Abstract
Objective
To investigate the differences in autonomic nervous system (ANS) dysfunction, arterial stiffness, and the degree of delay in post-exercise heart rate recovery (HRR) according to the level of spinal cord injury (SCI), and propose preventive measures against cardiovascular diseases after SCI.
Methods
This retrospective study included 51 patients with SCI. Based on the neurological level of injury (NLI), patients were divided into two groups: Group A (NLI at and above T6) and Group B (NLI below T6). To assess ANS dysfunction, the head-up tilt test and 24-hour ambulatory blood pressure monitoring were conducted. Arterial stiffness was measured using the pulse wave velocity test. The exercise tolerance test was conducted to measure post-exercise HRR.
Results
Group A had significantly higher values in the head-up tilt test and 24-hour ambulatory blood pressure monitoring. In the pulse wave velocity test, both sides (left and right) had significantly higher values in Group B. One minute after the exercise tolerance test, Group A had significantly slower HRR (18.8 ± 11.1 beats/minute) than Group B.
Conclusion
Understanding the impact of ANS dysfunction and arterial stiffness on HRR in SCI according to NLI may provide insights for clinical management and preventative strategies for cardiovascular diseases.
Keywords
Introduction
Cardiovascular disease (CVD) is a leading cause of death worldwide, and spinal cord injury (SCI) has emerged as a recognized risk factor for CVD. 1 In the general population, hypertension, diabetes, dyslipidemia, and smoking can influence CVD. 2 However, for patients with SCI, physical inactivity, obesity, changes in body composition, and delayed heart rate recovery (HRR) after exercise are also considered risk factors, in addition to the previously known risk factors for CVD.3,4 Post-exercise HRR indicates the rate at which the heart rate decreases in the first few minutes after stopping exercise. Clinically, post-exercise HRR has been extensively studied in healthy adults. When adjusted for other risk factors, such as dyslipidemia, it has been identified as an independent risk factor for CVD, 5 and is also a predictive factor for the occurrence of vascular diseases, such as stroke (including CVD). 6 In previous studies of healthy or asymptomatic adults with no history of heart failure, an HRR < 12 beats/minute at 1 minute after exercise is associated with an increased risk of death, and a causal relationship between post-exercise HRR and CVD has been demonstrated.5,7 Post-exercise HRR is influenced by both the autonomic nervous system and peripheral arterial stiffness.8,9 In patients with SCI, these two factors often function abnormally, thereby affecting heart rate.
When SCI occurs, autonomic nervous system function can be disrupted, resulting in cardiovascular complications such as orthostatic hypotension and autonomic dysreflexia. 10 In addition, autonomic nervous system damage can delay HRR in patients with SCI. 11 Early intervention for the prevention and management of complications in these patients may therefore be feasible. This may involve evaluating orthostatic hypotension and autonomic dysreflexia to identify autonomic nervous system damage, and investigating the degree of delay in HRR. This approach would allow for the early identification of patients at a high risk of CVD.
In patients with SCI, arterial stiffness delays HRR. 12 Although arterial stiffness is not a fatal condition, it can lead to coronary artery disease and stroke. 13 When arteries become stiff, vascular elasticity decreases, which then reduces coronary artery perfusion and increases the myocardial oxygen demand. 14 This causes arteriosclerosis and delays post-exercise HRR. 15 The evaluation of arterial stiffness in patients with SCI may therefore help to identify those at high risk of CVD, thus allowing for early intervention to prevent and manage complications.
Autonomic nervous system dysfunction has been widely reported in patients with cervical and upper thoracic SCI. 16 These patients experience symptoms such as dizziness, visual disturbances, and syncope as a result of orthostatic hypotension, as well as headache, sweating, anxiety, and worry as a result of autonomic dysreflexia. In general, patients with injuries below the lower thoracic spinal cord have a greater degree of arterial stiffness than those with injuries to the cervical or upper thoracic spinal cord. 17 The incidence rate of autonomic nervous system dysfunction and the degree of arterial stiffness thus vary according to the site of SCI.
The association between the incidence of autonomic nervous system dysfunction or the degree of arterial stiffness and HRR remains unclear; however, it is hypothesized that autonomic nervous system dysfunction and arterial stiffness have different effects on HRR depending on the level of SCI. In a previous study, HRR in the first 30 s after exercise was almost completely promoted by the sudden vagal reactivation that begins immediately after exercise. 18 Furthermore, a linear regression analysis revealed an association between HRR and increased arterial stiffness. 9 Autonomic nervous system dysfunction and increased arterial stiffness are common in SCI patients. However, autonomic nervous system dysfunction is more common in cervical and upper thoracic SCI patients, whereas increased arterial stiffness is more common in lower thoracic SCI patients. This can make it confusing to understand the relationship between HRR and the neurological level of injury (NLI) in SCI patients. In the present study, we therefore aimed to categorize patients based on the NLI relative to the sixth thoracic vertebrae (T6), to investigate the occurrence of autonomic nervous system dysfunction and differences in arterial stiffness on HRR, and to propose appropriate methods for preventing CVD in each patient group.
Patients and methods
Patients
This was a retrospective study that included patients who received treatment for SCI at the Department of Rehabilitation Medicine, Wonkwang University Hospital, between March 2018 and February 2022. The inclusion criteria were as follows: 1) patients diagnosed with SCI through radiological or electrodiagnostic tests; and 2) patients within the first 6 months after onset. The exclusion criteria were as follows: 1) patients who were unable to receive rehabilitation because of comorbidities such as pneumonia, urinary tract infections, or unstable neurological conditions; and 2) patients taking medications, such as beta-blockers or non-dihydropyridine calcium channel blockers, that can affect heart rate. All patient details have been de-identified, and all procedures were conducted in compliance with the relevant regulations. Approval was obtained from the Institutional Review Board of Wonkwang University Hospital (IRB Number: WKUH 2018-07-008-011).
Methods
To assess the general characteristics of the patients, demographic characteristics such as age, sex, type of SCI, and NLI were investigated. The patients were divided into two groups based on NLI: Group A (patients with an NLI at or above T6) and Group B (patients with an NLI below T6). To assess the presence of autonomic nervous system dysfunction, the head-up tilt test and 24-hour ambulatory blood pressure monitoring were performed. To measure arterial stiffness, pulse wave velocity (PWV) was measured. Furthermore, to measure post-exercise heart rate, patients exercised using an arm ergometer after 3 weeks of sufficient training, and an exercise tolerance test was performed during week 3.
The reporting of the present study conforms to Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 19
Evaluation of autonomic nervous system function
Head-up tilt test
To evaluate orthostatic hypotension, the head-up tilt test was performed. Before the test, the patient rested in the supine position for 5 minutes, and the baseline systolic and diastolic blood pressures (SBP and DBP, respectively) and heart rate were measured. Subsequently, the tilt table was raised to a 60° angle. After maintaining this position for 3 minutes, the blood pressure and heart rate were measured again. Orthostatic hypotension was defined as a decrease in SBP of more than 20 mmHg or a decrease in DBP of more than 10 mmHg compared with the baseline blood pressure, after 3 minutes of tilting.20,21 If the patient experienced symptoms such as dizziness, sweating, or syncope during the test and was unable to maintain the position for 3 minutes, their vital signs were immediately monitored, and they were considered to have orthostatic hypotension if they had a decrease in SBP of more than 20 mmHg or a decrease in DBP of more than 10 mmHg compared with the baseline blood pressure.
Twenty-four-hour ambulatory blood pressure monitoring
To assess autonomic dysreflexia, 24-hour ambulatory blood pressure monitoring was conducted. The BR 102 plus device (Schiller-Reomed AG, Obfelden, Switzerland) was attached to each patient’s nondominant upper limb using an appropriate blood pressure cuff. Blood pressure and heart rate were then measured for 24 hours, and the data were analyzed using MT-300 software (Schiller-Reomed AG). Daytime was defined as the period from 7 a.m. to 7 p.m., and nighttime was defined as the other 12 hours. SBP, DBP, and heart rate were measured every 30 minutes, and if the patient experienced symptoms of autonomic dysreflexia (such as headaches, sweating, or anxiety), they were instructed to press a button to record the event. Autonomic dysreflexia was defined by an increase in SBP of more than 20 mmHg compared with the baseline blood pressure.22,23 Baseline blood pressure was determined as the average blood pressure value measured during the daytime and nighttime. The frequency of autonomic dysreflexia was measured as the total number of occurrences throughout the monitoring period.
Evaluation of arterial stiffness
Arterial stiffness was measured in both the upper arms and ankles in a supine position, using a pulse wave meter (VP1000, Colin Co., Komaki, Japan). Participants had to stay in bed for 5 minutes before the measurement, for stabilization purposes. PWV was used to measure arterial stiffness using the formula baPWV = (L/Δt) × 0.8, where baPWV is the brachial-ankle PWV, L is the distance between the upper arm and the ankle (m), and Δt is the difference between the time at which the ascending wave appeared in the brachial and tibial arteries. The coefficient 0.8 was used to adjust for the difference between the actual path length and the straight-line distance between the two measurement points. Arterial stiffness was considered abnormal when baPWV ≥ 1750 cm/s, and this was used as a criterion for evaluating the risk of CVD. 24
Exercise tolerance test and evaluation of post-exercise HRR
To measure post-exercise HRR, an exercise tolerance test was conducted using an arm ergometer (Ergometrics 800; Ergoline, Bitz, Germany; or Monark Rehab Trainer 881E; Monark, Varberg, Sweden). The patients sat in a posture similar to that when riding a bicycle and rotated the handle in a circular motion, gradually increasing the load. The basic load of the exercise tolerance test was 10 W, and the test was performed at the same level regardless of the NLI and regardless of whether the patient had experienced a complete or incomplete injury. The exercise intensity was increased passively; that is, rather than the intensity being controlled by technologies such as computers, the patients themselves gradually increased their intensity until they reached their maximum capacity. The height of the arm ergometer was adjusted to match each patient’s shoulder height, and patients who had difficulty gripping the handle because of limb paralysis used gloves to secure their hands to the handle during the test. The target exercise time was 8 to 12 minutes, and patients were recommended to maintain a speed of 40 rpm. The increase in exercise load was determined individually for each patient, and the test continued until a patient reached their limit. During the test, a 12-lead electrocardiograph was attached to record each patient’s electrocardiogram.
During the test, the electrocardiogram machine provided a continuous record of the heart rate. 25 The exercise tolerance test provided cardiopulmonary exercise parameters, including the respiratory exchange ratio (RER), maximal oxygen consumption (V̇O2Peak), minute ventilation (VE), metabolic equivalents (MET), and W values. 26
Post-exercise HRR was calculated as the difference in heart rate between the maximal heart rate during exercise and the heart rate at different time points after exercise (1, 3, 5, or 8 minutes), as follows:
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, version 26.0 (IBM Corp., Armonk, NY, USA). Independent sample t-tests were used to compare continuous variables between patient groups, such as general characteristics, autonomic nervous system dysfunction, and arterial stiffness. Chi-squared tests were used for categorical variables. Independent-sample t-tests were used to compare the degree of HRR at 1, 3, 5, and 8 minutes between the two groups. The normality of distribution was confirmed using the Shapiro–Wilk test. The Bonferroni procedure was used to perform post hoc comparisons between groups. P < 0.5 was taken to indicate significance.
Results
Demographic characteristics
In the present study, 102 patients participated. After applying the exclusion criteria, the final sample comprised 25 patients in Group A and 26 patients in Group B (Figure 1). The initiation of rehabilitation therapy after injury occurred at 30.4 ± 7.6 days for Group A and at 33 ± 9.3 days for Group B, with no significant differences in sex, type of injury, NLI, or any other general characteristics between the two groups (Table 1).

Data extraction process. ETT, exercise tolerance test; HR, heart rate; NLI, neurological level of injury; RER, respiratory exchange ratio; RM, rehabilitation medicine. Presence of factors affecting HR: patients taking medications (such as beta-blockers or non-dihydropyridine calcium channel blockers) that can affect heart rate. Patients who cannot complete rehabilitation program: patients who were unable to receive rehabilitation because of comorbidities such as pneumonia, urinary tract infections, or unstable neurological conditions.
General characteristics of patients with spinal cord injury.

AIS, American Spinal Injury Association Impairment Scale.
Group A, level of injury at or above T6; Group B, level of injury below T6.
Autonomic nervous system dysfunction
The head-up tilt test revealed that 15 (60%) patients in Group A and six (23.1%) patients in Group B had orthostatic hypotension. The results of 24-hour ambulatory blood pressure monitoring showed that 11 (44%) patients in Group A and four (15.4%) patients in Group B had autonomic dysreflexia. These differences in prevalence between the two groups were significant (orthostatic hypotension, P = 0.02; autonomic dysreflexia, P = 0.03; Table 2).
Results of autonomic nervous system dysfunction and mean arterial stiffness.

baPWV, brachial-ankle pulse wave velocity; Lt, left; Rt, right.
Group A, level of injury at or above T6; Group B, level of injury below T6; *P < 0.05.
Comparison of mean arterial stiffness
The PWV test revealed that in Group A, the right side had a velocity of 1691.0 ± 460.2 m/s, and the left side had a velocity of 1746.3 ± 502.4 m/s. In Group B, the right side had a velocity of 2216.2 ± 810.0 m/s, and the left side had a velocity of 2215.5 ± 827.5 m/s. Group B had significantly higher PWV on both the left and right sides compared with Group A (right side, P < 0.01; left side, P = 0.02; Table 2), indicating greater mean arterial stiffness in Group B than in Group A.
Exercise tolerance test
Resting heart rate before the exercise tolerance test was 80.5 ± 15.2 beats/minute in Group A and 86.7 ± 12.7 beats/minute in Group B, with no significant difference between the two groups (Table 3). Resting blood pressure was 111.7 ± 17.6 mmHg/78.0 ± 14.4 mmHg in Group A and 123.8 ± 20.1 mmHg/78.7 ± 11.6 mmHg in Group B. Although there was a significant difference in SBP between the two groups (P = 0.03; Table 3), there was no significant difference in DBP (Table 3).
Results of the exercise tolerance test (resting and exercising).

BP, blood pressure; HR, heat rate; MET, metabolic equivalents; RER, respiratory exchange ratio; VE, minute ventilation; V̇O2Peak, maximal oxygen consumption.
Group A, level of injury at or above T6; Group B, level of injury below T6; *P < 0.05.
After the exercise tolerance test, the maximal heart rate was 121.4 ± 26.1 beats/minute in Group A and 128.8 ± 27.3 beats/minute in Group B. Blood pressure values in the two groups were 137.8 ± 31.6 mmHg/116.5 ± 19.3 mmHg and 144.8 ± 30.1 mmHg/124.3 ± 22.0 mmHg, respectively. There were no significant differences in the maximal heart rate, SBP, or DBP between the two groups. In the exercise tolerance test, cardiopulmonary exercise parameters—including the RER, V̇O2Peak, VE, MEs, and W values—were not significantly different between the groups (Table 3).
HRR over time
One minute after the exercise tolerance test, Group A had an HRR of 18.8 ± 11.1 beats/minute, whereas Group B had an HRR of 26.4 ± 15.2 beats/minute. At 3, 5, and 8 minutes after the test, Group A had HRRs of 29.1 ± 14.9, 32.3 ± 16.8, and 35.0 ± 18.5 beats/minute, respectively, and Group B had HRRs of 33.5 ± 16.1, 35.5 ± 16.9, and 38.4 ± 20.2 beats/minute, respectively. Although there was a significant difference in HRRs at 1 minute after the exercise tolerance test (P = 0.04), there were no significant differences in HRRs at 3, 5, or 8 minutes after the test (Table 4).
Post-exercise HRR.
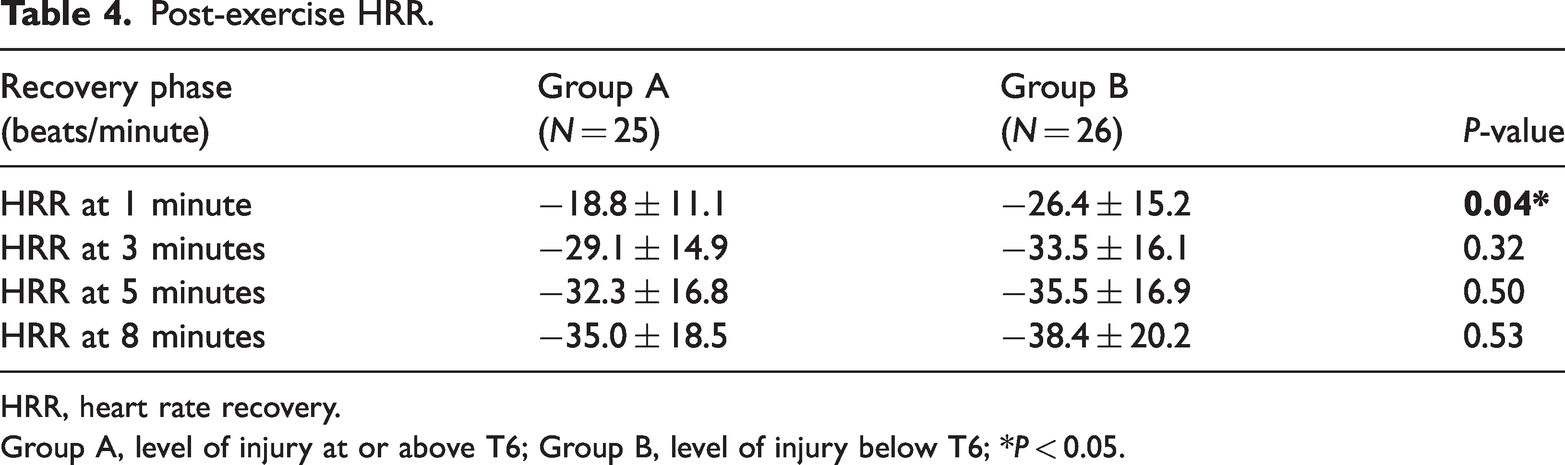
HRR, heart rate recovery.
Group A, level of injury at or above T6; Group B, level of injury below T6; *P < 0.05.
Discussion
The autonomic nervous system is the most important factor influencing heart rate. The autonomic nervous system contains two interacting systems, and sympathetic and parasympathetic neurons exert antagonistic effects on the heart. The sympathetic nervous system releases norepinephrine, which increases the heart rate and myocardial contractility. By contrast, the parasympathetic nervous system releases acetylcholine, which decreases the heart rate. 27 In previous studies, an abnormal HRR was identified as a predictive factor for autonomic nervous system dysfunction. Compared with other risk factors, abnormal HRR associated with autonomic nervous system dysfunction has been suggested to have a greater impact on CVD development. 28 Another study has reported that individuals with autonomic nervous system dysfunction experience delayed post-exercise HRR compared with those without autonomic nervous system dysfunction. The authors concluded that this may be because autonomic nervous system dysfunction can reduce the ability of the cardiovascular system to adapt to the needs of exercise, thus reducing venous return to the heart, which can then reduce cardiac output during exercise and delay post-exercise HRR. 7 Another important factor influencing HRR is arterial stiffness. This refers to a condition in which the elasticity of the arterial walls decreases or the rigidity increases, making it difficult for the arteries to expand and contract with each heartbeat. Consequently, the ability to effectively circulate blood during exercise is reduced, leading to a delay in HRR. 9 An increase in arterial stiffness thus results in decreased vascular elasticity, which subsequently restricts the supply of oxygen and nutrients to cardiac muscles—including skeletal muscles—during exercise. These consequences lead to increased fatigue, decreased exercise performance, and—ultimately—delayed HRR. 15
In the present study, compared with increased arterial stiffness, autonomic nervous system dysfunction had a larger impact on HRR at 1 minute after exercise. The exact mechanism underlying these results remains unclear. However, it may be that autonomic nervous system dysfunction causes reduced activity of the parasympathetic nervous system, which plays an essential role in post-exercise HRR; this may then lead to secondary overactivity or abnormal responses in the sympathetic nervous system. In patients with SCI at or above T6, this phenomenon may contribute to delayed post-exercise HRR. By contrast, in patients with SCI below T6, who have normal autonomic nervous system function, it is presumed that reactivation of the parasympathetic nervous system is possible even with high arterial stiffness, thus leading to relatively faster HRR.
Based on these results and assumptions, an exploration of different strategies to improve HRR in Groups A and B is required. In Group A, delayed HRR was attributed to autonomic nervous system dysfunction. Numerous studies have reported that improved endurance is associated with enhanced HRR.29,30 This effect of endurance exercise has also been observed in patients with SCI. In a previous study, patients with SCI and autonomic nervous system dysfunction showed improvements in various indicators (including heart rate, SBP, DBP, stroke volume, and cardiac output) when subjected to endurance exercises.31,32
To improve delayed HRR in Group B, arterial stiffness needs to be addressed. Increased arterial stiffness is associated with delayed HRR and is a risk factor for various conditions, including CVD. Several studies have aimed to improve arterial stiffness through exercise, such as aerobic exercise or resistance training. However, the results of these studies have been inconsistent.33–35 Currently, lifestyle and dietary changes are the most effective methods for improving arterial stiffness. Previous studies have demonstrated that weight loss, smoking cessation, and omega-3 fatty acid consumption are effective for reducing arterial stiffness in patients with obesity. By contrast, few studies have reported a reduction in arterial stiffness in patients with SCI; however, when considering the improvement methods that are successful in the general population, we hypothesize that weight loss and smoking cessation may help to lower arterial stiffness and further improve HRR in Group B. 36
Studies of post-exercise HRR in patients with SCI are relatively limited. A previous study compared differences in post-exercise heart rate among ambulatory patients without SCI, patients with SCI and tetraplegia, and patients with SCI and paraplegia. Post-exercise HRR was most delayed in patients with tetraplegia, followed by those with paraplegia and then by ambulatory patients without SCI. 11 Similar results were observed in the present study. However, in the previous study, significant differences were noted among the three groups in variables such as the maximal heart rate, RER, V̇O2Peak, VE, MEs, and W values during the exercise tolerance test. In addition, unlike in the present study, there was a significant difference in the maximal heart rate after exercise between patients with paraplegia and those with tetraplegia. 11 These results are therefore limited because when differences in the maximal heart rate are observed, it is difficult to objectively evaluate variations in HRR. In the current study, to achieve enhanced objectivity compared with previous studies, we thus established a consistent starting point for the maximal heart rate, and measured the variability in HRR. In the previous study, patients with SCI were also not provided sufficient practice time for the exercise tolerance test. 11 By contrast, patients in the present study had an ample practice period (21 days) before the exercise tolerance test was conducted. We also set criteria for sufficient exercise capacity using an RER value ≥ 0.9 and compared the results among groups, thereby increasing the reliability of the test. In this way, we attempted to optimize the objectivity of the results by comparing only the results of patients with SCI who had sufficient exercise abilities.
The current study has some limitations. First, we did not analyze all risk factors for CVD. CVD can result not only from hypertension and diabetes, which were investigated in the present study, but also from factors such as dyslipidemia, family history, and smoking status. However, we did not conduct lipid tests, thus preventing a comparative analysis of patients with low- and high-density cholesterol levels. In addition, because the majority of patients with SCI experience trauma, our investigation into the family history of patients with CVD was insufficient. However, to overcome the aforementioned limitations, our study included patients in the acute or subacute phase, approximately 30 days after injury. Most previous studies have evaluated HRR in chronic patients, whereas the present study was performed after sufficient training for acute/subacute patients. In patients with SCI, conditions such as dyslipidemia, hypertension, and diabetes mostly result from chronic conditions arising from post-injury immobility and reduced physical activity. The timing of the assessments in the current study corresponded to the acute and subacute phase, suggesting that conditions such as hypertension and dyslipidemia had not yet manifested. To some extent, the present study therefore partially addressed the limitations associated with not considering the effects of these factors. Nevertheless, future studies that consider all risk factors for CVD occurrence—including smoking status, degree of obesity, and uncontrollable factors such as family and medical histories—are required. Second, in the current study, we measured blood pressure after 3 minutes of tilting. Given that we did not undertake continuous measurements, it is thus possible that we missed the orthostatic hypotension episode. In addition, the occurrence of non-symptomatic orthostatic hypotension or delayed orthostatic hypotension remains unknown because it was not investigated; future studies are therefore required. Third, because we performed a single center study, the number of samples was relatively small. Large-scale studies on autonomic dysreflexia, orthostatic hypotension, arterial stiffness, and HRR in SCI patients are therefore needed.
Conclusions
In the present study, HRR measured at 1 minute after exercise was slower in patients with SCI with an NLI at or above T6, and arterial stiffness was higher in those with SCI with an NLI below T6. Our findings suggest that approaches for enhancing HRR to prevent CVD in patients with SCI should tailored using the NLI as a guide. Specifically, for patients with SCI below T6, lifestyle improvements focusing on reducing arterial stiffness may be crucial. By contrast, for patients with SCI at or above T6, endurance exercises—including aerobic exercises performed at maximal or submaximal intensity—are likely vital for overcoming autonomic nervous system dysfunction.
Footnotes
Acknowledgements
This paper was supported by Wonkwang University in 2022.
Author contributions
SYC: study design, data collection, statistical analysis, data interpretation, writing manuscript. CYK: data interpretation, writing manuscript. MCJ: study design, supervision of findings, editing manuscript. We confirm that the manuscript has been read and approved by all authors. The corresponding author had full access to all data in the study and had final responsibility for the decision to submit for publication.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.

