Abstract
Background
This study aimed to develop a mortality risk model for older adult patients with acute poisoning, addressing the lack of age-specific tools.
Methods
A retrospective study was conducted to develop and validate a nomogram for predicting in-hospital mortality. The training cohort included 452 patients (62.2% men; 51.3% ≥70 years; main poisoning types: pesticides, 39.9%; drugs, 28.2%), with 133 patients in an external validation cohort. Independent risk factors were identified through logistic regression, and the nomogram was developed in R. Model performance was assessed using area under the curve, sensitivity, specificity, calibration, and decision curve analysis; external validation was performed using data from another hospital.
Results
Multivariate analysis identified independent risk factors for in-hospital mortality: age ≥70 years (odds ratio = 2.66), heart rate <60 or ≥110 bpm (odds ratio = 2.00), lactate ≥3.5 mmol/L (odds ratio = 2.61), white blood count ≥15 × 109/L (odds ratio = 3.46), and myoglobin ≥1000 ng/mL (odds ratio = 8.62). Internal validation showed an area under the curve of 0.824, sensitivity of 72.4%, and specificity of 76.1%. External validation showed an area under the curve of 0.854, sensitivity of 85.3%, and specificity of 75.8%.
Conclusion
The nomogram effectively predicts in-hospital mortality in older adult patients with acute poisoning, outperforming conventional scores. It enables rapid risk stratification within 24 h of admission to guide early intervention.
Introduction
Older adults are more susceptible to acute poisoning, whether intentional or accidental, because of declines in cognitive abilities, self-care capacity, and organ function as well as the presence of multiple chronic comorbidities compared with younger individuals. 1 A comprehensive study 2 conducted in 2005 reported that the mortality rate from acute poisoning in older adults is ninefold higher than that in younger patients. Older adults also experience longer intensive care unit (ICU) stays and present with more severe conditions. The 2021 National Poison Data System (NPDS) report indicated that older adults (≥60 years) accounted for 9.54% of the 2.08 million toxic substance exposures in the United States. 3 Similarly, a Chinese study indicated that the mortality rate among older adults with acute poisoning could reach 16.9%, 4 with significantly higher intensive care and in-hospital mortality rates than those in younger groups. 5
With the global increase in life expectancy, the proportion of older adults is expected to rise, with the population aged 65 years projected to reach 12% by 2030, 16% by 2050, and 23% by 2100. 6 Consequently, the incidence and mortality associated with acute poisoning among older adults are also expected to increase, posing significant challenges to national healthcare systems and the economy. Therefore, promptly identifying and assessing older adults with acute poisoning at hospital admission is essential for reducing mortality.
With advances in artificial intelligence and information technology, computer algorithms and model construction have been increasingly applied in various medical fields, providing accurate clinical guideline and operational standards. 7 Predictive models have also demonstrated value in the management of emergency poisoning. 8
In cases of acute poisoning, critical illness assessment systems such as the Poisoning Severity Score (PSS), Sequential Organ Failure Assessment (SOFA), Simplified Acute Physiology Score (SAPS), and Acute Physiology and Chronic Health Evaluation (APACHE) are widely used.9,10 However, the prognostic performance of these scoring systems—including the PSS, SOFA, SAPS II, SAPS III, and APACHE II—in predicting mortality among patients with acute poisoning has not been specifically evaluated in the older adult population, and no clear recommendation exists regarding the optimal scoring tool.
Among these systems, the PSS requires assessment of 12 organ systems. Its complexity and reliance on subjective parameters limit its reliability and clinical practicality.11,12 The APACHE II is also cumbersome and time-consuming, while the SOFA requires continuous data updates. Consequently, these systems are not well suited for the rapid initial risk assessment of older adults with acute poisoning at admission.
Currently, only a limited number of new scoring methods have been developed to asses the fatality risk of acute poisoning. These systems often present inherent limitations, including small sample sizes, insufficient emphasis on the older adult population, inadequate external validation, and uncertain clinical practicality, all of which warrant further multicenter verification to establish their reliability and applicability. In 2021, the Korea Centers for Disease Control and Prevention introduced a new poisoning mortality scoring system (new-PMS) comprising 10 predicted indicators, which demonstrated good performance. 13 However, subsequent evaluations indicated that its predictive capability was inferior to that of the SOFA score, underscoring the need for additional validation of its clinical value. 14 Many existing prediction models identify age as a major risk factor for severe outcomes and death in patients with acute poisoning.12,13 Nevertheless, there remains a lack of models specifically designed to predict mortality due to acute poisoning in the older adult population.
Given this context, an externally validated mortality prediction model for acute poisoning specifically tailored to older adults would provide a reliable foundation for early clinical risk assessment and intervention. Such a model could enhance clinical diagnosis and treatment, thereby reducing mortality and easing the burden on healthcare systems and the broader economy.
Materials and methods
Study design
This retrospective study aimed to develop and validate a predictive model for in-hospital mortality in older adults with acute poisoning. The study cohort included patients aged ≥60 years who were admitted to the emergency ICU (EICU) and the emergency department of The First Affiliated Hospital of Wenzhou Medical University between 1 May 2012 and 1 May 2023, with a confirmed diagnosis of acute poisoning. Consecutive sampling was employed, and patient data were retrospectively obtained from the electronic medical record system. Diagnoses of acute poisoning were based on the history of toxic exposure, clinical manifestations, and laboratory and auxiliary examination results. 15 An external validation cohort comprised older adults with acute poisoning admitted to Lishui People’s Hospital during the same period.
Sample size
The sample size for the derivation cohort was calculated using G*Power version 3.1.9.4 (https://www.gpower.hhu.de/), with in-hospital mortality as the primary outcome. Based on Kaeley et al. 4 , the in-hospital mortality rate among older adults with acute poisoning was 16.9%, which was conservatively rounded to 17.0% for reference. The calculation assumed a two-tailed analysis, 80% power, a 5% margin of error, and a Z-score of 1.96 for a 95% confidence level, resulting in a required sample size of 217. To account for potential attrition due to incomplete medical records or exclusion criteria, an additional 10% was added, yielding a final sample size of 239 patients.
Inclusion criteria
Patients were included if they met the following criteria: aged ≥60 years and diagnosed with acute poisoning according to the 2016 Chinese Expert Consensus on Diagnosis and Treatment of Acute Poisoning.
16
Diagnostic criteria included the following:
Toxic exposure history. Information obtained from patients, family members, or on-site witnesses to allow rapid preliminary identification of the poisoning etiology; Clinical manifestations. Diagnosis based on patients’ symptoms and signs, identification of specific odors, and exclusion of other conditions with similar presentations; Laboratory examinations. Toxicological testing of samples such as on-site residual poisons, gastric contents, vomitus, blood, urine, and feces to assist in confirming the diagnosis.
All included patients had a clear discharge diagnosis of acute poisoning.
Exclusion criteria
Patients were excluded if they met any of the following criteria: (a) presentation more than 24 h after poisoning onset; (b) unclear type of poison; (c) incomplete clinical data; or (d) uncertain discharge outcomes (Figure 1).

Flowchart of patient inclusion in this study.
Ethical consideration
The study was conducted in accordance with the Declaration of Helsinki and approved by the Medical Ethics Committees of The First Affiliated Hospital of Wenzhou Medical University (Ethical Approval No.: KY2023 - R238) and Lishui People’s Hospital (Ethical Approval No.: 2023 - 169). The need for informed consent was waived due to the retrospective nature of the study.
Data collection and definitions
Two senior emergency physicians independently reviewed medical records according to the 2016 Chinese Expert Consensus on Diagnosis and Treatment of Acute Poisoning to determine eligibility based on the inclusion and exclusion criteria. Discrepancies were resolved by the quality control group. Demographic and clinical data were collected, including sex, age, medical history, complications, type of poison, route of exposure, time from poisoning to hospital presentation, initial vital signs, and clinical parameters obtained within 24 h of admission. Key parameters included the SOFA score, APACHE II score, arterial blood gases, lactate levels, complete blood count, liver and kidney function tests, and coagulation profile. In-hospital costs, length of stay, and discharge outcomes were also recorded. The primary outcome was in-hospital mortality, defined as death from any cause during hospitalization, including patients who died after admission or were automatically discharged following withdrawal of resuscitation efforts due to inevitable death. Patients who died during hospitalization were classified as the death group, while survivors were classified as the survival group.
Statistical analysis
Measurement data were assessed for normality and homogeneity of variance. Normally distributed data were expressed as mean ± standard deviation (mean ± SD) and compared between the survival and death groups using independent-samples t-tests. Non-normally distributed data were presented as median and interquartile range (M (Q1, Q3)) and compared using the Mann–Whitney U test. Categorical data were expressed as number (n, %) and compared using the chi-squared test or Fisher’s exact test. All statistical analyses were conducted using Statistical Package for the Social Sciences (SPSS) version 26.0 (IBM Corp, Armonk, NY, USA) and R version 4.2.2 (http://www.r-project.org/), with P-values <0.05 considered statistically significant.
Model development
Variables significantly associated with in-hospital mortality in univariate logistic regression analysis (P < 0.05) were included in a stepwise regression model. 17 Key variables were selected based on the Akaike information criterion (AIC) and Bayesian information criterion (BIC) to balance model fit and complexity. Variables with P < 0.05 or those that improved AIC/BIC were retained, while variables with P > 0.10 or that worsened AIC/BIC were removed, 18 resulting in a preliminary prediction model for in-hospital mortality.
Model performance was evaluated using the receiver operating characteristic (ROC) curve and the area under the curve (AUC). The ROC curve illustrated the trade-off between the false positive rate (FPR) and true positive rate (TPR), while the AUC quantified overall discriminative ability. To assess model robustness and adaptability to random variations, 1000 bootstrap resampling iterations were performed, and the mean AUC with its 95% confidence interval (CI) were calculated. 19 Calibration curves and the Hosmer–Lemeshow test were used to evaluate model accuracy. Decision curve analysis (DCA) was conducted to assess the clinical applicability and reliability of the model.
Model performance evaluation and validation
The clinical value of the newly developed prediction model was assessed by comparing its discriminative performance with two established vital sign-based early warning scores: the SOFA score and APACHE II score, calculated according to published criteria (https://www.mdcalc.com).
External validation was conducted using a cohort of 133 older adults with acute poisoning from Lishui People’s Hospital in Zhejiang Province. Model robustness and adaptability were assessed by calculating the mean AUC with a 95% CI. Performance was further evaluated using calibration curves, the Hosmer–Lemeshow test, and DCA to assess both reliability and clinical applicability.
Results
General information of older adults with acute poisoning
Clinical data from 688 older adults with acute poisoning were reviewed, with 452 patients ultimately included in the study (Table 1). The overall in-hospital mortality rate was 20.21%. Among the 376 survivors, 62.23% were men, while 56.58% of the 76 patients in the death group were men. The survival group had a lower proportion of patients aged ≥70 years than the death group (51.33% vs. 67.11%, P < 0.05). There were no significant differences in medical history between the groups (P > 0.05). The survival group had a higher incidence of pulmonary infection (38.56% vs. 22.37%, P < 0.05) but a lower incidence of multiple organ dysfunction syndrome (MODS) (2.66% vs. 57.89%, P < 0.05). No significant differences were observed in the incidence of gastrointestinal bleeding, shock, or acute kidney injury (P > 0.05). Patients in the death group required more invasive mechanical ventilation (56.58% vs. 30.05%, P < 0.05) and blood purification (92.11% vs. 56.91%, P < 0.05). The survival group had a longer median hospital stay (7 days vs. 1 day, P < 0.05). No significant differences were noted in time from poisoning to hospital presentation or hospitalization costs (P > 0.05).
Comparison of general information of older adults with acute poisoning.

a: Significance at P < 0.05, b: Significance at P < 0.01, c: Significance at P < 0.001.
Data are represented as median (IQR) or numbers (n; %).
MODS: multiple organ dysfunction syndrome; IQR: interquartile range; AKI: acute kidney injury.
Poisoning substances and causes in older adults with acute poisoning
The overall in-hospital mortality rate for common acute poisonings was 20.2%. In the survival group, the most frequently encountered substances were pesticides (39.9%), drugs (28.19%), natural organisms (12.5%), and mixed poisoning (6.19%). In the death group, pesticides were the leading cause (80.2%), followed by mixed poisoning (11.84%) and drug poisoning (6.59%). No significant mortality was observed for rodenticide, alcohol, or natural organism poisoning. Significant differences were observed between the two groups for pesticide, drug, and natural organism poisonings (P < 0.05). The primary exposure route in both groups was the digestive tract (88.03% vs. 88.16%), with no significant differences in mortality associated with airway or dermal exposure (P > 0.05). Intentional ingestion was the predominant cause of poisoning in both groups (78.46% vs. 89.47%), with no statistically significant differences in the cause of exposure (P > 0.05) (Table 2).
Comparison of poison types and causes of exposure in older adults with acute poisoning.
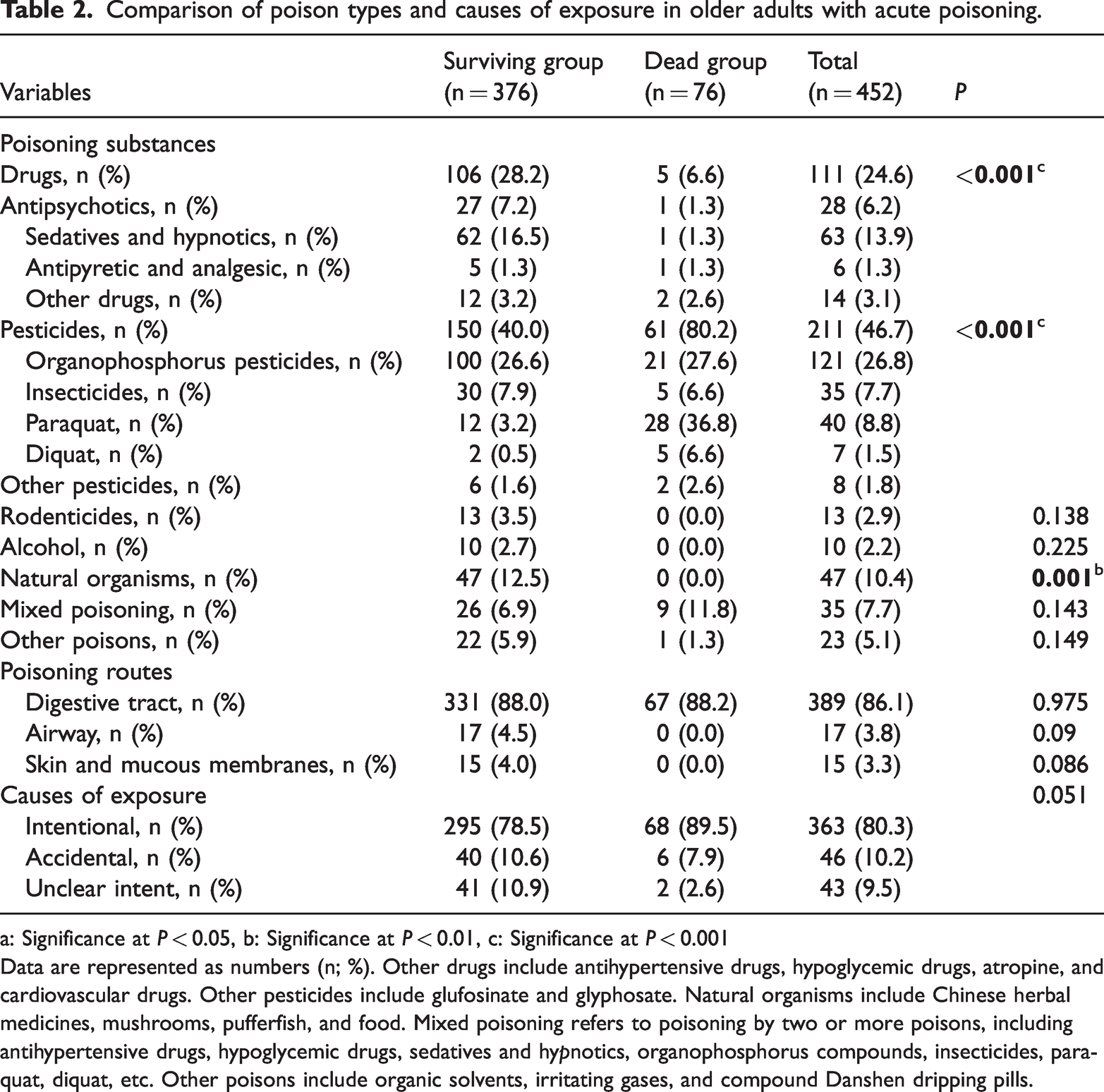
a: Significance at P < 0.05, b: Significance at P < 0.01, c: Significance at P < 0.001
Data are represented as numbers (n; %). Other drugs include antihypertensive drugs, hypoglycemic drugs, atropine, and cardiovascular drugs. Other pesticides include glufosinate and glyphosate. Natural organisms include Chinese herbal medicines, mushrooms, pufferfish, and food. Mixed poisoning refers to poisoning by two or more poisons, including antihypertensive drugs, hypoglycemic drugs, sedatives and hypnotics, organophosphorus compounds, insecticides, paraquat, diquat, etc. Other poisons include organic solvents, irritating gases, and compound Danshen dripping pills.
Comparison of vital signs and auxiliary examinations in older adults with acute poisoning
Significant differences in vital signs were observed between the groups. Patients in the death group were more likely to present with a body temperature <36.0°C or ≥39.0°C, heart rate (HR) <60 or ≥110 bpm, and respiratory rate <12 or ≥22 breaths/min (P < 0.05). No significant differences were noted in mean arterial pressure (P > 0.05) (Table 3).
Comparison of vital signs and laboratory examinations between surviving and death groups of older adults with acute poisoning.
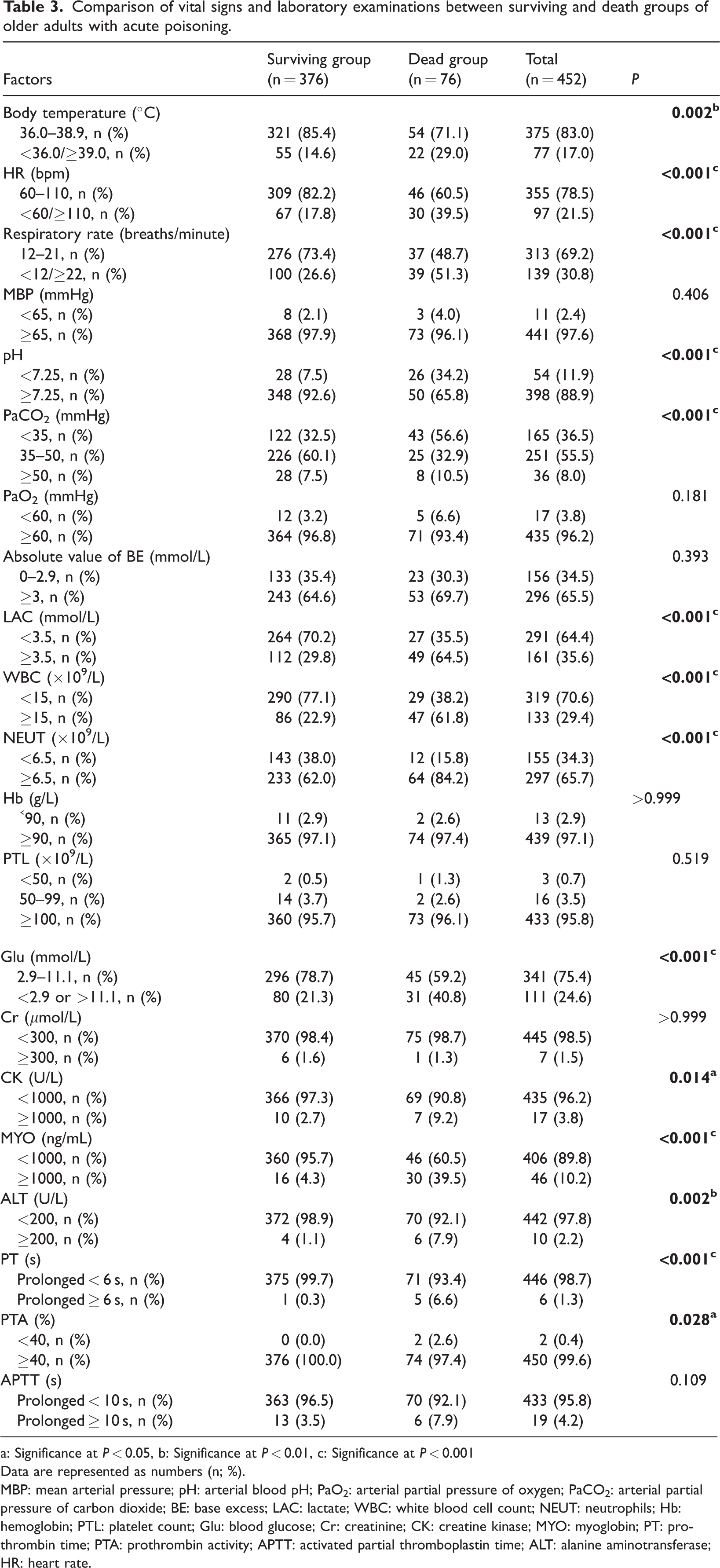
a: Significance at P < 0.05, b: Significance at P < 0.01, c: Significance at P < 0.001
Data are represented as numbers (n; %).
MBP: mean arterial pressure; pH: arterial blood pH; PaO2: arterial partial pressure of oxygen; PaCO2: arterial partial pressure of carbon dioxide; BE: base excess; LAC: lactate; WBC: white blood cell count; NEUT: neutrophils; Hb: hemoglobin; PTL: platelet count; Glu: blood glucose; Cr: creatinine; CK: creatine kinase; MYO: myoglobin; PT: prothrombin time; PTA: prothrombin activity; APTT: activated partial thromboplastin time; ALT: alanine aminotransferase; HR: heart rate.
Laboratory findings in the death group showed notable abnormalities, including pH < 7.25, lactate (LAC) ≥ 3.5 mmol/L, partial pressure of carbon dioxide in arterial blood (PaCO2) < 35 mmHg or ≥ 50 mmHg, white blood cell count (WBC) ≥ 15 × 109/L, neutrophil count (NEUT ) ≥ 6.5 × 109/L, abnormal glucose (Glu) levels, creatine kinase (CK) ≥ 1000 U/L, myoglobin (MYO) ≥ 1000 ng/mL, alanine aminotransferase (ALT) ≥ 200 U/L, and prothrombin time (PT) ≥ 6 s (P < 0.05). No significant differences were observed for partial pressure of oxygen in arterial blood (PaO2), base excess (BE), hemoglobin (Hb), platelets (PTL), creatinine (Cr), PT activity (PTA), or activated partial thromboplastin time (APTT) (P > 0.05) (Table 3).
Parameters of in-hospital mortality in older adults with acute poisoning
Univariate regression analysis identified 20 significant risk factors for in-hospital mortality, including age, sedatives and hypnotics, organophosphorus pesticides, paraquat, MODS, pulmonary infection, blood purification therapy, invasive mechanical ventilation, body temperature, respiratory rate, HR, LAC, WBC, NEUT, Glu, CK, MYO, ALT, and PT.
Multivariate logistic regression analysis showed that age ≥70 years (odds ratio (OR) = 2.66, P = 0.002), HR <60 or ≥110 bpm (OR = 2.00, P = 0.028), LAC ≥3.5 mmol/L (OR = 2.61, P = 0.002), WBC ≥15 × 109/L (OR = 3.46, P < 0.001), and MYO ≥1000 ng/mL (OR = 8.62, P < 0.001) were independent risk factors for in-hospital mortality (Table 4).
Multivariate logistic regression analysis of the prediction model for in-hospital mortality in older adults with acute poisoning.

a: Significance at P < 0.05, b: Significance at P < 0.01, c: Significance at P < 0.001.
HR: heart rate; LAC: lactate; WBC: white blood cell count; MYO: myoglobin; OR: odds ratio; CI: confidence interval.
Development and validation of the nomogram prediction model
Five independent risk indicators identified by multivariate logistic regression analysis were used to develop a nomogram for predicting in-hospital mortality in older adults with acute poisoning (Figure 2).

Nomogram prediction model for in-hospital mortality of older adults with acute poisoning.
The model demonstrated an AUC of 0.824 (95% CI: 0.774–0.837), with a sensitivity of 72.4% and a specificity of 76.1%. For comparison, the APACHE II score had an AUC of 0.738 (95% CI: 0.684–0.793), and the SOFA score had an AUC of 0.689 (95% CI: 0.636–0.741), indicating superior performance of the nomogram (Figure 3). Internal validation using a calibration curve yielded a Hosmer–Lemeshow test result of χ2 = 1.884, P = 0.984 (Figure 4( a)). DCA further indicated strong clinical applicability of the model (Figure 4(b)).

Comparison of ROC curves of different scoring systems for predicting in-hospital mortality of older adults with acute poisoning. ROC: receiver operating characteristic.
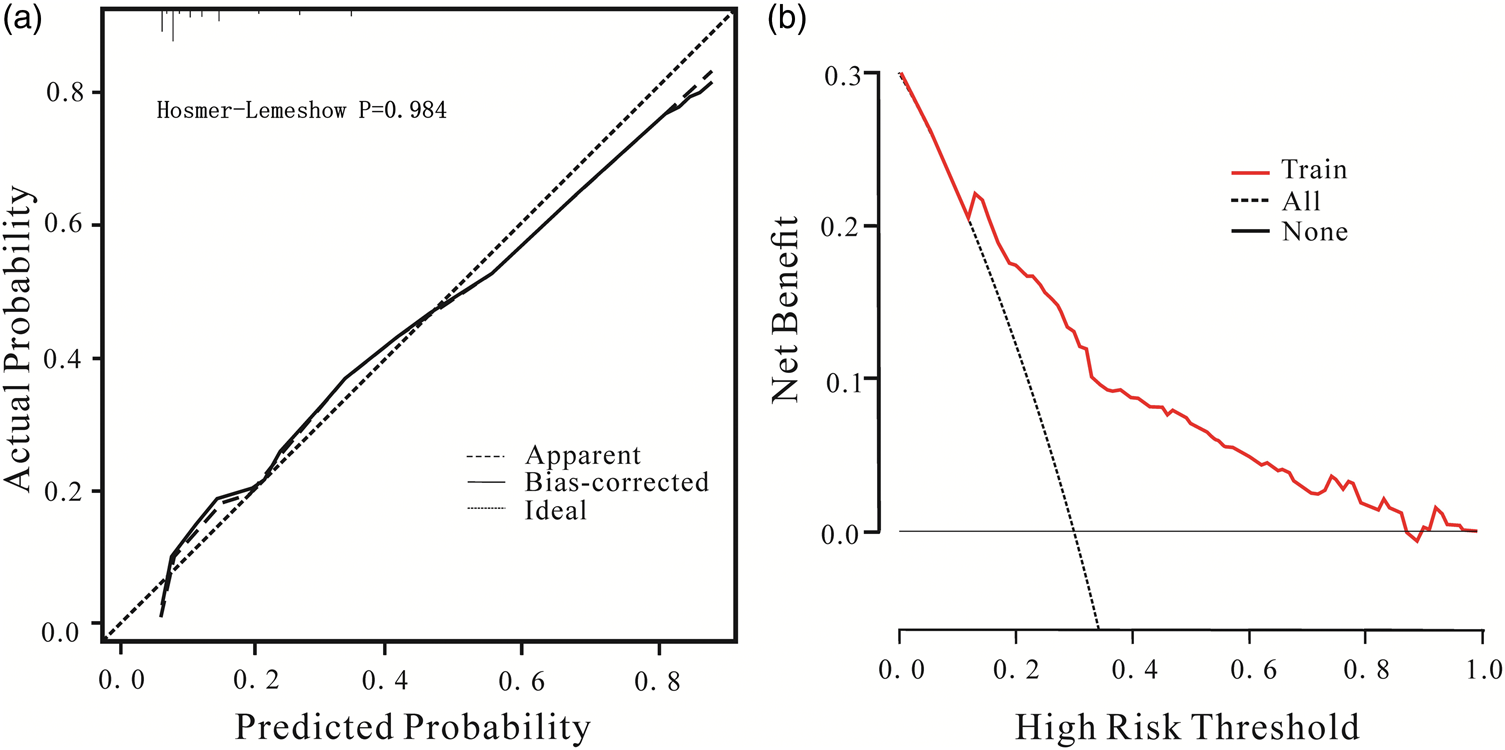
Calibration curve (a) and DCA curve (b) of the nomogram model for predicting in-hospital mortality risk of older adults with acute poisoning. DCA: decision curve analysis.
External validation using 133 older adults from Lishui People’s Hospital (May 2012–May 2023) demonstrated an AUC of 0.854 (95% CI: 0.779–0.929), with a sensitivity of 85.3% and specificity of 75.8%, confirming the clinical predictive value and generalizability of the nomogram (Figure 5). The calibration curve for external validation showed a Hosmer–Lemeshow test result of χ2 = 11.788, P = 0.108, further supporting the model’s reliability (Figure 6(a)). DCA comparing the development and validation cohorts indicated the model’s substantial clinical utility (Figure 6(b)).
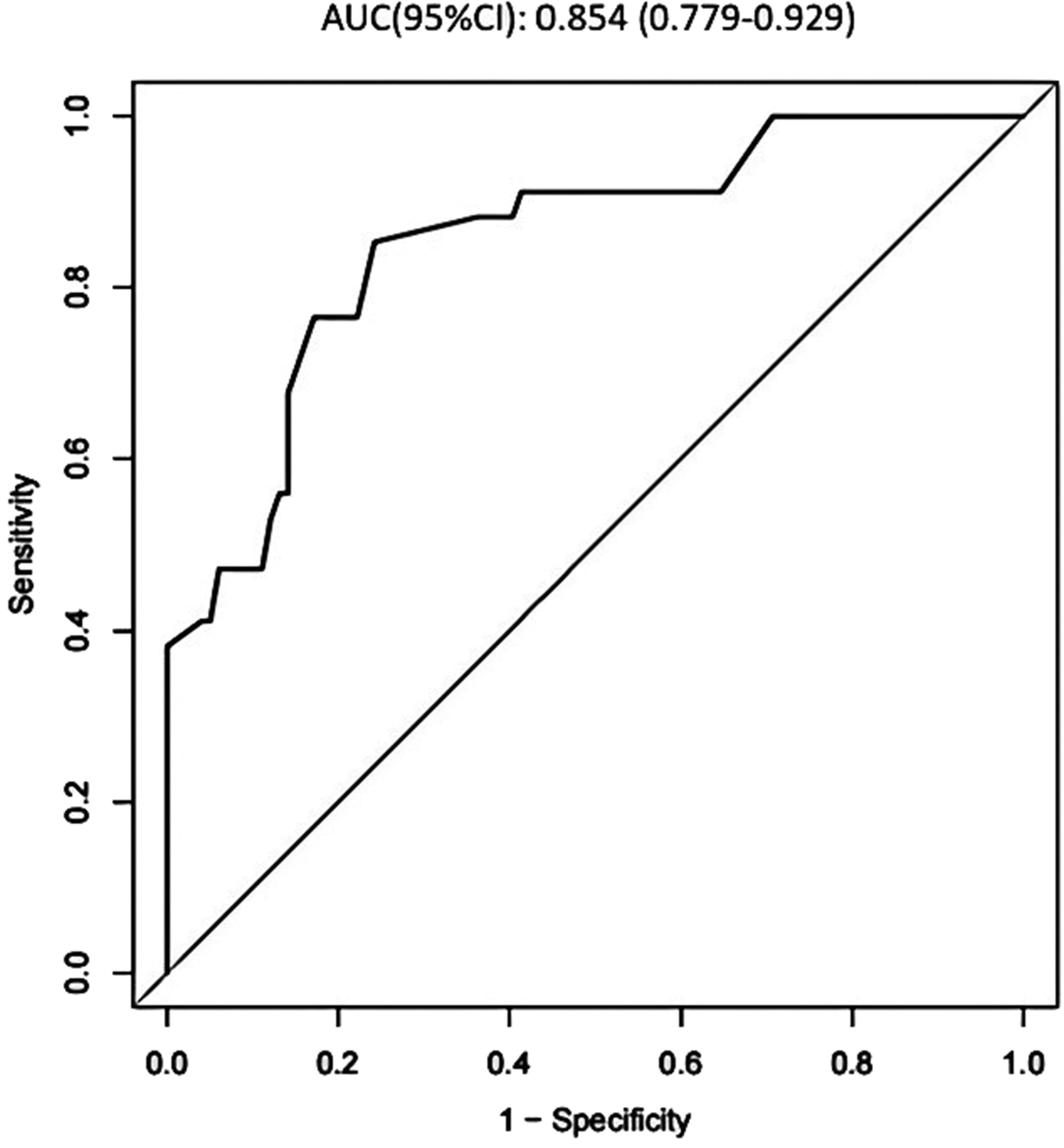
ROC curve of the validation group of the nomogram prediction model. ROC: receiver operating characteristic.
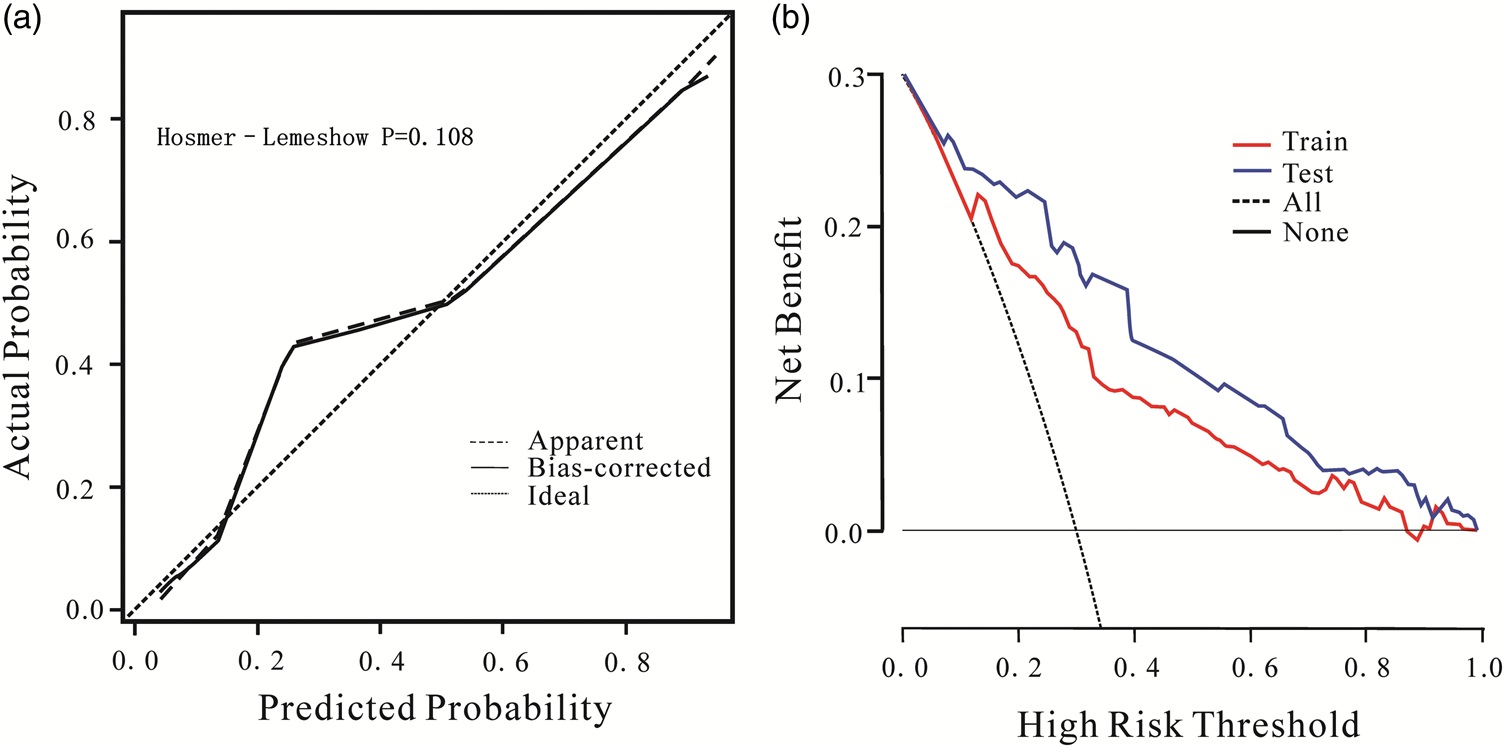
Calibration curve of the validation group of the nomogram prediction model (a) and comparison of DCA curves between the modeling group and the validation group (b). DCA: decision curve analysis.
Discussion
This two-center study focused on older adults with acute poisoning and developed an in-hospital mortality prediction model, which was externally validated. This model incorporates MYO as a novel predictor of mortality. Advances in artificial intelligence have significantly expanded the application of machine learning in emergency medicine, improving predictive models in clinical practice. 20 Nomograms have shown considerable utility in toxicology over the past 8 years.21–23 The nomogram developed in this study for predicting mortality in acute poisoning follows established research methodologies and addresses the notable gap of previous tools that were not specifically designed for older adults.
This mortality prediction model developed in this study includes five objective and quantifiable risk factors. This approach minimizes evaluation inaccuracies commonly associated with cognitive decline, communication difficulties, and other challenges prevalent among older adults. By incorporating readily available laboratory and clinical data, clinicians can perform early evaluations within 24 h of patient admission, making the model highly applicable in clinical practice.
Our study found an in-hospital mortality rate of 20.21% among older adults with acute poisoning. The primary poisoning agents were pesticides, mixed poisonings, and drugs, with organophosphorus pesticide poisoning being the most prevalent at 27.63%, followed by insecticide poisoning at 6.48%. Although scoring systems such as the PSS, APACHE II, SAPS II, and SOFA have been used to predict mortality in poisoning cases, there is no conclusive evidence supporting their superiority in specific poisoning scenarios. Therefore, a novel and more generalizable mortality prediction model is needed, particularly for the older adult population.
The study’s results showed that patients in the acute poisoning death group were older and had HRs that were either markedly elevated or reduced compared with those in the survival group. This finding is consistent with previous research on APACHE II and SOFA scores, where age and HR have been identified as independent predictors of mortality in specific poisoning scenarios, including pesticide exposure, 24 carbon monoxide poisoning, 25 and bee stings. 26 Our model further highlights the impact of abnormal HRs (<60 or ≥110 bpm) on the severity of acute poisoning, confirming the clinical relevance of this parameter.
WBC plays a critical role in inflammatory responses, toxin clearance, and immune defense in acute poisoning. Elevated WBC counts are closely associated with poisoning severity and poor prognosis, increasing the risk of mortality. 27 This relationship is particularly evident in paraquat poisoning, where oxidative stress induces the release of inflammatory mediators, resulting in organ dysfunction and higher mortality.28–30 Similar correlations have been observed in other types of poisoning, including diquat, 8 organophosphorus poisoning, 31 and envenomation. 32
LAC, a byproduct of anaerobic glucose metabolism, rises significantly in patients with acute poisoning, especially in the presence of severe complications such as shock, liver failure, severe hypoxemia, seizures, and tissue necrosis. Elevated LAC levels are closely linked to patient prognosis and mortality. Studies have demonstrated that LAC is a reliable early warning and predictive marker for short-term prognosis and in-hospital mortality in poisoned patients.33–35 Notably, in acetaminophen poisoning, a serum LAC level ≥3.5 mmol/L can predict mortality upon admission or shortly thereafter. 36
This study identifies elevated MYO as an independent risk factor for mortality in older adults with acute poisoning. MYO influences vascular tone and permeability, leading to endothelial dysfunction; in peripheral arterial disease, levels ≥100 ng/mL are associated with increased mortality. 37 MYO rises markedly in rhabdomyolysis, which can precipitate acute kidney injury (AKI) through tubular obstruction, lipid peroxidation, and reactive oxygen species-mediated cytotoxicity, potentially causing severe vasoconstriction. 38 Rhabdomyolysis-induced AKI is linked to higher mortality, 39 a relationship particularly relevant in severe poisoning cases—including paraquat, 40 diquat, 41 carbon monoxide, 42 poisonous mushrooms, 43 bee stings, 44 and snake bites 45 —where rhabdomyolysis, myocardial injury, and AKI frequently occur. In snake bites, rhabdomyolysis-related infection further elevates mortality risk, 46 supporting MYO’s direct and indirect association with poisoning severity and risk of death.
This study developed a mortality prediction model based on the clinical characteristics of older adults with acute poisoning admitted to the emergency department. By incorporating readily available laboratory and observational data, the model is highly practical for widespread implementation across hospitals. It achieved an AUC of 0.824, outperforming the APACHE II (0.738) and SOFA scores (0.689), which are commonly used to predict mortality in acute poisoning. The model demonstrated strong predictive value in external validation, achieving an AUC of 0.854.
Several limitations should be acknowledged. First, the study was based on single-center data with a relatively small sample size, which may introduce selection and retrospective biases. The limited sample also reduces the reliability of subgroup analyses, particularly for rare poisoning types comprising less than 5% of the cohort, and limits the generalizability of the findings. Multicenter prospective studies are therefore recommended. Second, although toxin dose is an established predictor of mortality in other models, uniform dose analysis was not feasible due to heterogeneous poisoning types and incomplete dose documentation. Further research with standardized dose data is needed to improve predictive accuracy. Third, the model does not fully account for variations in substance toxicity, particularly for high lethal agents such as paraquat. The general model’s performance for specific highly toxic substances remains unverified, highlighting the need for future studies to focus on specific poisons and develop tailored sub-models.
Conclusion
Older adults with acute poisoning exhibit high mortality rates, primarily due to pesticide, mixed, and drug poisonings, which often lead to rapid MOD and frequently require invasive interventions. This study developed a mortality prediction model incorporating age, HR, LAC, WBC count, and MYO. The model demonstrates strong clinical utility, enabling early identification of high-risk patients within 24 h of admission and allowing clinicians to implement timely, targeted interventions to improve outcomes and reduce mortality.
Footnotes
Acknowledgments
The authors thank E-transtar (Beijing) Information Technology Co., Ltd for assistance with language polishing.
Author contributions
Zhongqiu Lu contributed to conceptualization, funding acquisition, methodology, and project administration. Ke Li was responsible for conceptualization, data curation, formal analysis, investigation, software, visualization, and writing the original draft. Jianping Ye performed data curation, resources, and validation. Yahui Tang and Longwang Chen contributed to supervision and writing—review and editing.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interest
The authors declare no conflicts of interest.
Ethical statement
This study was conducted in accordance with the Declaration of Helsinki. Approval was granted in November 2023 by the Institutional Review Board (IRB) of the First Affiliated Hospital of Wenzhou Medical University, serving as the central IRB. The review outcomes were accepted by the IRBs of all participating institutions (KY2023-R238). Given the retrospective nature of the study, the central IRB approved a waiver of informed consent.
Funding
This study was supported by the Science and Technology Plan Project jointly established by the State Administration of Traditional Chinese Medicine and Zhejiang Administration of Traditional Chinese Medicine (NO. GZY-ZJ-KJ-23031), the Zhejiang Provincial Medical and Health Project (NO. 2024KY585), and the Self-funded Public Welfare Technology Application Research Project of Lishui Municipal Science and Technology Bureau (NO. 2024SJZC116).
