Abstract
In recent years, radiomics has emerged as a novel research methodology that plays a crucial role in the diagnosis and treatment of ischemic stroke. By integrating multimodal medical imaging techniques such as computed tomography and magnetic resonance imaging, radiomics offers in-depth insights into aspects such as the extent of brain tissue damage and hemodynamics. These data help physicians to accurately assess patient condition, select optimal treatment strategies, and predict recovery trajectories and long-term prognoses, thereby enhancing treatment efficacy and reducing the risk of complications. With the anticipated further advancements in radiomic technology, this methodology has great potential for expanded applications in the early detection, treatment, and prognosis of ischemic stroke. The present narrative review explores the burgeoning field of radiomics and its transformative impact on ischemic stroke.
Ischemic stroke
Stroke is the second leading cause of death globally and ranks as the third major cause of death and disability. Its incidence is progressively increasing, especially in developing nations. 1 In China, a 2020 study revealed a 2.6% stroke prevalence among adults over 40 years, highlighting the increasing burden of stroke—predominantly ischemic stroke—and emphasizing the need for improved prevention and management strategies.2,3 The majority of stroke cases manifest as ischemic strokes, which are primarily derived from arterial occlusions. 4 The definition of stroke encapsulates the acute onset of focal functional impediments in the brain, retina, or spinal cord persisting beyond 24 hours, or of any duration if imaging (computed tomography [CT] or magnetic resonance imaging [MRI]) or autopsy reveals a focal infarction or hemorrhage congruent with the observed symptoms. 5 Stroke can be broadly categorized into hemorrhagic stroke (such as cerebral hemorrhage) and ischemic stroke (cerebral infarction). Ischemic stroke is a consequence of cerebral vascular insufficiency leading to ischemia and hypoxia, and constitutes roughly 80% of all strokes. It results in localized cerebral tissue necrosis or softening, and commonly encompasses cerebral thrombosis, lacunar infarction, and cerebral embolism. Research indicates that pivotal risk factors for stroke encompass a history of hypertension, smoking, high waist-to-hip ratio, high dietary risk scoring, low regular physical activity, and diabetes. 6 Targeted interventions, such as blood pressure reduction, smoking cessation, promotion of physical activity, and adoption of a wholesome diet, can significantly alleviate the burden of stroke. 7 Furthermore, recent investigations have underscored the importance of early identification, emergent intervention, and treatment within specialized stroke centers for acute ischemic stroke; these can substantially mitigate stroke incidence and fatality. 8
The foremost direct etiology of ischemic stroke primarily stems from thromboembolism, which can be divided into cerebral thrombosis and cerebral embolism based on their distinct causative origins. Cerebral thrombosis is precipitated by the stenosis or occlusion of cerebral arteries as a result of cerebral atherosclerosis. This leads to the abrupt cessation of local cerebral blood supply, thereby causing local neurological symptoms and signs. From a broader perspective, other than atherosclerosis, factors like small vessel disease, arterial dissection, and cerebral vasculitis may also incite stroke by affecting cerebral vasculature.9,10 By contrast, cerebral embolism denotes the acute occlusion of cerebral arteries caused by various emboli—solid, liquid, or gaseous—that traverse the bloodstream and enter intracranial arteries. Cardioembolic emboli predominate, and often arise from conditions such as atrial fibrillation, patent foramen ovale, or infective endocarditis. 11 Notably, recent research indicates that thrombus imaging characteristics can predict post-thrombectomy recanalization efficacy following acute ischemic stroke. 12
Foundational notions of radiomics
In the context of ischemic cerebrovascular stroke, treatment efficacy depends on the early identification of etiology and the prompt administration of reperfusion methods. 13 To diagnose stroke, it needs to be discriminated from an array of other neurological disorders that present similar clinical manifestations. Cerebral imaging therefore has a pivotal role in stroke diagnosis. 14 Multimodal medical imaging techniques are also important beyond diagnosis, such as in the treatment and prognostic assessment of stroke patients.
Compared with evaluations using conventional clinical radiographic scans, imaging radiomics has advantages in terms of its non-invasiveness, non-destructiveness, cost-effectiveness, rapid analysis, and ease of serialization. 15 Imaging radiomics thus constitutes a rapidly evolving research domain, and is centered around the quantifiable extraction of metrics, referred to as radiomic features, from medical images. These radiomic features capture tissue and pathological attributes, such as heterogeneity and shape, and are amenable to synergistic amalgamation with artificial intelligence for clinical problem-solving (i.e., the basic radiomic workflow). 16 The course of imaging radiomics typically encompasses the formulation of clinical queries, acquisition and pre-processing of imaging data, image segmentation, feature extraction, and model construction and validation. 17 Within this framework, conventional radiologists typically engage in the visual interpretation of medical images, and draw upon their own intuitive and extensive experience to diagnose ailments. This approach can lead to a range of differences as a result of individual variances in expertise. Nonetheless, these imaging datasets inherently encapsulate a wealth of objective latent information, and this is where the quintessential stages of radiomics come into play; the extraction of high-throughput features is a requisite for analyzing substantive attributes within regions of interest. Such an approach, underpinned by these substantive attributes, can yield heightened diagnostic uniformity.
There are many reported examples of the application of radiomics in the context of neurological disorders. 17 In patients with brain cancer, radiomics has demonstrated potential for non-invasively discerning prognostic biomarkers, performing automated response assessments, and discriminating treatment-related changes linked to tumor progression. 18 Within the realm of other central nervous system disorders, the use of regional radiomics similarity networks has allowed the identification of distinct subtypes and aberrant patterns associated with mild cognitive impairment. 19 Furthermore, in the context of acute ailments, the integration of cranial CT information and clinical data in prognostic models has enabled the prediction of long-term outcomes following severe traumatic brain injury. 20 The neural system’s relative susceptibility and severe prognoses require non-invasive diagnostic and therapeutic paradigms, formed upon existing datasets, that cater to the needs of clinical practice.
The exponential surge in medical imaging data has created an ideal setting for machine learning and data-driven scientific endeavors. 21 Imaging radiomics has great potential and many advantages across diverse domains, including disease diagnosis, molecular differentiation, and prognostic determination.22–24 There is thus a need to encapsulate the recent imaging radiomic-related developments in the domain of ischemic cerebrovascular stroke, not only as a retrospective summary, but also as a guide for future advancements in related fields (Figure 1).

Basic workflow of ischemic stroke imaging radiomics.
In the present narrative review, we describe clinical issues related to stroke and discuss rapidly advancing technologies, such as radiomics. We also explore the application of radiomics for addressing clinical problems associated with stroke. Currently, many potential applications of radiomic analysis have been reported in various aspects of stroke, including for the diagnosis of stroke lesions, early prediction of outcomes, and assessment of long-term prognosis. Here, we aim to provide an overview of the research findings related to stroke radiomic analysis. We specifically focus on the key methods and research approaches that have been obtained through the application of radiomics in the early diagnosis of stroke, prediction of treatment outcomes, and assessment of prognosis. Additionally, we discuss the shortcomings and gaps present in the existing literature.
Search strategy
In the present study of the application of radiomics in ischemic stroke, we implemented an extensive search strategy with the following retrieval keywords: (“radiomics”[MeSH Terms] OR “radiomics”[All Fields] OR “radiomic”[All Fields]) AND (“ischemic stroke”[MeSH Terms] OR (“ischemic”[All Fields] AND “stroke”[All Fields]) OR “ischemic stroke”[All Fields]).
We focused our search on the MEDLINE and PubMed databases and targeted articles published between 2017 and 2023. Our search terms were carefully selected to capture studies related to ischemic stroke and the emerging field of radiomics. To ensure thorough coverage, we also conducted manual searches and checked the references of key papers. The initial screening involved reviewing titles and abstracts; this was followed by a full-text assessment of potentially relevant articles. The final inclusion of papers in our study was based on a consensus among all coauthors, with a focus on works that explored the applications of radiomics in diagnosing, treating, and assessing prognosis in ischemic stroke. This strategy was designed to provide a comprehensive and relevant collection of literature to support our analysis.
Application of imaging radiomics in ischemic cerebrovascular stroke
The importance of imaging information in the context of ischemic cerebrovascular stroke, and the robust complementarity provided by imaging radiomic features to conventional imaging data, have accelerated the advancement of diagnosis and treatment in this domain. The following discussion centers on common clinical imaging data and progress within this field (Figure 2), encompassing three main facets: early detection, treatment, and prognosis (Table 1).
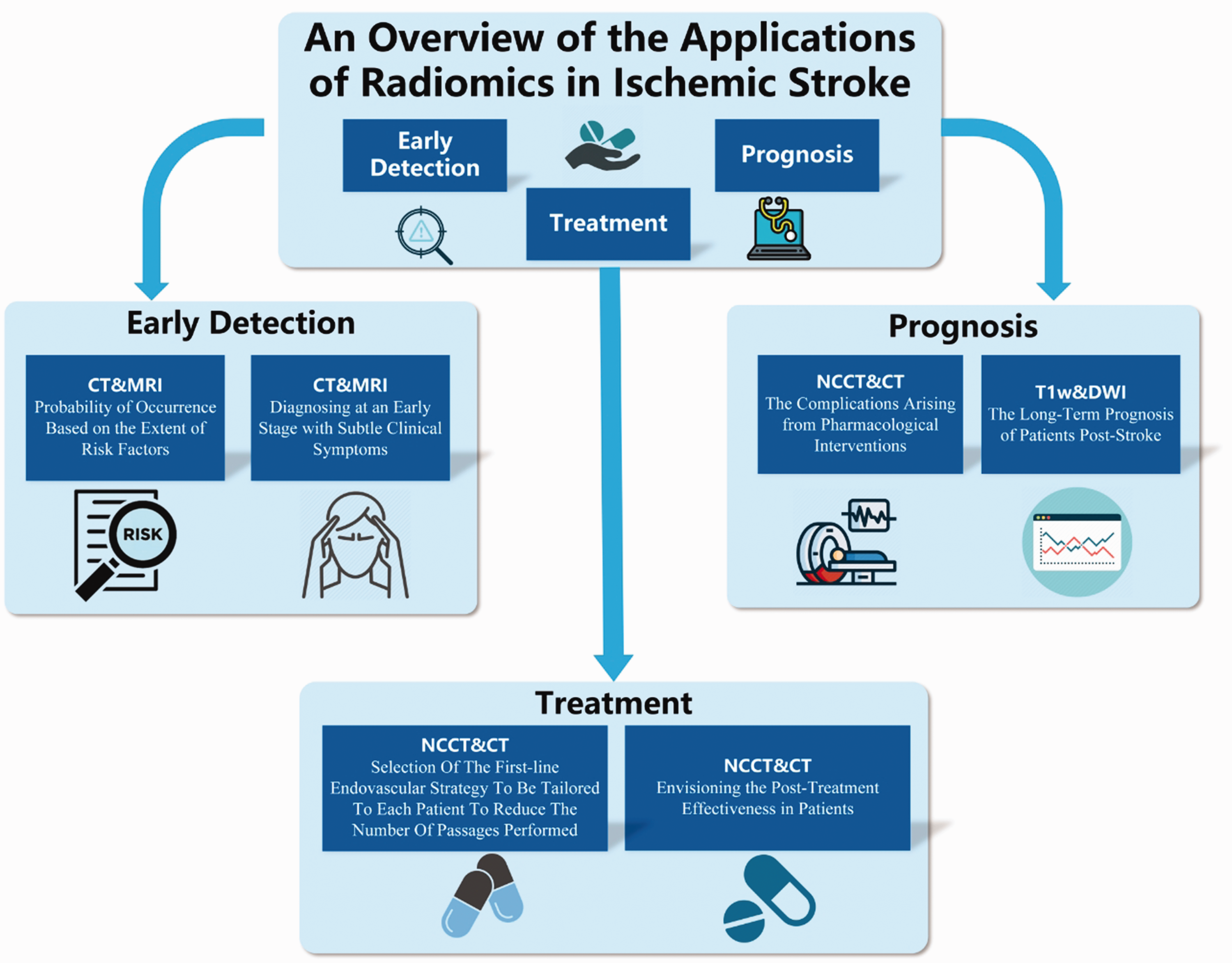
Overview of the applications of radiomics in stroke. CT, computed tomography; DWI, diffusion-weighted imaging; MRI, magnetic resonance imaging; NCCT, non-contrast computed tomography; T1w, T1-weighted.
Studies into the applications of radiomics for ischemic stroke management.

CT, computed tomography; DSC-PWI, dynamic susceptibility contrast perfusion-weighted imaging; DWI, diffusion-weighted imaging; FLAIR, fluid-attenuated inversion recovery; MRI, magnetic resonance imaging; NCCT, non-contrast computed tomography; No., number; T1w, T1-weighted.
Early detection
Ischemic cerebrovascular stroke can be highly debilitating and fatal, and requires early detection and intervention for effective treatment. A study has suggested that during the COVID-19 pandemic, delayed treatment—likely because of the perceived risk of infectious disease—correlated with higher in-hospital mortality rates. 25 Early detection embodies two aspects: predicting the probability of occurrence based on the extent of risk factors, and diagnosing at an early stage based on subtle clinical symptoms.
To predict the probability of stroke based on risk factors, Bretzner et al. 26 proposed predicting stroke risk using the relative brain age, derived from T2-fluid-attenuated inversion recovery images acquired during acute stroke clinical assessments. These authors established that T2-fluid-attenuated inversion recovery-related radiomic features can be used to predict brain age and calculate relative brain age; the predicted brain age then serves as an indicator of cumulative cardiovascular risk factors and post-stroke outcomes. Additionally, Su et al. successfully built a risk model to predict future ischemic stroke in asymptomatic lacunar infarction patients using radiomic feature extraction from CT images; they identified that factors including age, blood lipids, and multiple infarctions were independently associated with future ischemic stroke. 27
When considering the diagnosis of stroke at an early stage, evidence supports the benefits of intravenous tissue plasminogen activator administration within 4.5 hours after symptom onset in ischemic stroke patients. 28 Wen and colleagues 29 leveraged this knowledge to extract radiomic features from CT images to determine the infarction onset time in patients with acute middle cerebral artery occlusion. They concluded that radiomic features were able to guide thrombolytic therapy in patients with uncertain stroke onset time. Furthermore, to recognize early ischemic stroke, Kuang et al. 30 designed a radiomics-related scoring system to objectively score patients’ CT scans and aid in their early diagnosis. Similarly, Ortiz-Ramón and colleagues 31 used 1800 sets of three-dimensional MRI data from three prospective studies to determine the presence of ischemic lesions using the radiomic features of 114 structural attributes, such as brain ventricles and basal ganglia. Moreover, Zhang et al. 32 constructed a predictive model for the onset time of ischemic stroke based on MRI radiomic features, achieving an area under the curve (AUC) of 0.754 for predicting whether stroke had occurred within 4.5 hours. These findings may facilitate stroke assessment and treatment decisions. In addition, through the construction of these models, it becomes possible to quantify the post-stroke time in patients, thus enabling the optimal timing for medication administration and enhancing patient prognosis.
The Alberta Stroke Program Early CT Score is often used to assess the severity of early ischemic stroke in patients. Kuang et al. 30 devised an image-based model with intertwined radiomic features, thereby achieving the automated scoring of patients’ CT scans. Encouragingly, the intragroup correlation coefficient between the model’s outcomes and those of expert evaluations was 0.76. The expansion of radiomics as a field is largely thanks to its easy integration with artificial intelligence, thus facilitating automated image segmentation and result generation. In this way, it can help clinical practitioners to make clinical judgments.
Radiomics is also very useful from a binary classification perspective, such as for discerning the presence or absence of ischemic cerebral infarction lesions. For example, Ortiz-Ramón et al. 31 harnessed 1800 sets of three-dimensional MRI data from three prospective studies of stroke mechanisms (one study) and cognitive aging (two studies). The authors assessed 114 structural features, including cerebrospinal fluid and deep gray matter, which culminated in the identification of stroke-type lesions. Additionally, Dogan et al. 33 used computations and standardization to quantify the mean apparent diffusion coefficient of ischemic voxels within the ischemic territory of patients with isolated pontine infarction. By comparing two cohorts—one displaying neurological deterioration and the other non-deterioration—they ascertained a close correlation between the average relative apparent diffusion coefficient value within ischemic lesions and early neurofunctional deterioration in patients with isolated pontine infarction.
Treatment
Radiomics and related technologies play a crucial role in the treatment of ischemic stroke. These technologies are typically used to ascertain the potential benefit of specific therapeutic interventions for individual patients. Additionally, they are instrumental in predicting the therapeutic efficacy of various pharmacological agents.
The application of radiomics for identifying patients who are likely to benefit from specific treatment methods is gaining importance. Mechanical thrombectomy is a reference treatment for acute ischemic stroke, and there are currently various endovascular treatment strategies. 34 However, a quantitative method to select the optimal endovascular strategy or to predict the difficulty of thrombus removal is not yet available. To investigate this, Hofmeister et al. 35 constructed a retrospective training cohort (n = 109) and a prospective validation cohort (n = 47), and then developed two machine learning models. Following the selection of radiomics-related features, these models were used to predict the success of first-pass recanalization via thrombus aspiration, as well as the total number of passes required for successful recanalization using mechanical thrombectomy devices. The performance of these models was then evaluated in the prospective validation cohort. The study concluded that radiomics based on the thrombus can predict the success of mechanical thrombectomy-based recanalization strategies in acute ischemic stroke, thereby enabling the better selection of mechanical thrombectomy strategies and identifying the patients most likely to benefit from these interventions (Figure 2).
Venous thrombolysis is the only widely approved systemic reperfusion therapy for patients with acute ischemic stroke. 36 Alteplase is the most common pharmacotherapeutic approach for acute ischemic stroke patients. 37 Many investigations have highlighted the potential of combining radiomic and clinical data features into clinical prediction models, which may allow the precise prediction of patients who are likely to have favorable reperfusion outcomes.38–40
It has been reported that thrombus radiomic features may be useful for predicting early recanalization following venous alteplase administration. 41 Derived from non-contrast CT and CT angiography, these radiomic features have superior predictive abilities for venous alteplase recanalization compared with conventional thrombus imaging attributes such as length, volume, and permeability. Notably, an amalgamation of radiomic features from non-contrast CT, CT angiography, and radiomic changes has emerged as the most closely aligned with the success of early venous alteplase recanalization (AUC = 0.85).
Prognosis
The mortality rate following stroke is approximately 15% at 1 month, 25% at 1 year, and 50% at 5 years. 5 Moreover, according to the 10-year Oxford Vascular Study, approximately 40% of stroke survivors experience disabling stroke between 1 month and 5 years after a cerebral hemorrhage, and 20% exhibit pre-stroke disabilities. 42 Precisely gauging a patient’s prognosis is therefore an essential part of the stroke diagnostic and therapeutic process. When an unfavorable prognosis is likely, early intervention measures can be escalated, thereby maximizing the chances of patient survival.
In the present section, two aspects of prognosis are primarily discussed: first, complications arising from pharmacological interventions, and second, the long-term prognosis of patients post-stroke (Figure 2). Systemic thrombolysis via intravenous administration methods is a common treatment for patients with acute ischemic stroke. However, venous thrombolysis is often accompanied by the complication of intracerebral hemorrhage transformation. 43 To predict hemorrhage transformation, Meng et al. 44 introduced a multifaceted MRI radiomics-based model coupled with machine learning. Four distinct predictive models were cultivated within a primary training cohort and subsequently validated using an independent dataset. The combined model, comprising both clinical and radiomic features, was particularly good at predicting hemorrhage transformation among patients with acute ischemic stroke (AUC = 0.911). By contrast, when features were extracted solely from anomalous regions (i.e., excluding those originating from normal regions of interest) the classification accuracy of predictions was significantly worse (AUC = 0.831).
Another severe complication after post-ischemic stroke is malignant cerebral edema, which leads to adverse prognoses or fatality. Using a methodology termed image patch-based net water uptake, Fu et al. 45 used non-contrast CT exclusively for the prediction of malignant cerebral edema. This approach may help to identify patients who are likely to benefit from decompressive hemicraniectomy, thereby improving their prognoses.
The differentiation between contrast agent extravasation and post-thrombectomy hemorrhage is often difficult using imaging features; this challenge was addressed in studies by Ma et al. 46 and Chen et al. 47 By integrating radiomic features and incorporating multiple independent predictive factors, these researchers constructed models that were proficient at distinguishing between hemorrhage and contrast agent extravasation. These models are clinically important because they facilitate the swift identification of aberrant signals in images, thereby helping clinicians to make prompt and accurate decisions and enhancing patient treatment.
Betrouni et al. 48 revealed that that imaging features derived from the texture analysis of T1-weighted images are promising biomarkers for post-stroke cognitive impairment, with higher sensitivity than atrophy measurements. In a cohort of 327 patients from stroke and cognition consortia in France, the UK, and other nations, predictive modeling demonstrated that texture features extracted from routine clinical MRI are reliable early prognostic factors for post-stroke cognitive impairment. Furthermore, a more accurate predictive model was constructed when these features were combined with other demographic and clinical predictive factors.
Liang et al. 49 divided 1716 acute ischemic stroke patients from two centers into training and validation sets before constructing the CrrScore model, which used both clinical and radiological features. The CrrScore model was able to effectively predict outcomes for patients with acute ischemic stroke and might be useful for guiding rehabilitation treatments in patients with different risks of adverse outcomes. Liu et al. 50 used modeling data from 612 diagnosed acute ischemic stroke patients to devise a model for predicting ischemic stroke recurrence within 1 year of discharge. In this model, multiple imaging and clinical features that influence acute ischemic stroke recurrence were identified.
As gradual advancements and novel iterations of prognosis prediction models continue to emerge, 51 our understanding of ischemic stroke will improve, as will our comprehension and assessment of patient prognoses.
Summary
Current limitations and constraints
Many different studies have used diverse imaging acquisition and feature extraction protocols, 1 and multiple research efforts have highlighted the effects of differences in data collection, image reconstruction, and post-processing on downstream analyses. 52 Reproducibility of data outcomes is also lacking, and some radiomic studies have issues such as small sample sizes and inadequate dataset and test set divisions. These factors might lead to overly optimistic results and the overestimation of research outcomes. 53 Furthermore, the majority of existing radiomic research entails the manual segmentation of images by experienced radiologists. This manual image segmentation process is not only resource-intensive in terms of time and effort, but is also inherently prone to interobserver discrepancies. It is therefore imperative to develop a sophisticated, fully automated segmentation methodology that is characterized by high precision and robustness. Such an approach should aim to mitigate these errors to the greatest possible extent.
We also note that many radiomic models lack interpretability in their outputs. For example, critical thresholds are arbitrarily set at specific percentiles, such as the median, to define high-risk and low-risk groups, and the connections between clinical outcomes and these grouping methods are often disregarded. 54
Although a variety of radiomic models have been developed for the diagnosis and treatment of ischemic cerebrovascular stroke, certain deficiencies remain in terms of clinical application and acceptance. These issues fundamentally arise from the absence of unified standards within the field. With the diverse array of imaging protocols, data preprocessing methods, feature extraction techniques, and model validation procedures used by disparate researchers during their investigations, the resultant variability might impede the use of radiomic research outcomes within a singular research domain. Similarly, in the absence of a universally embraced set of standards, the credibility and dependability of radiomic models may face scrutiny from clinical practitioners and healthcare professionals.
Future prospects
In recent years, numerous innovative research findings—including from the field of imaging—have been applied to ischemic cerebrovascular stroke. These results suggest that the application of radiomics in ischemic stroke may help to further narrow diagnostic time windows and enable more precise prognostic assessments. Furthermore, to address the drawbacks of traditional MRI, such as lengthy procedures, high costs, and susceptibility to interference, researchers have used radiomics and other methods to generate MRI images from CT. 55 This finding illustrates the breakthrough potential and flexibility of radiomic technology. In addition, the increasing prevalence of mobile stroke units and advanced neuroimaging may enhance the number of patients receiving thrombolysis, both by shortening the time from onset to treatment and by identifying salvageable penumbra regions. 56 Radiomics-based applications may further reduce this time.
Sepsis can lead to arrhythmia, with atrial fibrillation as the most prevalent manifestation. 57 In turn, atrial fibrillation is an important etiological factor for cerebral embolism. The prompt detection of post-stroke cerebral embolism endeavors to ameliorate prognosis, because a considerable portion of patients face unfavorable prognoses. As mentioned earlier in the present review, the performance of combined clinical- and radiomics-based models tends to surpass that of models based solely on clinical variables. The use of radiomic techniques therefore enhances the prediction of adverse outcomes in patients with acute stroke.
Although the field of radiomics is currently facing challenges and difficulties, its application remains promising. Moreover, the integration of various omics analyses—including radiomics—will undoubtedly represent a more comprehensive use of data.
The field of medical imaging informatics is currently flourishing. In this emerging domain, any endeavors to forcibly establish uniform standards might impede the emergence of novel ideas and methodologies. However, we are not opposed to the establishment of universally acknowledged norms. Rather, we believe that innovation should be encouraged across various facets of medical imaging informatics, from image segmentation to feature extraction. This approach diverges from the mere replication of techniques within the same domain, and advocates for genuine, substantive innovations. Furthermore, we note that current related research has usually been conducted using retrospective studies. Building upon the achievements to date, multicenter prospective studies should be performed to further validate the effectiveness and advantages of radiomics.
Footnotes
Acknowledgements
We would like to acknowledge Medpeer (www.medpeer.cn) for assistance in creating ![]() .
.
Author contributions
Jie Yang: Writing-Original draft preparation; Huabo Cai: Conceptualization, Methodology; Ning Liu: Writing-Reviewing and Editing; Jiajie Huang: Writing-Reviewing and Editing; Yun Pan: Visualization; Bo Zhang: Manuscript Revision; Mingfeng Tong: Investigation and Editing; Zhongheng Zhang: Supervision.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
Zhongheng Zhang received funding from the China National Key Research and Development Program (2022YFC2504503, 2023YFC3603104), a collaborative scientific project co-established by the Science and Technology Department of the National Administration of Traditional Chinese Medicine and the Zhejiang Provincial Administration of Traditional Chinese Medicine (GZY-ZJ-KJ-24082), the General Health Science and Technology Program of Zhejiang Province (2024KY1099), the Huadong Medicine Joint Funds of the Zhejiang Provincial Natural Science Foundation of China (LHDMD24H150001), the National Natural Science Foundation of China (82272180), and the Project of Drug Clinical Evaluate Research of Chinese Pharmaceutical Association (CPA-Z06-ZC-2021–004). Ning Liu was supported by the Fundamental Research Funds for the Central Universities (226-2023-00123). Mingfeng Tong was supported by Key Project of Jinhua City (2020-3-038, 2023-3-90, 2022-3-102, 2023-1-099).
