Abstract
This case report evaluated the efficacy and safety of dupilumab, a monoclonal antibody targeting interleukin-4 and interleukin-13, in a patient with atopic dermatitis (AD) and a history of metastatic renal cancer. Over 20 months of follow-up, the patient experienced significant improvement in AD symptoms without any recurrence of cancer. In addition to not stimulating carcinogenesis, dupilumab might synergistically inhibit tumor-induced immunosuppression, thereby potentially preventing tumor progression. This case suggests that dupilumab could be a viable treatment option for patients with AD and a history of solid neoplasms, filling a critical gap in current therapeutic approaches. Current evidence indicates that dupilumab does not promote cancer progression, but further research is necessary to confirm its long-term safety in this population. To establish robust scientific evidence and develop precise clinical guidelines, future studies should be randomized, controlled, and large-scale. This will help provide a definitive understanding of the safety and efficacy of dupilumab in patients with a history of cancer and inform clinical practice. In summary, although the initial findings are promising, comprehensive research is essential to ensure the optimal management of AD in patients with a history of malignancies.
Keywords
Introduction
AD is a chronic inflammatory skin condition that significantly affects patients’ quality of life. The pathophysiology of AD involves the complex interplay of genetic, immunological, and environmental factors, with dysregulation of the Th2 immune response playing a central role. This dysregulation leads to excessive production of cytokines such as interleukin (IL)-4 and IL-13, which contribute to inflammation and skin barrier dysfunction.1–3 As a chronic disease, AD is associated with various comorbidities over a patient’s life, complicating management. These comorbidities include solid malignancies, the incidence of which has been increasing in developed countries. Managing these patients is challenging because of the lack of evidence supporting the use of current systemic treatments.4,5
Dupilumab, approved in 2017, is a fully human monoclonal antibody that binds to the alpha subunit of the IL-4 receptor, blocking IL-4 and IL-13 signaling and thereby modulating the Th2 inflammatory response.3,4,6,7 The efficacy of dupilumab is well documented, making it a valuable therapeutic option for patients with severe AD. 8 Despite its efficacy, there is a paucity of literature on the use of dupilumab in patients with a history of malignancies or those with currently active malignancies.4,9
Case report
This case involved a 61-year-old man with a history of mild AD that was controlled with emollients and topical corticosteroids. In 2015, he was diagnosed with clear cell renal cell carcinoma of the right kidney, classified as T3a N0 M0 according to the pathological TNM staging system. He underwent radical nephrectomy to manage the cancer. During surveillance in 2018, he developed extensive inguinal lymph node metastasis, leading to the initiation of treatment with pazopanib, a tyrosine kinase inhibitor targeting VEGFR1–3. By 2020, the disease had spread to the mediastinal lymph nodes, prompting treatment with the PD-1 inhibitor nivolumab. Although nivolumab effectively controlled the cancer, it markedly worsened the patient's AD.
AD was managed with oral prednisolone (15–30 mg daily) for 2 years until he was referred to our center. At the time of referral, the patient had an Eczema Area and Severity Index (EASI) score of 27 and a Numeric Rating Scale (NRS) for itch of 8/10. Dupilumab treatment was initiated at an initial dose of 600 mg followed by 300 mg every 2 weeks.
After 4 months of dupilumab therapy, significant improvements were observed in the patient’s AD, with the EASI score decreasing to lower than 7 and the NRS for itch decreasing to 2/10. The patient was able to discontinue oral corticosteroids, and dupilumab treatment was continued for 20 months while maintaining symptom improvement (EASI < 3 and NRS for itch of 2/10). Nivolumab was discontinued because the patient’s cancer was adequately controlled by dupilumab. Importantly, the patient’s cancer did not progress during this period.
The patient provided written informed consent for both his treatment and the publication of this case report. It should be noted that treatment decisions were made in cooperation with an oncology specialist. This case report follows the CARE (CAse REport) guidelines to ensure comprehensive and transparent documentation of the clinical case. 10
Discussion
AD requires continuous and effective management to improve patients’ quality of life. Non-biologic systemic treatments, such as cyclosporine and methotrexate, are often associated with toxicity and long-term complications, including significant immunosuppression, which can potentially increase the risk of cancer development or recurrence. JAK inhibitors should be avoided in this population because of the potential risk of exacerbating cancer progression or recurrence.5,7 By contrast, the mechanism of action of dupilumab, which blocks IL-4 and IL-13 signaling without broadly suppressing the immune system, offers a more targeted and potentially safer therapeutic approach for this patient population. The most common side effects of dupilumab reported in clinical trials are conjunctivitis and local injection site reactions, which usually are easily managed. 1
The clinical experience with dupilumab in patients with AD and a history of cancer has been encouraging, but it remains limited. Case studies and series have demonstrated that dupilumab can safely and effectively control AD without causing cancer recurrence or progression.2–4,8,9,11,12 A case series published in 2021 found that patients with various types of cancer who received dupilumab did not experience neoplastic progression during treatment over an average of 54 weeks of follow-up, suggesting that modulating IL-4 and IL-13 pathways does not promote carcinogenesis. 8 Interestingly, a study highlighted the potential synergistic effect of dupilumab when used in combination with PD-1/PD-L1 inhibitors, particularly in non-small cell lung cancer. IL-4 blockade by dupilumab reduces immunosuppression mediated by myeloid-derived suppressor cells, thereby enhancing the anti-tumor response. In laboratory models, dupilumab modulated the immune system, thereby reducing the immunosuppressive environment created by tumors and promoting the infiltration of CD8+ T cells into the tumor. This suggests that dupilumab can both prevent cancer progression and potentiate the effects of immunotherapies. 12 IL-13 appears to be associated with the pathogenesis and progression of various cancers. 6 For this reason, it is expected that dupilumab, by blocking signaling by this cytokine, can prevent the progression of malignancies. Recent studies suggested a link between dupilumab and the onset or progression of cutaneous T-cell lymphoma (CTCL).6,13 However, subsequent findings revealed that some patients initially diagnosed with AD were later correctly diagnosed with CTCL.5,7
Recently, a cohort study conducted at the University Hospital of Turin analyzed 24 patients with moderate-to-severe AD and a history of cancer who received dupilumab. This study highlighted that, although some patients developed new cancers during treatment, there were no reports of tumor progression or recurrence attributable to dupilumab. Most patients exhibited a positive response to AD treatment with dupilumab, indicating good tolerability and efficacy in this context. 11
Conclusion
This clinical case demonstrates that dupilumab can be safely and effectively used in patients with AD and a history of metastatic renal cancer, providing disease control without evidence of neoplastic recurrence over 20 months of follow-up. Until more data are available, the decision to use dupilumab should be based on a careful individualized assessment involving a multidisciplinary team, including dermatologists, oncologists, and other relevant specialists, with continuous monitoring during treatment.
In conclusion, after reviewing current published evidence, the use of dupilumab in patients with a history of cancer appears to be a viable and safe option, particularly in patients with solid neoplasms, as dupilumab can effectively control AD without promoting cancer progression. However, additional studies are necessary to confirm the long-term safety of dupilumab in this specific population, for which limited systemic therapeutic options are available despite its growing size. To establish robust scientific evidence and form precise and objective guidelines, future studies should be randomized, controlled, and large-scale.
Footnotes
Acknowledgements
We would like to thank the staff at the Department of Dermatology, Centro Hospitalar Universitário do Porto, for their invaluable support in the management of the case described.
Author contributions
César Ferreira, Egídio Freitas, and Tiago Torres conceived the idea for the article, performed the literature search, data analysis, and drafted as well as critically revised the manuscript.
Consent to publish
The patient in this manuscript has given written informed consent to the publication of his case details.
Data availability statement
Summary of reported cases of dupilumab treatment for atopic dermatitis in patients with a previous history of cancer.
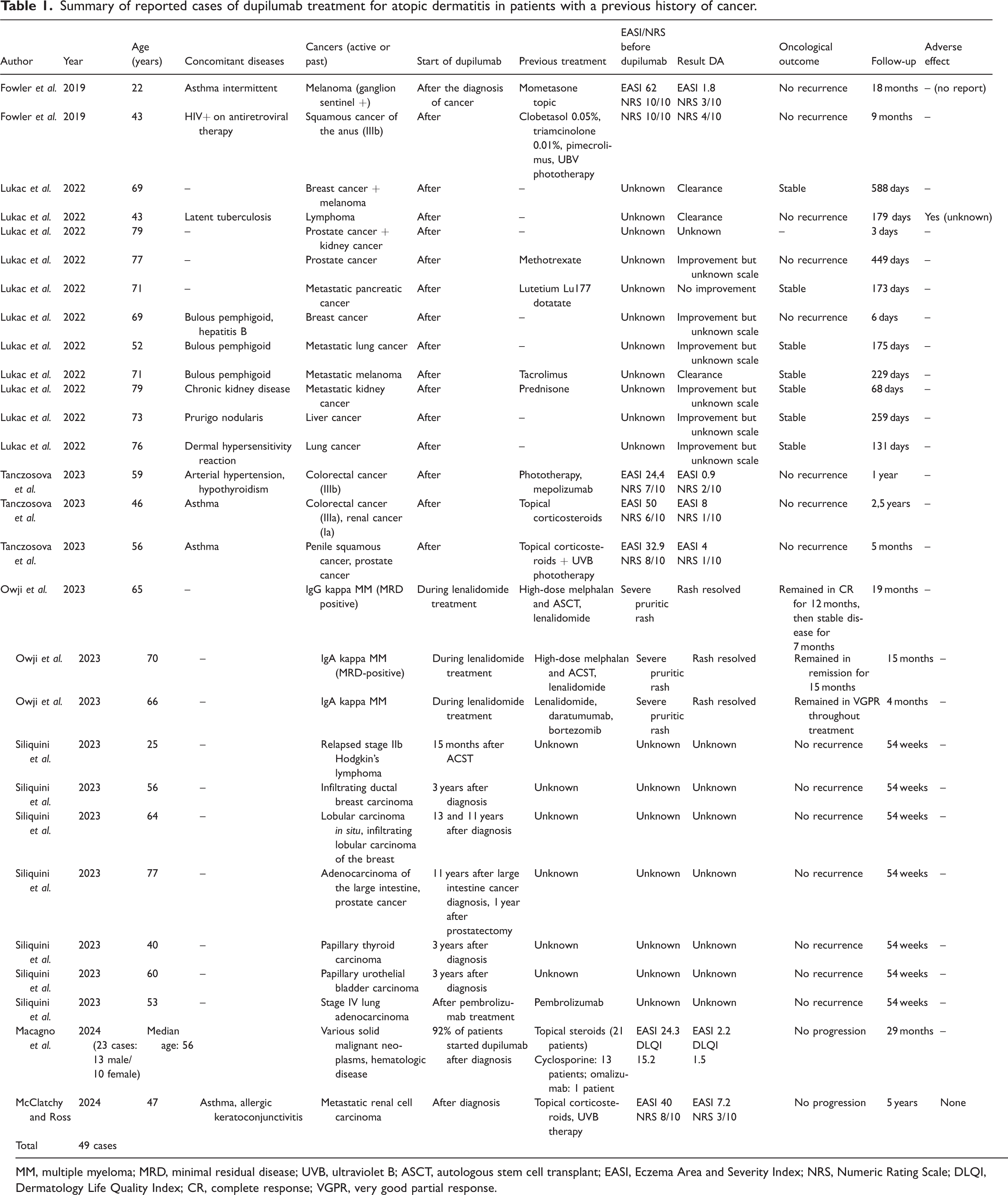
| Author | Year | Age (years) | Concomitant diseases | Cancers (active or past) | Start of dupilumab | Previous treatment | EASI/NRS before dupilumab | Result DA | Oncological outcome | Follow-up | Adverse effect |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Fowler |
2019 | 22 | Asthma intermittent | Melanoma (ganglion sentinel +) | After the diagnosis of cancer | Mometasone topic | EASI 62NRS 10/10 | EASI 1.8NRS 3/10 | No recurrence | 18 months | – (no report) |
| Fowler |
2019 | 43 | HIV+ on antiretroviral therapy | Squamous cancer of the anus (IIIb) | After | Clobetasol 0.05%, triamcinolone 0.01%, pimecrolimus, UBV phototherapy | NRS 10/10 | NRS 4/10 | No recurrence | 9 months | – |
| Lukac |
2022 | 69 | – | Breast cancer + melanoma | After | – | Unknown | Clearance | Stable | 588 days | – |
| Lukac |
2022 | 43 | Latent tuberculosis | Lymphoma | After | – | Unknown | Clearance | No recurrence | 179 days | Yes (unknown) |
| Lukac |
2022 | 79 | – | Prostate cancer + kidney cancer | After | – | Unknown | Unknown | – | 3 days | – |
| Lukac |
2022 | 77 | – | Prostate cancer | After | Methotrexate | Unknown | Improvement but unknown scale | No recurrence | 449 days | – |
| Lukac |
2022 | 71 | – | Metastatic pancreatic cancer | After | Lutetium Lu177 dotatate | Unknown | No improvement | Stable | 173 days | – |
| Lukac |
2022 | 69 | Bulous pemphigoid, hepatitis B | Breast cancer | After | – | Unknown | Improvement but unknown scale | No recurrence | 6 days | – |
| Lukac |
2022 | 52 | Bulous pemphigoid | Metastatic lung cancer | After | – | Unknown | Improvement but unknown scale | Stable | 175 days | – |
| Lukac |
2022 | 71 | Bulous pemphigoid | Metastatic melanoma | After | Tacrolimus | Unknown | Clearance | Stable | 229 days | – |
| Lukac |
2022 | 79 | Chronic kidney disease | Metastatic kidney cancer | After | Prednisone | Unknown | Improvement but unknown scale | Stable | 68 days | – |
| Lukac |
2022 | 73 | Prurigo nodularis | Liver cancer | After | – | Unknown | Improvement but unknown scale | Stable | 259 days | – |
| Lukac |
2022 | 76 | Dermal hypersensitivity reaction | Lung cancer | After | – | Unknown | Improvement but unknown scale | Stable | 131 days | – |
| Tanczosova |
2023 | 59 | Arterial hypertension, hypothyroidism | Colorectal cancer (IIIb) | After | Phototherapy, mepolizumab | EASI 24,4NRS 7/10 | EASI 0.9NRS 2/10 | No recurrence | 1 year | – |
| Tanczosova |
2023 | 46 | Asthma | Colorectal cancer (IIIa), renal cancer (Ia) | After | Topical corticosteroids | EASI 50NRS 6/10 | EASI 8NRS 1/10 | No recurrence | 2,5 years | – |
| Tanczosova |
2023 | 56 | Asthma | Penile squamous cancer, prostate cancer | After | Topical corticosteroids + UVB phototherapy | EASI 32.9NRS 8/10 | EASI 4NRS 1/10 | No recurrence | 5 months | – |
| Owji |
2023 | 65 | – | IgG kappa MM (MRD positive) | During lenalidomide treatment | High-dose melphalan and ASCT, lenalidomide | Severe pruritic rash | Rash resolved | Remained in CR for 12 months, then stable disease for 7 months | 19 months | – |
| Owji |
2023 | 70 | – | IgA kappa MM (MRD-positive) | During lenalidomide treatment | High-dose melphalan and ACST, lenalidomide | Severe pruritic rash | Rash resolved | Remained in remission for 15 months | 15 months | – |
| Owji |
2023 | 66 | – | IgA kappa MM | During lenalidomide treatment | Lenalidomide, daratumumab, bortezomib | Severe pruritic rash | Rash resolved | Remained in VGPR throughout treatment | 4 months | – |
| Siliquini |
2023 | 25 | – | Relapsed stage IIb Hodgkin’s lymphoma | 15 months after ACST | Unknown | Unknown | Unknown | No recurrence | 54 weeks | – |
| Siliquini |
2023 | 56 | – | Infiltrating ductal breast carcinoma | 3 years after diagnosis | Unknown | Unknown | Unknown | No recurrence | 54 weeks | – |
| Siliquini |
2023 | 64 | – | Lobular carcinoma |
13 and 11 years after diagnosis | Unknown | Unknown | Unknown | No recurrence | 54 weeks | – |
| Siliquini |
2023 | 77 | – | Adenocarcinoma of the large intestine, prostate cancer | 11 years after large intestine cancer diagnosis, 1 year after prostatectomy | Unknown | Unknown | Unknown | No recurrence | 54 weeks | – |
| Siliquini |
2023 | 40 | – | Papillary thyroid carcinoma | 3 years after diagnosis | Unknown | Unknown | Unknown | No recurrence | 54 weeks | – |
| Siliquini |
2023 | 60 | – | Papillary urothelial bladder carcinoma | 3 years after diagnosis | Unknown | Unknown | Unknown | No recurrence | 54 weeks | – |
| Siliquini |
2023 | 53 | – | Stage IV lung adenocarcinoma | After pembrolizumab treatment | Pembrolizumab | Unknown | Unknown | No recurrence | 54 weeks | – |
| Macagno |
2024 (23 cases: 13 male/10 female) | Median age: 56 | Various solid malignant neoplasms, hematologic disease | 92% of patients started dupilumab after diagnosis | Topical steroids (21 patients) Cyclosporine: 13 patients; omalizumab: 1 patient | EASI 24.3DLQI15.2 | EASI 2.2DLQI1.5 | No progression | 29 months | – | |
| McClatchy and Ross | 2024 | 47 | Asthma, allergic keratoconjunctivitis | Metastatic renal cell carcinoma | After diagnosis | Topical corticosteroids, UVB therapy | EASI 40 NRS 8/10 | EASI 7.2 NRS 3/10 | No progression | 5 years | None |
| Total | 49 cases | ||||||||||
MM, multiple myeloma; MRD, minimal residual disease; UVB, ultraviolet B; ASCT, autologous stem cell transplant; EASI, Eczema Area and Severity Index; NRS, Numeric Rating Scale; DLQI, Dermatology Life Quality Index; CR, complete response; VGPR, very good partial response.
Declaration of conflicting interests
César Ferreira and Egídio Freitas declare no conflicts of interest.
Tiago Torres declares the following conflicts of interest: AbbVie, Amgen, Almirall, Arena Pharmaceuticals, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, Janssen, Biocad, LEO Pharma, Eli Lilly, MSD, Novartis, Pfizer, Samsung-Bioepis, Sanofi-Genzyme, and Sandoz.
Funding
No funding has been received for the preparation of this manuscript.
