Abstract
Malignant hyperthermia during surgery represents a significant challenge for anesthesiologists due to its high mortality rate and the risk of long-term physiological damage. Despite preventive measures, malignant hyperthermia cases continue to be reported. The standard and effective response involves the prompt administration of dantrolene, a specific antidote. However, dantrolene availability can differ across countries and regions; not all healthcare facilities can guarantee its immediate availability for early intervention. This case report details the successful management of malignant hyperthermia in an adult during surgery without the use of dantrolene.
Introduction
Malignant hyperthermia (MH) is a rare yet life-threatening condition that can manifest during surgical anesthesia and is associated with a high mortality rate. 1 The genotype prevalence of MH is 1 in 500, while the phenotype prevalence is 1 in 10,000. The diagnosis is based on both genetic studies as well as skeletal muscle contracture testing, which is “presumptive” without these tests. This condition is often initiated by specific anesthetic agents, which provoke a drastic increase in skeletal muscle metabolism. The central mechanism involves the dysregulated handling of calcium ions within muscle cells. Anesthetic drugs, notably volatile inhalation anesthetics and succinylcholine, trigger a substantial release of calcium ions, causing prolonged muscle contractions, elevated heat production, and severe metabolic disturbances. 2
The primary management strategy for MH includes the immediate discontinuation of triggering agents and the intravenous delivery of dantrolene, the specific antidote for MH. This treatment is complemented by cooling, correcting metabolic imbalances, and supporting the affected organs. 3 Given the rarity of MH and the substantial cost associated with dantrolene use, not all healthcare facilities maintain an on-hand drug supply. In this case report, we describe the successful management of MH in a patient that was achieved via early detection and swift intervention despite the absence of dantrolene.
Case report
The treatment and publication of this case study required written consent from the patient. The patient details have been deidentified. The reporting of this study conforms to the Case Report (CARE) guidelines. 4 The patient was a male in his early 60 s who weighed approximately 60 kg and was admitted to the Meizhou Traditional Chinese Medicine Hospital in Guangdong Province 4 h after sustaining swelling, bleeding, deformity, and restricted movement in the right lower leg due to a car accident in June 2014. The patient has no history of hypertension, diabetes, surgeries, or allergies; the patient and his family denied a history of MH. The following vital signs were recorded upon admission: T 36.7°C, P 96 beats per minute (bpm), blood pressure (BP) 130/70 mmHg. The patient was conscious, with equal and reactive pupils (2.5 mm in diameter), a soft neck, a midline trachea, clear bilateral breath sounds, and no abnormal rales. No abnormal heartbeats were heard upon auscultation, and no pathological murmurs were detected. The abdomen was soft, with no tenderness or rebound pain. There was mild swelling and deformity of the left lower leg. A 1-cm long wound was observed on the medial lower calf, communicating with a tibial fracture. There was a 2-cm long soft tissue contusion on the lateral and posterior aspects, with exposed subcutaneous muscle, chronic hemorrhage, and good blood circulation in the right lower extremity, including a palpable dorsalis pedis pulse. The X-ray showed a comminuted fracture of the right tibia and fibula. Complete blood count: white blood cell 19 × 109/L, creatine kinase (CK) 150 U/L. The admission diagnosis was as follows: Open comminuted fracture of the right tibia and the fibula. Upon admission, preoperative preparations were made, and the patient was urgently sent to the operating room for “debridement, fracture reduction, and internal fixation surgery.”
Surgical details
The treatment process is summarized in Table 1.
Treatment process.
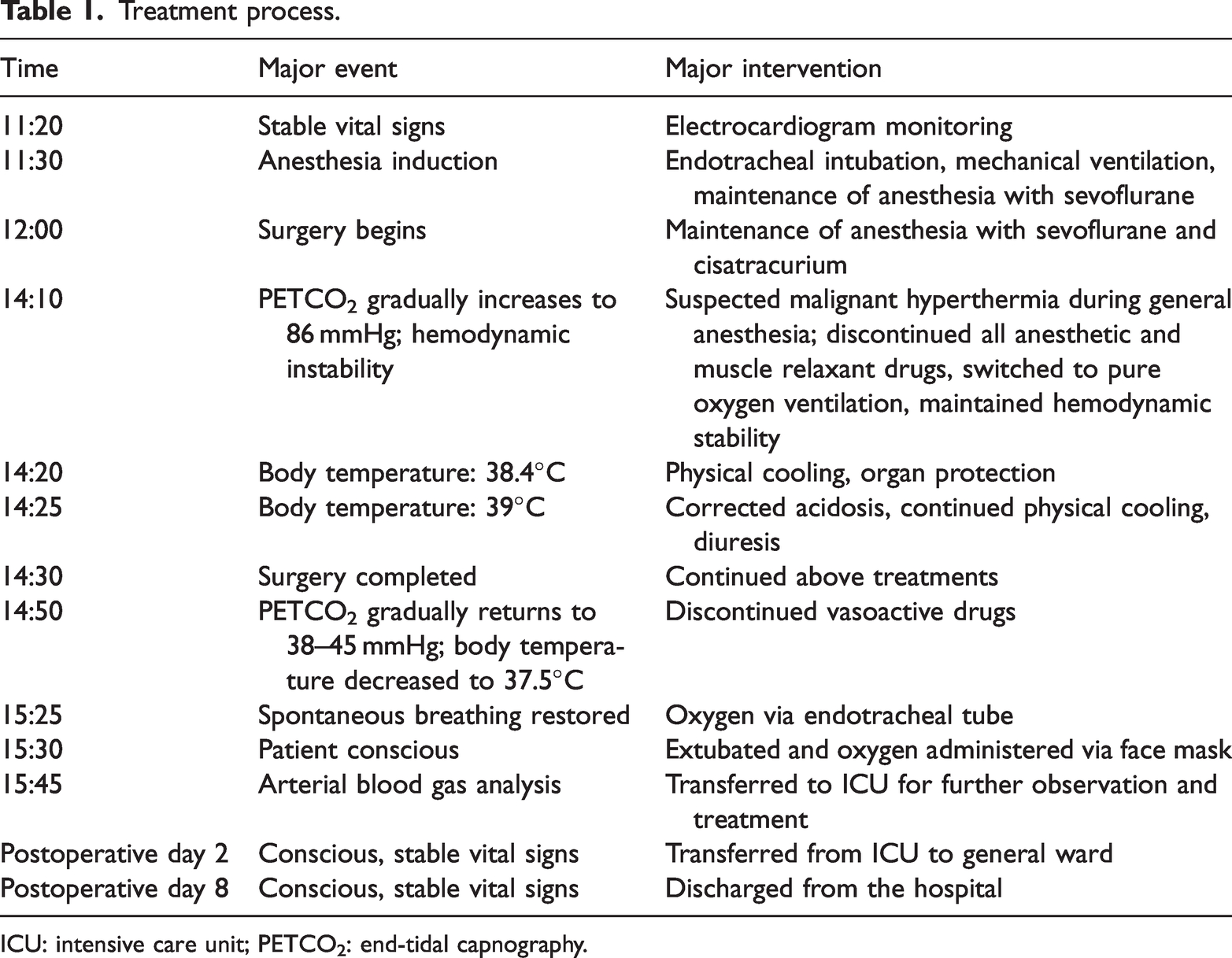
ICU: intensive care unit; PETCO2: end-tidal capnography.
At 11:20, the patient entered the operating room with stable vital signs as follows: consciousness intact, smooth respiration, BP 130/70 mmHg, heart rate 90 bpm, regular rhythm, oxygen saturation (SPO2) 99%. General anesthesia with endotracheal intubation was planned.
At 11:30, anesthesia induction began with the following agents: sufentanil 25 µg, etomidate 18 mg, and cisatracurium 10 mg. Endotracheal intubation was successfully performed. The ventilator parameters were set to a tidal volume of 400 mL, a frequency of 12 breaths/min, and an I:E ratio of 1:2. Anesthesia maintenance was achieved with sevoflurane 1%–3%, remifentanil 0.2 µg/kg/min, and cisatracurium 6 mg/h.
At 12:00, the surgery commenced. BP was maintained at 95–110/55–65 mmHg, heart rate at 75–95 bpm, end-tidal capnography (PETCO2) at 38–45 mmHg, SPO2 at 99%, and airway pressure at 18 mmHg.
At 14:10, the patient’s PETCO2 gradually increased. The tidal volume increased to 650 mL, and the respiratory rate was adjusted to 20 breaths/min. Despite replacing the soda lime, PETCO2 continued to escalate, peaking at 86 mmHg, while the airway pressure rose to 28 mmHg. Concurrently, the heart rate increased from 89 bpm to 145 bpm, and BP fell to 92/50 mmHg, subsequently reaching a minimum of 67/35 mmHg. Norepinephrine was administered to sustain BP at 80/50 mmHg, maintaining a heart rate of 140 bpm. These developments led to a suspected diagnosis of MH during general anesthesia administration. Consequently, all anesthetic and muscle relaxant drugs were ceased, and the anesthesia machine was switched to pure oxygen ventilation. The surgery and rescue were carried out simultaneously.
At 14:20, the patient exhibited symptoms, including facial flushing, mild sweating, and a body temperature of 38.4°C. Cooling measures were applied; alcohol wipes and ice packs were placed across the body. Additionally, dexamethasone 30 mg was injected, and 125 mL of mannitol was infused intravenously to protect brain function. Procedures for radial artery and right internal jugular vein catheterization were performed. Arterial blood gas analysis was conducted, and myocardial enzymes were measured (CK 1579 U/L, CK-MB 53.3 U/L).
By 14:25, the arterial blood gas analysis results were as follows: pH 7.159, partial pressure of carbon dioxide (PCO2) 62.6 mmHg, partial pressure of oxygen (PO2) 393.7 mmHg, potassium (K+) 5.75 mmol/L, BE −6 mmol/L, lactate (Lac) 5 mmol/L. The temperature had escalated to 39°C. Sodium bicarbonate was administered to correct the acidosis, and potassium levels were regulated with furosemide 20 mg intravenously to ensure a urine output greater than 1 mL/kg/h and to support kidney function. Physical cooling measures continued, leading to the stabilization of vital signs.
At 14:30, the surgery ended.
At 14:50, PETCO2 gradually returned to 38–45 mmHg after the treatment, and body temperature dropped to 37.5°C. The heart rate stabilized to 110–126 bpm. BP was maintained at 138–110/98–75 mmHg, and intravenous norepinephrine infusion was stopped.
At 15:25, the patient resumed spontaneous breathing with a tidal volume of 400 mL, a frequency of 15 breaths/min, and SPO2 of 98%. The patient was given nasal cannula oxygen and continued to be observed.
At 15:30, the patient was awake with stable vital signs. The endotracheal tube was removed, and oxygen was administered via mask.
Intraoperative fluid intake: 3000 mL of lactated Ringer’s, 250 mL of 5% sodium bicarbonate, 125 mL of mannitol, and 500 mL of hydroxyethyl starch.
Intraoperative measurements indicated a minimal blood loss of 20 mL and a substantial urine output of 2600 mL. At 15:45, arterial blood gas analysis revealed pH 7.347, PCO2 43 mmHg, PO2 107 mmHg, K+ 5.22 mmol/L, BE −1.3 mmol/L, and Lac 2.1 mmol/L. The heart rate stabilized to 100–120 bpm, and BP was maintained at 130–110/80–65 mmHg. The urine was clear, showing no signs of hematuria. To prevent hyperthermia recurrence, the patient was transferred to the intensive care unit (ICU) for continued observation and treatment.
ICU results were significant, with myoglobin levels exceeding 1000 ng/mL, BNP <100 pg/mL, and procalcitonin level of 0.29 ng/mL. Following treatment, the patient regained consciousness, breathed smoothly, and cooperated during the physical examination, exhibiting no signs of fever or chills.
His vital signs were as follows: T at 37°C, P at 89 bpm, BP at 140/70 mmHg, SPO2 at 98%. The patient was transferred out of the ICU on the second postoperative day and was discharged on the eighth postoperative day.
Discussion
MH is a rare, potentially fatal syndrome, with an incidence of approximately 1 in 100,000 cases. 5 This condition is commonly triggered during anesthesia administration due to potent inhalational anesthetics and depolarizing muscle relaxants. MH is characterized by a rapid increase in body temperature and severe metabolic disturbances, with mortality rates ranging from 79% to 91%. 6 The underlying mechanism is believed to involve abnormalities in the calcium release channels of the sarcoplasmic reticulum in the skeletal muscle. Clinical manifestations of MH vary and largely depend on factors, such as the anesthetic drugs used, patient age, and environmental conditions. The caffeine-halothane contracture test on skeletal muscle in vitro is recognized as the gold standard for screening and diagnosing MH. 7 However, this test is available only in a limited number of medical institutions in China, and it is challenging to perform during an emergency or for uncooperative patients. Consequently, diagnosis often relies on clinical symptoms and laboratory findings. After the patient’s recovery, we recommended that the patient and their family visit a specialized medical institution for a caffeine-halothane ex vivo skeletal muscle contraction test to confirm the diagnosis. However, they declined the recommendation. Therefore, based on clinical symptoms and laboratory results, we can only infer that the patient most likely experienced MH. In this particular case, general anesthesia was maintained with sevoflurane, a potent inhalational anesthetic, which could have been the primary trigger for the MH episode. 8 Non-depolarizing muscle relaxants, such as cisatracurium, may also have contributed to MH onset. 9 The clinical course and presentation in this case were typical, with laboratory findings supporting the diagnosis of MH.
The key diagnostic points were as follows: (a) a sharp increase in PETCO2 within 2 h of general anesthesia administration, unresponsive to routine management; (b) abnormal temperature elevation, with an increase of more than 0.5°C within 15 min; (c) tachycardia during the procedure; (d) metabolic and respiratory acidosis, hyperkalemia; (e) significant elevation of myocardial enzymes; and (f) rapid disease progression.
The application of dantrolene has been shown to reduce the mortality rate of MH from 80% to less than 5%, 10 highlighting the critical importance of this medication. However, despite the absence of dantrolene treatment in this case, the patient still achieved a positive outcome. The successful treatment was based on the following key factors: 1. Early detection of the rapid increase in PETCO2 during surgery with routine measures proving ineffective. After excluding other causes, the possibility of MH was immediately suspected, and all anesthetic and muscle relaxant drugs were discontinued. The sharp rise in PETCO2 is often the earliest and most easily observed clinical sign of MH, serving as an excellent early warning signal. 11 This phenomenon should be closely monitored during the process of anesthesia administration. Further confirmation of the diagnosis was obtained through continuous temperature monitoring. 12 2. The prompt initiation of cooling measures effectively controlled the body temperature, which helped mitigate complications that could have arisen from the effect of hyperthermia on major organs. 13 3. High-dose steroid therapy effectively reduced the inflammatory response, protecting the organs from further damage, particularly the brain, with mannitol being used for additional brain protection. 14 4. Early invasive arterial and venous monitoring allowed for tight monitoring of acid-base and electrolyte levels, facilitating proactive management of the patient’s homeostasis. 5. Early administration of furosemide and urine alkalinization were crucial for protecting kidney function. 6. MH can recur within 24 h; therefore, to prevent a relapse, the patient was transferred to the ICU for continued close observation and treatment.
We persistently and strongly recommend that the patient and their family undergo a caffeine halothane ex vivo skeletal muscle contraction test for diagnostic confirmation because we have sufficient reason to believe they may be at high risk of MH. If the patient or their family members require general anesthesia in the future, they must inform the anesthesiologist so that an appropriate anesthesia plan can be adopted to prevent MH.
Conclusion
In China, the availability of dantrolene is limited, making timely access of this medication for MH patients challenging. Nonetheless, the likelihood of successful treatment can be significantly enhanced through rigorous monitoring, vigilance maintenance, early diagnosis, and prompt symptomatic treatment, particularly effective cooling measures. This approach is crucial for reducing complications and improving the overall survival rate in MH patients.
Footnotes
Acknowledgements
We thank the patient for his support.
Consent for publication
The publication was approved by the patient, and informed consent was obtained and signed.
Data availability
The corresponding author can provide detailed data.
Declaration of conflicting interests
Cailing Zhang wrote the manuscript; Huanbin Wang provided relevant literature; Zhi Chen contributed the data; and Yuennong Zhang designed and revised the manuscript.
Ethical approval
This study was approved by the Ethics Committee of Meizhou Traditional Chinese Medicine Hospital, Guangdong Province (Ethics No. 2025-08, Time January 2025).
Funding
Not applicable.
