Abstract
Objective
To investigate the neuroprotective effects of rosiglitazone in a rat traumatic spinal cord injury (SCI) model.
Methods
Adult Sprague–Dawley rats (n = 12/group) underwent laminectomy (sham), SCI, SCI and rosiglitazone treatment (2 mg/kg twice daily for 7 days), or SCI and saline injection (vehicle). SCI was induced via dural application of an aneurysm clip. Spinal cord apoptosis and levels of tumour necrosis factor-α (TNFα), interleukin (IL)-1β, myeloperoxidase (MPO) and the apoptosis-associated proteins B-cell leukaemia/lymphoma 2 (Bcl-2) and Bcl-2 associated X protein (Bax) were examined 24 h after SCI. Locomotor function was evaluated 3, 7, 10, 14 and 21 days after SCI.
Results
At 24 h after SCI, apoptosis and TNFα, IL-1β and MPO concentrations were significantly lower in the rosiglitazone group than in the vehicle and SCI groups. SCI resulted in an increase in Bax and a decrease in Bcl-2, which was reversed by rosiglitazone treatment. Rats in the rosiglitazone group had significantly better functional recovery than those in the vehicle and SCI groups.
Conclusion
Rosiglitazone significantly improved functional recovery, probably via attenuation of the local inflammatory reaction and reduced apoptosis.
Introduction
Spinal cord injury (SCI) usually results in catastrophic dysfunction and disability, with significant costs and burdens to families and society in general. 1 After the initial traumatic injury, many pathological events occur within the lesion that exacerbate neuronal death and neurological dysfunction, including inflammation, apoptosis, oxidative stress and excitotoxicity.2,3 Inflammation begins within minutes of injury and continues for several days,4–6 and is a major cause of secondary neuronal damage and motor dysfunction after experimental SCI. 7 Apoptosis within the injured spinal cord has been identified as another important component of secondary damage. 8 Apoptotic cell death along the longitudinal axis of the spinal cord may contribute to the delayed and prolonged demyelination process that leads to deterioration of spinal cord sensorimotor function. 9 A variety of strategies are being investigated to modulate the processes of inflammation and apoptosis after SCI.
The anti-inflammatory and antioxidant actions of methylprednisolone have led to its use in the treatment of human and experimental SCI.10,11 In spite of its widespread use, some investigators have expressed concern regarding the high incidence of complications and lack of evidence of neurological improvement following high-dose methylprednisolone treatment.12–14 Rosiglitazone is a synthetic agonist of peroxisome proliferator-activated receptor-γ (PPARγ) that is used in the treatment of type 2 diabetes, although its use is restricted due to side-effects including increased risk of stroke and heart failure.15,16 The anti-inflammatory and antioxidant properties of rosiglitazone have been studied in models of central nervous system injury and disease, including amyotrophic lateral sclerosis, 17 Parkinson’s disease 18 and cerebral ischaemia or haemorrhage. 19 Rosiglitazone has also been shown to prevent apoptosis in experimental traumatic brain injury. 20
The aim of the present study was to investigate the neuroprotective effects of rosiglitazone in a rat SCI model. Apoptosis and levels of proinflammatory cytokines (tumour necrosis factor-α [TNFα] and interleukin [IL]-1β), myeloperoxidase (MPO; a marker of neutrophil infiltration) and apoptotic signalling proteins (the antiapoptotic protein B-cell leukaemia/lymphoma 2 [Bcl-2] and the proapoptotic protein Bcl-2 associated X protein [Bax]) were examined in the injured spinal cord. In addition, post-traumatic function recovery was evaluated using the Basso, Beattie and Bresnahan (BBB) locomotor recovery scale 21 at 3, 7, 10, 14 and 21 days after injury.
Materials and methods
Study Design
Adult male Sprague–Dawley rats (n = 48, aged 12 weeks, weight range 200–250 g) were housed in a temperature-controlled room at 27°C, with free access to food and water. All surgical procedures were performed in an aseptic manner and were approved by the research animal resources and care committee of Zhejiang University, Hangzhou, China. Every effort was made to minimize the number of animals used and their suffering, in accordance with the Guide for the Care and Use of Laboratory Animals (available at http://grants.nih.gov/grants/olaw/Guide-for-the-care-and-use-of-Laboratory-animals.pdf). A preliminary study (unpublished) determined that 2 mg/kg rosiglitazone substantially attenuated the secondary damage following spinal cord injury, and this dose was therefore chosen for the present study. Rats were allocated to one of four groups (n = 12/group): (i) laminectomy only (sham group); (ii) laminectomy with SCI (SCI group); (iii) laminectomy, SCI and 2 mg/kg rosiglitazone (Avandia®; GlaxoSmithKline, Philadelphia, PA, USA) intraperitoneal (i.p.) injection (rosiglitazone group); (iv) laminectomy, SCI and 0.9% saline i.p. injection (vehicle group). The volume of each injection was 10 µl.
SCI Model
Rats were anaesthetized via 400 mg/kg chloral hydrate i.p. injection. A longitudinal incision was made on the midline of the back to expose the paravertebral muscles and the spinal cord was exposed via a four-level T5–T8 laminectomy. Compressive SCI was produced by transient (1-min) extradural application of an aneurysm clip (closing force 24
TNF-α and IL-1β ELISA and MPO Activity
Spinal cord segments (10 mm long, centred around the site of injury) were harvested from rats sacrificed 24 h after surgery (n = 2/group) and homogenized in 1 ml PBS (pH 7.4), containing protease inhibitors. Enzyme-linked immunosorbent assay (ELISA) for TNFα and IL-1β was performed in duplicate using the DuoSet® ELISA Development System (R&D Systems, Minneapolis, MN, USA), according to the manufacturer’s instructions. Optical density at 450 nm was determined using a microplate reader, and TNFα and IL-1β were quantified by reference to a standard curve.
Next, MPO activity was determined using an MPO activity assay kit (Nanjing Jiancheng Biological Institute, Nanjing, China) according to the manufacturer’s instructions. Optical density at 460 nm was determined using a spectrophotometer, and MPO activity was expressed as U/g wet tissue.
TUNEL Staining
Apoptotic cells were quantified by terminal deoxynucleotidyl transferase mediated dUTP nick end labelling (TUNEL) staining. In brief, a 60-mm segment of spinal cord with the lesion site at its centre was harvested 24 h after surgery (n = 2/group) and placed in 20% sucrose in PBS (pH 7.4) at 4°C until the blocks sank. Serial 20 -µm cross sections were mounted on slides and stained using a TUNEL detection kit (Roche, Basel, Switzerland), according to the manufacturer’s instructions. A total of six sections were examined for each animal (two sections from the centre of injury and two sections 2 mm away from the injury, on both sides). Slides were observed via light microscopy and neurons with brown-stained nuclei or containing apoptotic bodies were considered apoptotic. All TUNEL-positive cells were counted and examined for the typical pathological features of apoptosis. The mean number of TUNEL-positive cells in each group was calculated, and the apoptotic index was expressed as TUNEL positive cells/total cells × 100%. Independent scoring was performed by an investigator who was blinded to the study design.
Western Blotting
Spinal cord segments (10-mm long, centred around the site of injury) were harvested from rats sacrificed 24 h after surgery (n = 2/group), and Bcl-2 and Bax were quantified using Western blotting. Tissue was homogenized on ice in 10 mM TrisHCl (pH 7.4) containing 10 mM ethylenediaminetetra-acetic acid, 30% Triton X™-100, 10% sodium dodecyl sulphate (SDS) and 0.9% sodium chloride, then centrifuged at 12000
Functional Assessment
The BBB locomotor recovery scale 21 was used to assess functional recovery on days 3, 7, 10, 14 and 21 after surgery. This scale is based on the precise observation of hind-limb movements, stepping and co-ordination in an open field. Rats were observed for 4 min by an observer blinded to the treatment groups; behaviour was scored from 0 (no spontaneous locomotor activity) to 21 (normal movement co-ordinated gait with parallel paw placement). Scores were presented as the mean score of all animals in a group (n = 6) at each time point.
Statistical Analyses
Data were expressed as mean ± SEM. Multiple group comparisons of quantitative data were made using one-way analysis of variance followed by Dunnett's t test. Statistical analyses were performed using SPSS® software, version 13.0 (SPSS Inc., Chicago, IL, USA) for Windows®. A P-value < 0.05 was considered statistically significant.
Results
Tumour necrosis factor (TNF) α and interleukin (IL)-1β concentrations, myeloperoxidase (MPO) activity and apoptosis index in the spinal cords of rats 24 h after laminectomy (sham), laminectomy with spinal cord injury (SCI), SCI with rosiglitazone intraperitoneal (i.p.) injection (rosiglitazone) or SCI with saline i.p. injection (vehicle) (n = 2 animals/group per parameter).

Data presented as mean ± SEM of duplicate experiments.
P < 0.05 vs sham; bP < 0.05 vs SCI; cP < 0.05 vs vehicle: one-way analysis of variance followed by Dunnett’s t-test.
Apoptotic cells/total cells × 100%.
Locomotor function of rats, assessed using the Basso, Beattie and Bresnahan scale 21 0, 3, 7, 10, 14 and 21 days after laminectomy (sham), laminectomy with spinal cord injury (SCI), SCI with rosiglitazone intraperitoneal (i.p.) injection (rosiglitazone) or SCI with saline i.p. injection (vehicle) (n = 6 animals/group).
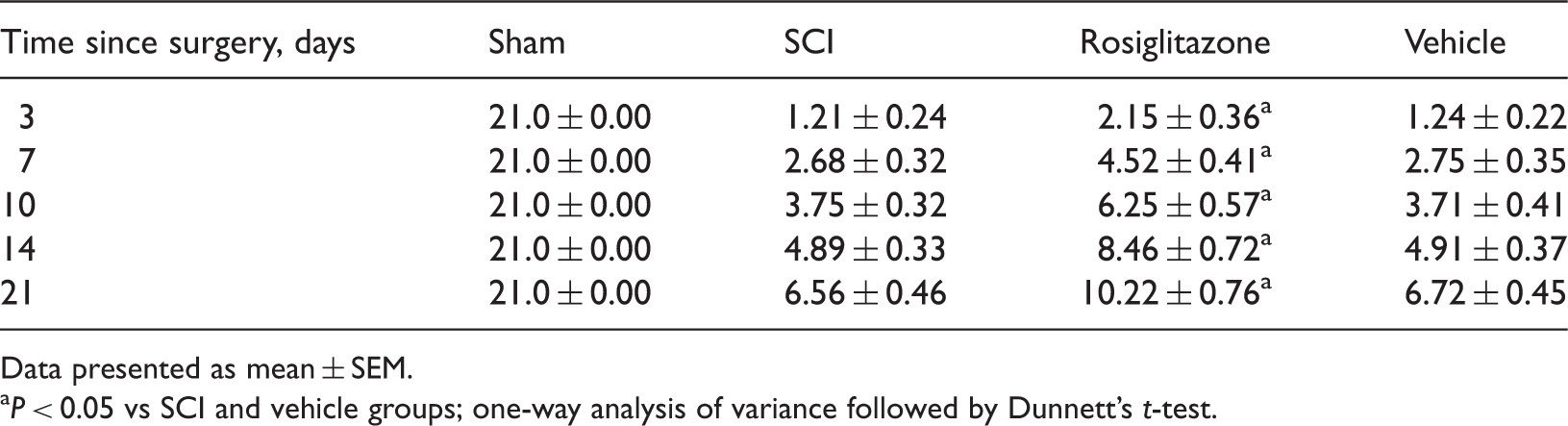
Data presented as mean ± SEM.
P < 0.05 vs SCI and vehicle groups; one-way analysis of variance followed by Dunnett’s t-test.
Levels of Bax were visibly increased in spinal cord tissue 24 h after SCI, and were significantly reduced by rosiglitazone treatment (P < 0.05; Figure 1). Bcl-2 levels were significantly lower in the SCI group than in sham-operated animals at 24 h post-injury (P < 0.05). This reduction in Bcl-2 levels was reversed by rosiglitazone-treatment (P < 0.05). There were no significant differences between SCI and vehicle groups in Bax or Bcl-2 levels.
(A) Representative Western blot for B-cell leukaemia/lymphoma 2 (Bcl-2) and Bcl-2 associated X protein (Bax) in spinal cord tissue samplesfrom rats 24 h after laminectomy (sham), spinal cord injury (SCI), SCI with vehicle or SCI with rosiglitazone (RSG). (B) Levels of Bax and Bcl-2 normalized to glyceraldehyde phosphate dehydrogenase (GAPDH) (n = 2 animals/group). aP < 0.05 vs sham, bP < 0.05 vs SCI and SCI + vehicle groups; one-way analysis of variance followed by Dunnett’s t-test.
Discussion
The attenuation of secondary degeneration following SCI is considered critical for improving patient outcome. 10 Secondary degeneration is affected by a variety of cellular and molecular mechanisms, including inflammation and cell death.4–7 PPARγ is a ligand-activated transcription factor of the nuclear hormone receptor superfamily, with important roles in glucose and lipid homeostasis. 22 The natural ligand of PPARγ is 15-deoxy-Δ12,14-prostaglandin J2 (15 d-PDJ2), 23 which has been shown to decrease neurological deficit after experimental intracerebral haemorrhage, 24 in addition to reducing inflammation and neuronal tissue damage associated with spinal cord trauma. 25 Thiazolidinediones (TZDs) are potent synthetic agonists of PPARγ, 22 two of which (rosiglitazone and pioglitazone) are currently approved by the US Food and Drug Administration for treatment of type-2 diabetes. TZDs have been reported to induce neuroprotection in animal models of chronic and acute neurological insult.17–20 Administration of rosiglitazone significantly improved levels of inflammatory and apoptotic markers, and resulted in better functional recovery than vehicle, after SCI in the present study.
An acute inflammatory response characterized by neutrophil infiltration and activation of microglia develops within hours of spinal cord injury. 26 These reactive cells produce several proinflammatory cytokines including IL-1, TNFα, IL-6, IL-3 and granulocyte-macrophage colony-stimulating factor, which mediate the inflammatory response and regulate cellular events after SCI.4–6 TNFα and IL-1β are significantly upregulated in the injured spinal cord as early as 1 h after SCI.4–6,27 MPO activity and levels of TNFα and IL-1β were significantly increased in the spinal cord tissue of injured rats compared with sham-operated animals 24 h after injury in the present study. In a finding similar to other studies,17–20,24 rosiglitazone treatment significantly reduced these increased levels of MPO, TNFα and IL-1β in the present study, thus protecting the spinal cord against secondary degeneration caused by inflammation. It is thought that TZDs confer their neuroprotective effects via the prevention of both microglial activation and inflammatory cytokine/chemokine expression, 28 in concurrence with the findings of the present study.
Cell death is an important component of secondary damage following SCI.29–33 Although necrosis is considered characteristic of secondary cell death, studies have revealed that much of the neuronal and glial cell loss after injury results from apoptosis.29–32 Apoptotic death of oligodendrocytes has been observed along the longitudinal axis of the spinal cord, and this may be involved in prolonged demyelination and deterioration of sensorimotor function. 33 Interventions that target apoptosis may therefore be beneficial for the prevention of secondary degeneration following traumatic SCI. Apoptosis initially occurs at the lesion centre 6 h after injury and lasts for several days. 32 A large number of apoptotic cells were detected in the SCI group at 24 h post injury in the current study, and significantly fewer were seen in animals treated with rosiglitazone.
Apoptosis can be triggered by both intracellular and extracellular signals, and involves a complex cascade of molecular and biochemical reactions in the cell. The proapoptotic protein, Bax, plays an important role in developmental cell death and central nervous system injury. 33 In addition, the administration of the antiapoptotic protein Bcl-2 into injured spinal cord tissue has been shown to increase neuronal survival, suggesting that SCI-induced decreases in Bcl-2 contribute considerably to neuronal death. 34 Compared with sham-operated animals, Bax levels were increased, and Bcl-2 was decreased, in spinal cord tissue 24 h after SCI in the present study. Treatment with rosiglitazone reversed these effects, reducing overall apoptotic cell death by preventing the loss of the antiapoptotic pathway and attenuating the activation of the proapoptotic pathway.
Locomotor function gradually recovered during the 21-day postinjury period in all injured animals in the present study, and function was significantly improved by treatment with rosiglitazone. Higher BBB scores indicate improvements in muscle co-ordination, sequencing and scaling of muscle activity, and interjoint and interlimb co-ordination. This improvement in locomotor recovery by rosiglitazone administration may be due to attenuation of the local inflammatory reaction and reduced apoptosis.
In conclusion, the present study demonstrated that rosiglitazone treatment following experimental SCI in rats significantly reduced levels of TNFα and IL-1β, MPO activity and apoptosis in the injured spinal cord, and improved functional recovery, compared with no treatment or vehicle. The PPARγ agonist rosiglitazone may be useful in the treatment of SCI, although the increased risk of stroke and heart disease associated with rosiglitazone should be noted in clinical practice.
Footnotes
Declaration of Conflicting Interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
