Abstract
Objective
While several laboratory variables have been used to assess COVID-19 disease, to our knowledge, no attempt has previously been made to compare differences across different patient groups. We attempted to evaluate the relationship between laboratory variables and severity of the disease as well as on prognosis.
Method
We searched BioLINCC database and identified three studies which had separately included outpatients, inpatients, and ICU patients. For this re-analysis, we extracted data on general demography, laboratory variables and outcome.
Result
In total, 2454 participants (496 outpatients [Study 1], 478 inpatients [Study 2], and 1480 ICU patients [Study 3]) were included in the analysis. We found three laboratory variables (i.e., creatinine, aspartate transferase, and albumin) were not only prognostic factors for outcome of inpatients with COVID-19, but also reflected disease severity as they were significantly different between inpatients and ICU patients. These three laboratory variables are an indication of kidney function, liver function, and nutritional status.
Conclusion
For patients with COVID-19, in addition to monitoring infectious disease indicators, we need to pay attention to liver function, renal function, and take timely measures to correct them to improve prognosis.
Introduction
The fight against coronavirus disease 2019 (COVID-19) has continued for years after the first outbreak, and the current emphasis is on diagnosis and treatment, including the use of traditional Chinese medicine. 1 Many clinical studies have been registered in the ClinicalTrials.gov database and have been performed since the first COVID-19 outbreak. 2 Since laboratory variables are often used to assess body function and disease severity, they are widely used in the assessment of COVID-19. 3 Increased levels of several inflammatory biomarkers, including C-reactive protein (CRP), have been found in patients with COVID-19 and are associated with an increased risk of severe disease and are described as a ‘cytokine storm’. 4
Several studies have evaluated COVID-19 using laboratory variables. Indeed, one study was able to distinguish laboratory variables for COVID-19 from community-acquired pneumonia. 5 In addition, several laboratory variables show abnormalities with COVID-19 disease progression. 6 Using this correlation, one study compared laboratory variables in mild vs severe COVID-19 disease and from patients who had died versus those who survived. 7 Some researchers have reported that severe cases were associated with old age, male sex, fever, cough, respiratory diseases, increased white blood cell count (WBC), CRP, D-dimer, aspartate aminotransferase (AST), and lactate dehydrogenase (LDH) levels. 8 Other researchers have compared clinical characteristics and laboratory findings of patients with and without COVID-19 complications and identified important risk factors for complications and death. 9 In another study, a risk stratification model was constructed based on laboratory variables to help diagnose, monitor, and predict severity in the early stages of COVID-19. 10 Furthermore, laboratory findings can be used to distinguish between COVID-19 and non-COVID-19 conditions. 10 However, existing studies have omitted certain laboratory variables, especially commonly used ones. Moreover, to our knowledge, no attempt has previously been made to compare differences across different patient groups such as outpatients, inpatients, and intensive care unit (ICU) patients. In this re-analysis of data from the BioLINCC public database (https://biolincc.nhlbi.nih.gov//) 11 we attempted to evaluate the relationship between laboratory variables and the severity of patients with COVID-19. We also evaluated the association between laboratory variables and outcome prognosis.
Methods
Data source
We searched the Biologic Specimen and Data Repository Information Coordinating Center (BioLINCC) resources for COVID-19 studies. BioLINCC contains individual-level data from more than 580,000 participants and more than 180 studies. 11 We identified three BioLINCC studies, COLCORONA, 12 PETAL-ORCHID, 13 and PETAL-RED CORAL, 14 which included outpatients, inpatients, and ICU patients, respectively.
Our analysis was performed on anonymized, clinical data from the BioLINCC database and so informed consent from the patients was not required. The study was approved by the Ethics Committee of Zhejiang Chinese Medical University (No:20230210-1). The reporting of this study conforms to STROBE guidelines. 15
Methodology
In this re-analysis of data from the BioLINCC public database, data were obtained from three COVID-19 studies that had separately included outpatients, inpatients, and ICU patients.12–14 Generally, for COVID-19 disease, outpatients are the least severe group, ICU patients are the most severe group, and inpatients are somewhere in the middle. The inclusion and exclusion criteria have been previously reported.12–14 Two researchers [LS and MH] independently extracted data from the BioLINCC database relating to the three studies. The following items were extracted: general demographic data; laboratory data; outcome data.
While creatinine levels were recorded in all three studies, only inpatients and ICU patients had the following variables measured: blood urea nitrogen (BUN); white blood cells (WBC); haemoglobin (Hb); platelet count (PLT); AST; alanine aminotransferase (ALT); total bilirubin (TBIL); albumin (ALB); partial thromboplastin time (PTT); international normalized ratio (INR).12–14 For units of the laboratory variables that were inconsistent, unit conversion was performed (Table 1). We determined ‘normal’, ‘low’, and ‘high’ values of the laboratory variables according to the relevant normal reference range.
Units and normal ranges for measured laboratory variables from the three studies.

ICU, intensive care unit; BUN, blood urea nitrogen; WBC, white blood cells; Hb, haemoglobin; PLT, platelet count; AST, aspartate transferase; ALT, alanine aminotransferase; TBIL, total bilirubin; ALB, albumin; PTT, partial thromboplastin time; INR, international normalized ratio.
To assess which laboratory variables could be prognostic factors for disease outcome, in the inpatient study 13 we grouped patients according to different outcomes (i.e., death or need for extracorporeal membrane oxygenation (ECMO), and survival/no need for ECMO) and compared differences in laboratory variables between the two groups.
Statistical analysis
The analysis was performed using GraphPad prism v9 and Sata v17.0 and P < 0.05 was considered statistically significant. Continuous variables were tested for normality and data that were skewed were described using median and interquartile ranges, and intergroup comparisons were performed using Wilcoxon rank-sum test. For normally distributed data, mean ± standard deviation (SD) were reported, and Student's t-test or analysis of variance (ANOVA) were used for intergroup comparison. Proportions were given as numbers and percentages. Comparison of groups was performed using ANOVA for continuous variables and χ2 test for categorical variables. To assess outcome predictors from the inpatient study, we used multivariate logistic regression analysis (trial intervention grouping as a correction variable and death/ECMO as the dependent variable) and calculated the odds ratio (OR) with 95% CIs.
Results
Study participants
In total, 2454 participants (496 outpatients [Study 1], 478 inpatients [Study 2], and 1480 ICU patients [Study 3]) were included in the analysis (Figure 1). The general demographic characteristics of the patients are presented in Figure 2. Proportions of male/female patients, ages and body mass index (BMI) were similar across the three studies.
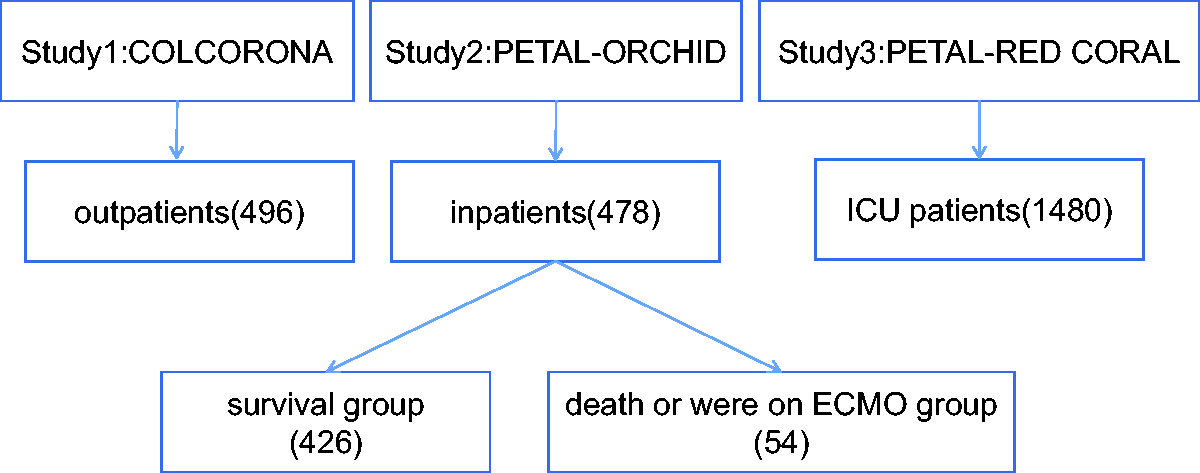
Study flow chart. Data were extracted from three BioLINCC studies, COLCORONA, 12 PETAL-ORCHID, 13 and PETAL-RED CORAL, 14 which included outpatients, inpatients, and intensive care unit (ICU) patients, respectively. Patients from the inpatient study 13 were separated into two groups according to outcomes (i.e., death or need for extracorporeal membrane oxygenation (ECMO) and survival/no need for ECMO).

(a–d) Demographic characteristics of the outpatients, inpatients, and intensive care unit (ICU) patients, (i.e., number, age, sex and body mass index [BMI]).
Differences in laboratory variables
There were statistically significant (P < 0.05) differences between the inpatient (Study 2) and ICU groups (Study 3) in several laboratory variables (i.e., Hb, PLT, AST, ALT, TBIL, and ALB,) (Table 2). Inpatients had higher median values than ICU patients for, PLT, AST, and ALT and lower median values for creatinine. There were also statistically significant (P < 0.05) differences between the inpatient and ICU groups in the proportion of patients with low, normal or high concentrations of several laboratory variables (i.e., Hb, PLT, AST, ALT, ALB, PTT and INR) (Table 3).
Comparison of laboratory variables between Study 2 (inpatients) and Study 3 (ICU patients).

ICU, intensive care unit; IQR, interquartile range; BUN, blood urea nitrogen; WBC, white blood cells; Hb, haemoglobin; PLT, platelet count; AST, aspartate transferase; ALT, alanine aminotransferase; TBIL, total bilirubin; ALB, albumin; PTT, partial thromboplastin time; INR, international normalized ratio.
Comparison of low/normal/high values of laboratory variables (with respect to normal ranges) between Studies 2 and 3.
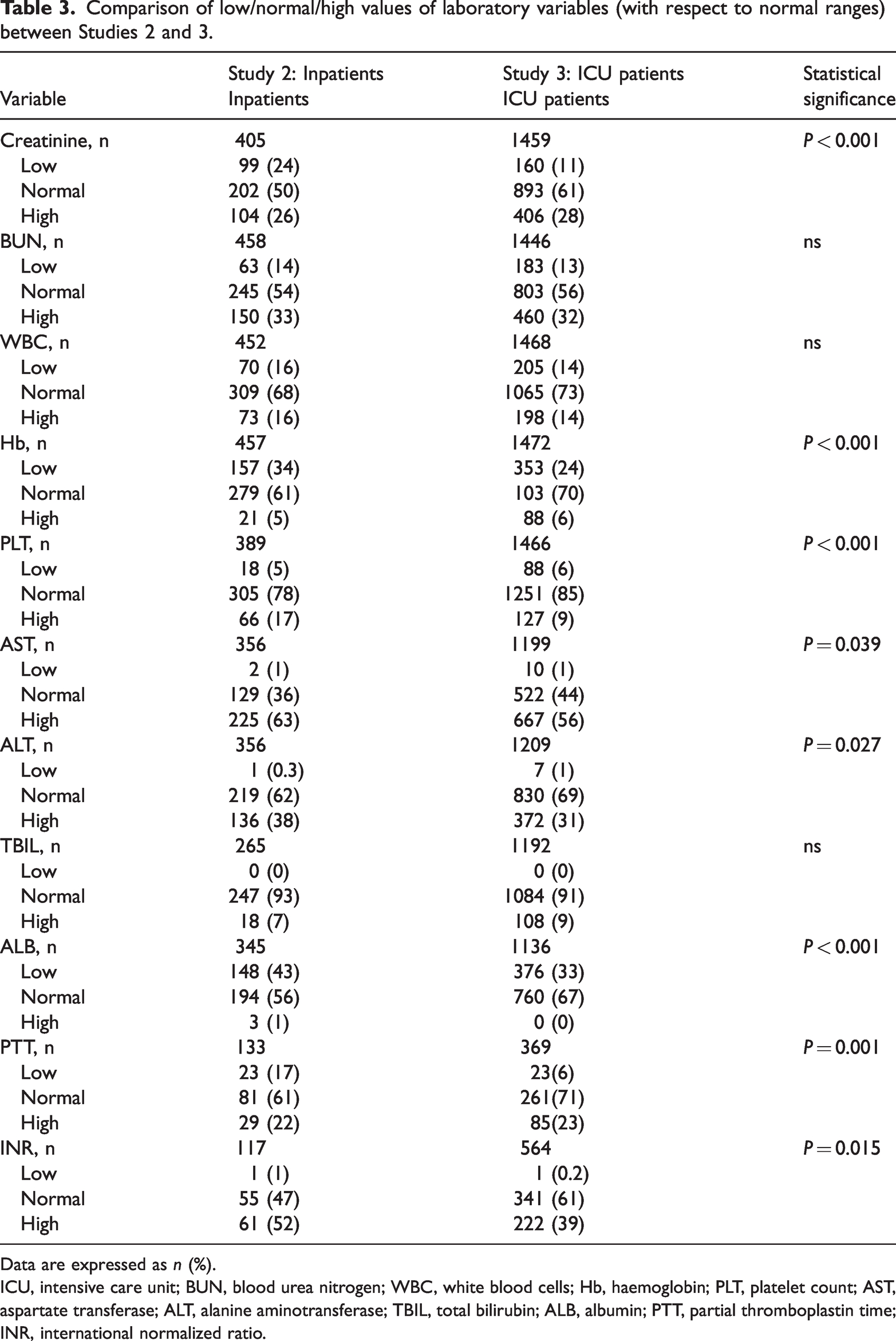
Data are expressed as n (%).
ICU, intensive care unit; BUN, blood urea nitrogen; WBC, white blood cells; Hb, haemoglobin; PLT, platelet count; AST, aspartate transferase; ALT, alanine aminotransferase; TBIL, total bilirubin; ALB, albumin; PTT, partial thromboplastin time; INR, international normalized ratio.
For creatinine, which was measured in all studies, the proportion of patients with a high creatinine level was the lowest in the outpatient group, compared with the other two groups (Figure 3). The proportion of patients with a low creatinine level was the highest in the inpatient group, compared with the other two groups.

(a and b) Comparison of creatinine (Cr) levels (mean ± SD) and proportion of patients with low, normal or high values relative to the normal range, from the three studies.
The proportion of patients with high AST levels was higher in the inpatient group (63%) compared with the ICU group (56%). The proportion of patients with low ALB levels was higher in the inpatient group (43%) compared with the ICU group (33%).
Prognostic factors
Of the 478 patients in the inpatient group (Study 2), 52 had a clinical endpoint (death/ECMO), while the others (n = 426) were categorized as the survival group (Figure 1). We compared laboratory variables and demographic data between these two groups (Table 4). Significant prognostic factors for outcome of inpatients with COVID-19 included: age; creatinine; BUN; AST; ALB.
Prognostic factors for outcome of COVID-19 disease in Study 2 (Inpatients).
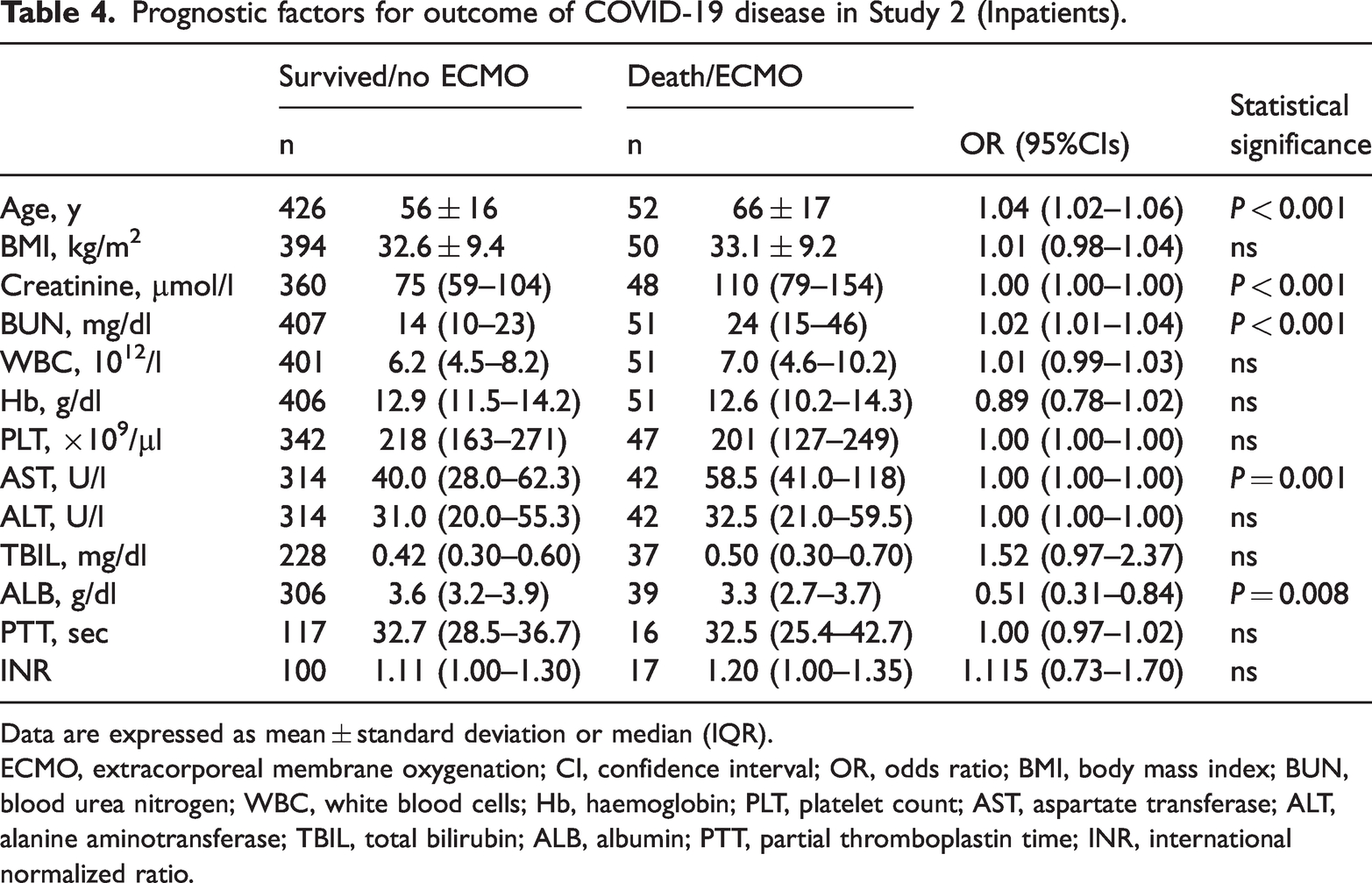
Data are expressed as mean ± standard deviation or median (IQR).
ECMO, extracorporeal membrane oxygenation; CI, confidence interval; OR, odds ratio; BMI, body mass index; BUN, blood urea nitrogen; WBC, white blood cells; Hb, haemoglobin; PLT, platelet count; AST, aspartate transferase; ALT, alanine aminotransferase; TBIL, total bilirubin; ALB, albumin; PTT, partial thromboplastin time; INR, international normalized ratio.
Discussion
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) caused significant morbidity and mortality worldwide.16,17 Laboratory variables 18 and demographic data 19 are often used as the bases for selecting treatment options. Indeed, WBC, ALB, and Hb levels are used to inform decisions regarding COVID-19 treatment.10,20 In this re-analysis of data from three studies in the BioLINCC public database, we identified some laboratory variables that could be used to represent the characteristics of inpatients and ICU patients. We also identified possible prognostic factors for the outcome of inpatients with COVID-19.
Creatinine is a product of muscle metabolism in the human body, and mainly excreted from the body through glomerular filtration. Following a decrease in protein intake or exercise volume, muscle metabolism decreases, which can cause a fall in creatinine levels. Low creatinine levels indicate malnutrition, whereas high levels indicate poor renal function. Our research found that the proportion of patients with high creatinine values in the inpatient group was greater than in the outpatient group, and the proportion of patients with low creatinine levels in the inpatient group was greater than that in the ICU patient group. These findings suggest that disease severity in inpatients is more severe than that of outpatients but less than that of ICU patients. In hospitalized patients, creatine levels are an important prognostic factor since high levels indicate acute kidney injury, which is a major risk factor for death and ECMO use.
Our study identified several common laboratory variables that could be used to assess disease severity or predict outcome, some of which have been previously reported.6,8,21,22 We found three laboratory variables (i.e., creatinine, AST, and ALB) were not only prognostic factors for outcome of inpatients with COVID-19, but also reflected disease severity as they were significantly different between inpatients and ICU patients. These three laboratory variables are an indication of kidney function, liver function, and nutritional status. We suggest that these variables should be carefully monitored in patients with COVID-19, especially those in hospital.
Our study had several limitations. For example, it was re-analysis of data from previously published research. Accordingly, the selected participants and laboratory variables were restricted to those from the original dataset. Therefore, other variables such as, estimated glomerular filtration rate (eGFR) were not considered; eGRF is more reliable in predicting renal function than creatinine levels since creatinine can be affected by many factors such as muscle mass, age, sex, and ethnicity. 23 However, this is the first study to extract data from several COVID-19 studies contained within the BioLINCC database, and our findings will enrich the evidence base on COVID-19 research. Perhaps the findings from our study will inspire further prospective studies to verify our results.
In summary, for patients with COVID-19, in addition to monitoring infectious disease indicators, we need to pay attention to liver function, renal function, and take timely measures to correct them to improve prognosis. Creatinine, AST, and albumin levels not only relate to severity but also prognosis of COVID-19 disease.
Footnotes
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This work was supported by the Department of Science and Technology of Zhejiang Province (No. 2023C25012) and the Health Commission of Zhejiang Province (No. 2021KY606), the People's Republic of China.
