MB differentiated into the following types: gangliocytoma (n=2), ganglioglioma (n=1), melanocyte (n=1), neuronal differentiation (n=2), and CMB (n=1). Gangliocytoma (WHO grade I) is well-demarcated and usually consists of large, mature neurons with a small glial component.10,14 Ganglioglioma (WHO grade I–III) is a rare tumor composed of both neuronal and glial components and characterized by large, mature neoplastic neurons; the glial component consists of astrocytic or oligodendroglial cells. 15 Gangliocytoma (n=2), ganglioglioma (n=1), and neuronal cells (n=2) are less malignant than MB; thus, DMB and MBEN could differentiate into less malignant cell types.
Differentiation of MB could be the result of radiotherapy and chemotherapy or may represent intrinsic properties of the MB. 16 Bernert et al. 17 reported a pathologically confirmed case of ganglioglioma differentiation with neither radiotherapy nor chemotherapy. Crawford and Levy 18 also reported a case of myogenic differentiation without adjuvant therapy, indicating an intrinsic potential for differentiation.
Adjuvant therapy, including chemotherapy and radiotherapy, varies based on the pathologic and molecular classifications.1,19 Chemotherapy is an important part of postoperative treatment of MB. In certain cases, chemotherapy could spare the patients from irradiation and the detrimental effects of irradiation on development. When adequate resection is not possible, chemotherapy should be guided by pathologic and molecular typing. The chemotherapy after radiotherapy is a cisplatin-based regimen that is administered for four to nine cycles. 20 The chemotherapy includes induction chemotherapy and consolidation chemotherapy. 5 Induction chemotherapy typically consists of cisplatin, vincristine, and cyclophosphamide in combination with mesna and etoposide. Consolidation chemotherapy includes high-dose carboplatin and thiotepa. Salet et al. 21 reported a case of Ewing sarcoma that differentiated into ganglioneuroblastoma and displayed neuronal maturation after chemotherapy. In our overall analysis that included the two patients at our institution and the six cases identified in the literature search, all patients received chemotherapy, suggesting that chemotherapy could affect maturation of MB cells.
Radiotherapy is the mainstay of treatment in patients aged >3 years. The regimen involves craniospinal irradiation at a total dose of 23.4 Gy followed by local boost to the posterior fossa at a total dose of up to 54.0 to 55.8 Gy. 22 Patients aged <3 years undergo chemotherapy first in an effort to avoid or at least postpone radiation, but this may decrease survival. 23 Stokman et al. 24 quantitatively evaluated radiotherapy-induced oral mucositis and found that epithelial cell maturity shifted from immature to mature due to radiotherapy. Previous studies have indicated that irradiation can affect cell maturation. In the current study, seven of eight patients (87.5%) underwent radiotherapy, suggesting that radiotherapy may affect mutation of MB cells. A variety of attempts have been made to reduce irradiation dosages.25,26 Large doses of radiotherapy may kill tumor cells but could also lead to mutation of tumor cells and further differentiation. 27 Molecular subgrouping has already been applied to clinical trials involving a variety of adjuvant therapy schemes, and this classic scheme is likely to be modified or supplanted by a scheme based on molecular subgroups. 28
MB is a highly malignant brain tumor with a poor prognosis in children. In a European study from 2000 to 2007, the 1-, 3-, and 5-year survival rates of pediatric patients with MB were 81%, 63%, and 56%, respectively. 29 The prognosis is the worst in infants and improves with increasing age. 29 Survival has improved since the adoption of molecular classification. Schwalbe et al. 30 stratified patients with MB into those with favorable risk, standard risk, high risk, and very high risk, and the corresponding 5-year progression-free survival rates were 91%, 81%, 42%, and 28%, respectively. In the current study, our literature review showed that one patient remained alive for >11 years, and the two patients in our institution survived for >2 and >6 years, respectively. In these two cases, MB differentiated into less malignant gangliocytoma after radiotherapy and chemotherapy. We are continuing to follow up both patients.
Conclusion
DMB and MBEN are more likely to differentiate into less malignant cell types. Maturation of MB may be affected by radiotherapy and chemotherapy.
Footnotes
Acknowledgement
We are grateful to Dr. Jin Zhu, Department of Pathology, and Dr. Yong Qin, Department of Radiology, for providing the relevant figures for these cases.
Disclosure of conflict of interest
The authors declare that there is no conflict of interest.
Funding
This research was funded by the Key Project of the Chongqing Natural Science Foundation (cstc2015jcyjbx0144).
