Abstract
Pyruvate dehydrogenase complex (PDHC) deficiency is a common genetic disorder leading to lactic acidosis, which can also result from several nongenetic conditions, such as septic shock. The present study reports a case of PDHC deficiency masked by septic shock-induced lactic acidosis. This case involved a 16-year-old adolescent with poor exercise tolerance compared with his peers, and no underlying diseases. The disease onset was characterized by cough, fever, and dyspnea, with hypotension and elevated lactate levels, which indicated septic shock. However, severe hypoglycemia and lactic acidosis persisted despite resolution of a pulmonary infection and correction of septic shock, requiring continuous intravenous infusion of 50% glucose. Although the patient did not experience acute kidney injury and had normal urine output, continuous renal replacement therapy was used to regulate the internal environment owing to the severity of the acidosis. The diagnosis of PDHC deficiency was considered on the basis of the persistent hypoglycemia and hyperlactatemia, before genetic mutation testing was completed. The clinical thinking process required a rich accumulation of pathophysiological knowledge. This article reports a case of PDHC deficiency masked by septic shock-induced lactic acidosis to raise awareness of the disease and avoid misdiagnosis and missed diagnosis.
Keywords
Introduction
Pyruvate dehydrogenase complex (PDHC) is an important mitochondrial energy metabolism enzyme. 1 PDHC deficiency is caused by mitochondrial PDHC gene mutations. 2 The activity of PDHC decreases with gene mutations, leading to the inability of pyruvate to enter mitochondria and obstruction of the tricarboxylic acid cycle. As a result, pyruvate is converted to lactate in the cytoplasm, inducing lactic acidosis. PDHC deficiency is characterized by decreased exercise tolerance, lactic acidosis, and chronic neurological disorders.2,3 Lactic acidosis is defined as a buildup of lactic acid in the blood and tissues, which can also result from several nongenetic conditions, such as septic shock. In the present study, we present a case of PDHC deficiency that was masked by septic shock-induced lactic acidosis. It was difficult to make a diagnosis on the basis of the available clinical data, and a wealth of pathophysiological knowledge was needed. The aim of this study was to raise awareness of the disease and avoid misdiagnosis and missed diagnosis.
Case report
This case report was approved for publication by the Research Ethics Committee of Guangdong Provincial People’s Hospital (approval no. GDREC2018424H(R1)). Written informed consent for treatment and publication was provided by the legal guardian (father) of the patient. The reporting of this study conforms to the CARE guidelines. 4
This case involved a 16-year-old adolescent who was admitted to the Emergency Department of Guangdong Provincial People’s Hospital on 23 December 2020. The patient had no underlying diseases but experienced decreased exercise tolerance compared with his peers. Two days before admission, he experienced paroxysmal cough with yellow purulent sputum and fever (up to 39°C). Half a day prior to admission, he developed shortness of breath, which was accompanied by dizziness, nausea, and overall weakness. He attended our clinic owing to persistent symptoms. Laboratory tests revealed the following: white blood cell count (WBC): 22.12 × 109/L (normal range: 4–10 × 109/L), procalcitonin (PCT): 233 pmol/L (normal range: 0–38 pmol/L), C-reactive protein (CRP): 565 nmol/L (normal range: 0–435 nmol/L), and serum lactate: 8.2 mmol/L (normal range: 0.5–1.7 mmol/L). Owing to low blood pressure (systolic pressure: 80 mmHg), he was admitted with a diagnosis of pulmonary infection and septic shock.
The physical examination and auxiliary inspection on admission supported the diagnoses indicated by the laboratory findings. The patient was conscious with acute distress and a temperature of 38.9°C, heart rate of 125 beats per minute, respiratory rate of 28 breaths per minute, and blood pressure of 86/52 mmHg on admission. Coarse breath sounds and wet rales were ausculted in the lower left lung. The results of auxiliary examinations 2 days after the first laboratory tests were as follows: WBC: 15.36 × 109/L, neutrophil ratio (NEUT): 0.975, PCT: 310 pmol/L, CRP: 452 nmol/L; chest computed tomography (CT): left lower lung infection focus; blood gas analysis: pH: 7.32 (normal range: 7.35–7.45), oxygen partial pressure (PaO2): 135 mmHg (normal range: 80–100 mmHg while inhaling low-flow oxygen), carbon dioxide partial pressure (PaCO2): 13 mmHg (normal range: 35–45 mmHg), oxygen saturation (SaO2): 100% (normal range: 95%–98%), bicarbonate (

Norepinephrine doses over time.
The pulmonary infection improved markedly after anti-infection treatment (Figure 2), but severe hypoglycemia (3.8–6.4 mmol/L, despite continuous intravenous infusion of 50% glucose for 24 hours) and lactic acidosis (lactate: 5.8–11.3 mmol/L) persisted (Figure 3). Continuous intravenous infusion of 50% glucose and blood purification were used to maintain internal environment stability. Blood gas analysis provided the trends for pH and PaCO2 over time (Figure 4). The following laboratory results were obtained 10 days after admission: WBC: 9.43 × 109/L, NEUT: 67.5%, PCT: 20 pmol/L, and CRP: 104 nmol/L. Chest CT indicated marked resolution of the pulmonary infection focus. After analyzing the findings in this case, the possibility of PDHC deficiency was considered, and genetic mutation analysis revealed an p.Arg302His mutation in the PDHA1 gene. The lactic acidosis resolved with therapy, and the patient’s condition improved. He was discharged after discontinuation of intravenous glucose and changing to a ketogenic diet.
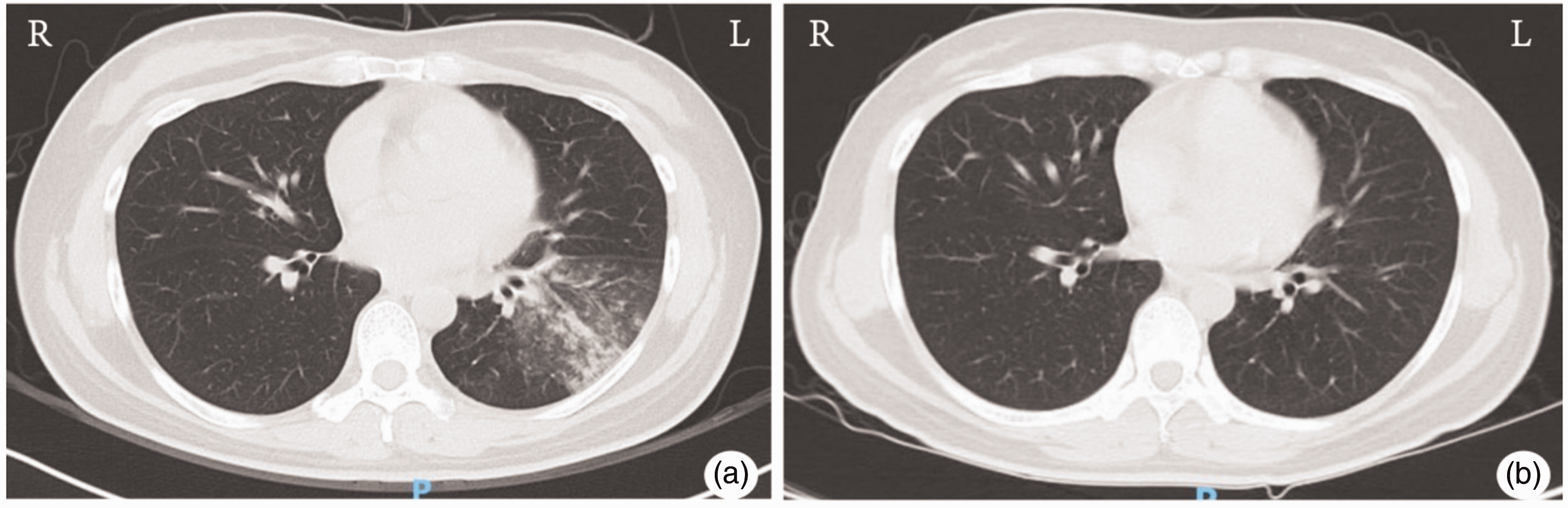
Chest CT images before (a) and after treatment (b). CT, computed tomography.

Blood glucose and lactate levels during hospitalization. The patient received continuous intravenous infusion of 50% glucose from day 2 to day 12.

pH and PaCO2 values over time during hospitalization. Continuous renal replacement therapy was used to regulate the internal environment from day 1 to day 12. PaCO2, carbon dioxide partial pressure.
Discussion
This case involved a 16-year-old adolescent with poor exercise tolerance compared with his peers and no underlying diseases. The onset of the disease was characterized by cough, fever, dyspnea, hypotension, and elevated lactate levels, which indicated septic shock. A low dose of norepinephrine was required to maintain systolic blood pressure at 100 mmHg to 125 mmHg. The septic shock was corrected quickly, and norepinephrine was no longer required. However, despite correction of a pulmonary infection and shock, severe hypoglycemia and lactic acidosis persisted, requiring continuous intravenous infusion of 50% glucose. Although hemodynamic stabilization was achieved quickly, lactate levels were consistently high, and pH was low, from the time of admission, which were markedly inappropriate compared with the hemodynamic stability. Because a definitive diagnosis could not be made at that time, septic shock remained the clinician’s first choice of diagnosis. Although the patient did not develop acute kidney injury and had normal urine output, continuous renal replacement therapy was used to regulate the internal environment owing to the severity of the acidosis. The diagnosis of PDHC deficiency was considered, with the persistent hypoglycemia and hyperlactatemia, before genetic mutation testing was completed. This clinical thinking process required a rich accumulation of pathophysiological knowledge, in this case. Genetic mutation analysis revealed a p.Arg302His mutation in the PDHA1 gene. Following discontinuation of intravenous glucose and initiation of a ketogenic diet, the lactic acidosis resolved, and the patient was finally discharged from the hospital.
Glucose is an important energy source that maintains physiological functions in the body. As glucose enters the body, it is transported to the cytoplasm and converted into pyruvate, nicotinamide adenine dinucleotide (NADH), hydrogen (H+), and adenosine triphosphate (ATP) through glycolysis, a non-oxygen-dependent metabolic pathway common to both anaerobic and aerobic metabolism. With sufficient oxygen, pyruvate and H+ are transferred into the mitochondria for the tricarboxylic acid cycle, which produces 36 molecules of ATP through oxidative phosphorylation. When the body is deprived of oxygen, pyruvate and H+ are obstructed from entering the mitochondria and are converted into lactate.5,6 This is why an elevated serum lactate level is considered a sign of tissue hypoxia. 7 However, in our patient, despite sufficient oxygen, pyruvate was still unable to enter the mitochondria for oxidative phosphorylation. Instead pyruvate was converted to lactate in the cytoplasm, which manifested clinically as resistant hypoglycemia and lactic acidosis. This clinical manifestation was initially difficult to understand. Are there other conditions necessary for pyruvate to enter the mitochondria other than oxygen? Thus, PDHC was considered. Pyruvate enters the mitochondria for oxidative phosphorylation under aerobic conditions; however, pyruvate is converted to lactate under anaerobic conditions. Through the analysis of this case, we are reminded that sufficient oxygen is not the only factor facilitating entry of pyruvate into the mitochondria. PDHC is also a necessary link in this process. The subsequent genetic mutation analysis confirmed a p.Arg302His mutation in the PDHA1 gene in our patient. The decreased activity of PDHC with this genetic mutation results in obstruction of pyruvate entering the mitochondria and the tricarboxylic acid cycle, causing pyruvate to be converted to lactate in the cytoplasm. This induces rapid consumption of glucose and severe lactic acidosis. However, we did not measure pyruvate levels in our patient, which is a limitation in this study.
PDHC deficiency is a rare inherited mitochondrial energy metabolism disorder. The clinical symptoms comprise paroxysmal movement disorders, which usually occur after exercise, and more than 50% of patients present with dystonia in the lower extremities, varying from 5 to 30 minutes in duration.8–10 PDHC deficiency may be accompanied by other neurological symptoms, such as ataxia or epilepsy, and magnetic resonance imaging often shows abnormal signals in the bilateral pallidum, which helps with the diagnosis.8–10 The symptoms and signs of PDHC deficiency vary dramatically depending on the age of onset. PDHC deficiency in neonates is characterized primarily by severe fatal hyperlactatemia. 11 The severity and the survival time in infants and young children are closely related to the lactate levels. 12 Symptoms in a small number of patients are relatively mild, observed mainly in children or adolescents, and present as ataxia, chorea-like movements, and dysarthria after high carbohydrate intake or exercise. 13 Similarly, our patient was 16 years old at the time of consultation, which is consistent with previous reports. 13 Our patient experienced mild symptoms, such as decreased exercise endurance. His condition improved rapidly after discontinuation of the glucose infusion and treatment with a ketogenic diet, vitamin B1, coenzyme Q10, and other supplements.
In summary, PDHC deficiency is a rare genetic disease, and the clinical symptoms vary with the age of onset, leading to misdiagnosis or missed diagnosis. PDHC deficiency should be considered when a person presents with hypoglycemia, hyperlactatemia, and muscle tone disorders after carbohydrate intake or exercise. Additionally, genetic mutation testing should be completed promptly. This article reports a case of PDHC deficiency in which lactic acidosis was masked by septic shock in the early stage. Despite the improvement of a pulmonary infection and correction of septic shock, severe hypoglycemia and lactic acidosis persisted, eventually leading to the diagnosis of PDHC deficiency. This case increases our understanding and awareness of rare diseases, contributing to timely diagnosis and proper treatment of PDHC deficiency.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241252112 - Supplemental material for Pyruvate dehydrogenase complex deficiency masked by septic shock-induced lactic acidosis: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605241252112 for Pyruvate dehydrogenase complex deficiency masked by septic shock-induced lactic acidosis: a case report by Hao Zhou, Yin Wen and Hongguang Ding in Journal of International Medical Research
Footnotes
Author contributions
Hao Zhou and Yin Wen designed the study; Hongguang Ding wrote the manuscript; Hao Zhou and Hongguang Ding collected the clinical information; Yin Wen revised the manuscript; all authors have read and approved the final manuscript.
Data availability statement
The data analyzed in this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation for Young Scientists of China [grant number 82002074], the China International Medical Foundation Cerebrovascular Disease Youth Innovation Fund [grant number Z-2016-20-2201], and the Natural Science Foundation of Guangdong Province [grant number 2023A1515010267].
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
