Abstract
Objective
Follicular lymphoma (FL) is an indolent, lymphoproliferative disease of B-cell origin that has a heterogeneous disease course with varying outcomes. Certain patients may undergo autologous stem cell transplantation. We investigated the outcome of autologous stem cell transplantation in patients with FL.
Methods
Patients who received autologous stem cell transplantation at the University of Debrecen’s Department of Hematology between 2004 and 2021 were retrospectively analyzed. The overall survival (OS) and progression-free survival (PFS) after transplantation of patients with FL were examined. Prognostic factors that may influence the course of the disease were chosen.
Results
Data were collected from 49 patients. OS was influenced only by age, whereas PFS was affected by age and the lymphocyte/monocyte ratio. The combination of age and lymphocyte/monocyte ratio defined a patient population with a particularly unfavorable prognostic risk profile: patients over 47 years of age with a pre-transplant lymphocyte/monocyte ratio greater than or equal to 2.675.
Conclusion
Age and lymphocyte/monocyte ratio were identified as useful prognostic factors for PFS in patients with FL following autologous stem cell transplantation.
Keywords
Introduction
Follicular lymphoma (FL) is the most common type of indolent lymphoma. The management of FL continues to be characterized by relapsing and remitting courses and a risk of high-grade transformation. Overall survival (OS) in FL has increased for the past two decades with the improvement of novel therapies, and the expected median OS may exceed 20 years. 1 The role of autologous stem cell transplantation (ASCT) in FL and the effectiveness of new targeted agents and allogeneic stem cell transplantation (alloSCT) still need further clarification.
Although rituximab-containing chemotherapy regimens such as R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, prednisolone), R-CVP (rituximab, cyclophosphamide, vincristine, prednisolone), and rituximab-bendamustin have solidified the role of rituximab as first-line therapy, the probability of relapse remains high in FL, with 33% of patients relapsing within 3 years of therapy. 2 However, even the OS in patients with relapsed FL who have risk factors or those who have had early treatment failure has been improving. Several trials have discussed the importance of ASCT in relapsing FL; large prospective trials have shown that the use of ASCT is no longer supported in first remission because of the absence of an OS benefit despite improvement in progression-free survival (PFS).2–5
High-dose chemotherapy (HDT) followed by ASCT in second remission appears to be the best strategy for timing ASCT. The advantage is greater than total-body irradiation-free conditioning (e.g., rituximab plus bis-chloroethylnitrosourea, carmustine [BCNU]) in younger patients with chemosensitive disease because the use of total-body irradiation conditioning appears to increase the risk of secondary malignancies such as acute myeloid leukemia/myelodysplastic syndrome. 2 Other options for managing the second remission are rituximab+ lenalidomide, rituximab or obinutuzumab with a different backbone, or rituximab/obinutuzumab maintenance. Lenalidomide demonstrated synergy with CD-20 antibodies. Rituximab+ lenalidomide is now considered standard therapy after relapse following anti-CD-20 or chemoimmunotherapy. 1
ASCT may be considered in patients who are in good condition or asymptomatic or patients under the age of 70 years in combination with high-dose chemotherapy, and long-term survival with therapy is expected. However, the outcome of these patients after transplantation and the subset of patients who are affected by the procedure remains unclear. 6
Patients and methods
We retrospectively investigated the prognostic factors of patients with FL who underwent ASCT between 2004 and 2021 at the University of Debrecen’s Department of Hematology. This retrospective analysis was approved by the Regional and Institutional Research Ethics Committee of the University of Debrecen (DE RKEB/IKEB 5694-2021). The patients provided prior written or verbal consent for the collection of their data. We de-identified all of the patient details. The reporting of this study conforms to the standards for reporting qualitative research. 7 We investigated factors that may have influenced post-transplant PFS and OS. We collected factors such as age, grade, stage, time elapsed until progression, transplant in first chemosensitive relapse, type of conditioning, status of disease before transplant (complete metabolic remission, complete remission, or partial remission), and maximum standardized uptake value (SUVmax) before transplant. Furthermore, the follicular lymphoma international prognostic index (FLIPI), bone marrow involvement, lymphocyte count, monocyte count, lymphocyte/monocyte (Ly/Mo) ratio, and hemoglobin, lactate dehydrogenase, and beta 2-microglobulin levels before transplant.
The patients were treated in accordance with the institutional guidelines and as previously described. 8 Treatment was initiated for patients who met the Groupe d’Etude des Lymphomes Folliculaires criteria. 9 Grades 1 and 2 patients received R-CVP until 2015 when bendamustine became widely available. Grade 3 patients received R-CHOP chemotherapy. Rituximab maintenance was administered to patients as described in the PRIMA trial 10 since 2013 when the drug became available. The patients with relapsed FL received dexamethasone, cytarabine, and cisplatin (DHAP) with either rituximab or obinutuzamab. Stem cell mobilization was performed while the patients received a second or third cycle of anti-CD20-DHAP. Given that we are a tertiary center, we were referred to patients with relapsed/refractory FL when stem cell collection was due. Patients who achieved a complete or at least partial response underwent ASCT. Before treatment initiation, all of the patients provided informed consent to have their data retrospectively collected and published in accordance with the Declaration of Helsinki.
Positron emission tomography/computed tomography (PET/CT) has been routinely used as an imaging modality at the University of Debrecen since May 2009. A work-up PET/CT was performed for every patient before transplantation since 2009, whereas a conventional computed tomography was conducted before 2009. The SUVmax measurements were performed using the Interview Fusion version 3.03.077.0007 software (Mediso, Budapest, Hungary), which has been cross-validated with the Philips ISP system. The SUVmax calculation was based on body weight, and the SUV dimensions were in g/mL. A nuclear medicine specialist measured the lesions on the fused images, and we placed the globus volume of interest onto the areas with the highest intensive uptake.
To exclude histological transformation, histology was performed from the most accessible site at the time of diagnosis and in the event of relapse or progression. The histology slides were not centrally reviewed; however, the results were verified by two expert hematopathologists.
The patient outcomes were analyzed for PFS and OS. PFS was calculated from the date of transplant to June 2021, relapse or progression of disease, histological transformation, or death, whereas OS was calculated from transplantation to June 2021 or death. Factors that could affect survival were evaluated using a multivariate analysis. Continuous variables were transformed into discrete variables either by exceeding normal values or calculating data cut-off values based on the receiver operating characteristic (ROC) curves. State variables were defined as the events defined for PFS. A multivariable Cox regression model with the Enter method was used to obtain the hazard ratio. Survival was estimated using the Kaplan–Meier method. The comparison of survival curves was based on the log-rank test. Statistical significance was set at p < 0.05.
Results
Forty-nine patients with FL and a median age of 48 years underwent ASCT at the University of Debrecen’s Department of Hematology between 2004 and June 2021. Table 1 includes patient characteristics. Most patients were diagnosed with histology grade 2 or 3a. The median Eastern Cooperative Oncology Group score was 0 and the median Karnofsky score was 100%. The vast majority of patients (46) received cyclophosphamide-containing first-line therapy (i.e., R-CVP or R-CHOP), and three patients received rituximab-bendamustin. Sixteen patients received rituximab maintenance. Thirty-eight patients showed disease progression within 24 months (POD24). Seventy-five percent of patients (37) underwent transplantation after the first chemosensitive relapse, whereas the remaining patients were transplanted in further lines. All of the patients received anti-CD20-BEAM (BCNU, etoposide, cytarabine, melphalan) as a conditioning regimen, except for one patient who was allergic to rituximab. More than half of the patients were in complete (i.e., metabolic) remission, most patients did not have bone marrow involvement, and most patients had a high FLIPI at the time of transplantation. The median length of hospitalization was 23 days (range: 17–40 days).
Patient characteristics.

Cy, cyclophosphamide; B, bendamustine; R, rituximab, POD24, progression of disease within 24 months; Tx, transplantation; Z-BEAM, zevalin, bis-chloroethylnitrosourea, carmustine, etoposide, cytarabine, melphalan; R-BEAM, rituximab, bis-chloroethylnitrosourea, carmustine, etoposide, cytarabine, melphalan; G-BEAM, obinutuzumab, bis-chloroethylnitrosourea, carmustine, etoposide, cytarabine, melphalan; CMR, complete metabolic remission; CR, complete remission; PR, partial remission; FLIPI, follicular lymphoma international prognostic index; ND, not determined.
With a median follow-up of 37 months (range: 1–196 months), the median PFS was 48 months post-transplant, whereas the median OS was not met (Figure 1). No difference in survival was observed when the period was divided into the intervals 2004 to 2010 and 2011 to 2021. Nine patients who reached the plateau phase of the curve and had a PFS of >98 months were considered long-time survivors and “functionally cured.” Given the retrospective nature of this evaluation, we did not have a control group of patients who did not undergo ASCT. Those who did not undergo transplantation in this period had significantly inferior clinical features; hence, the disease courses were biased and not comparable.
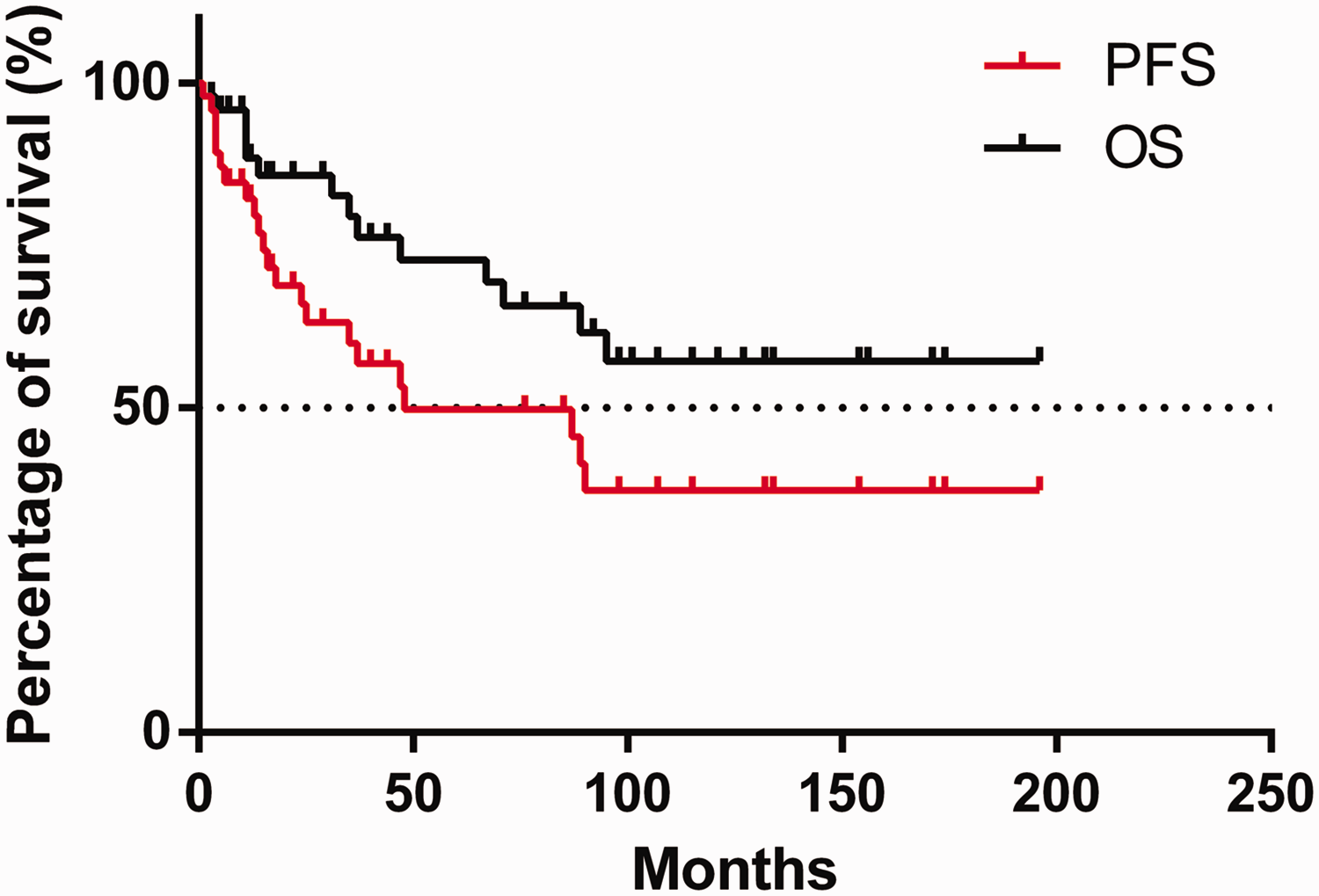
Progression-free survival and overall survival of follicular lymphoma patients post-transplant.
A multivariate analysis model indicated that age and the Ly/Mo ratio affected PFS, whereas only age affected OS. Both variables were independent prognostic factors for post-transplant relapse. A multivariate analysis further showed that a transplant in the first chemosensitive relapse was not prognostic for either PFS or OS (Table 2).
Multivariate analysis model for factors that may affect progression-free survival and overall survival in patients with follicular lymphoma post-transplant.

PFS, progression-free survival; OS, overall survival; CI, confidence interval; POD24, progression of disease within 24 months; Tx, transplantation; CR, complete remission; PR, partial remission; FLIPI, follicular lymphoma international prognostic index; Ly, lymphocyte; Mo, monocyte, Hgb, hemoglobin, LDH, lactate dehydrogenase.
The ROC curve showed a Ly/Mo ratio cut-off of 2.675 and age greater than 47 years as the best predictors of PFS; however, the difference was not significant (Figures 2 and 3). Nevertheless, when the two variables were combined, a significant difference (p = 0.0200) was observed in the PFS of the four subgroups (Figure 4). Importantly, the group of patients with FL who were older than 47 years of age and had a Ly/Mo ratio ≥2.675 had a poor prognosis; the difference in PFS between this group and the remaining patients was significant (p = 0.0019; Figure 5). The difference in survival was not translated into a significant difference in OS.

The receiver operating characteristic (ROC) curve showed a cut-off of 2.675 of the lymphocyte/monocyte ratio for best predicting follicular lymphoma patients’ progression-free survival, however, the difference was not significant.

The receiver operating characteristic (ROC) curve showed a cut-off of 47 years of age for best predicting follicular lymphoma patients’ progression-free survival, however, the difference was not significant.

When combining the cut-off of lymphocyte/monocyte ratio of 2.675 and an age of 47 years, a significant difference (p = 0.0200) was seen, illustrating progression-free survival of the 4 subgroups.

When we highlight one poor prognostic group of follicular lymphoma patients older than 47 years and having a Ly/Mo ratio >=2.675. The progression-free survival difference is significant (p = 0.0019).
No difference in disease status before transplant was observed between patients in complete metabolic remission and those in partial remission.
The patients who underwent autologous stem cell transplantation after POD24 of initiating first-line treatment had significantly worse PFS post-transplant (Figure 6). The difference did not translate into an OS disadvantage.
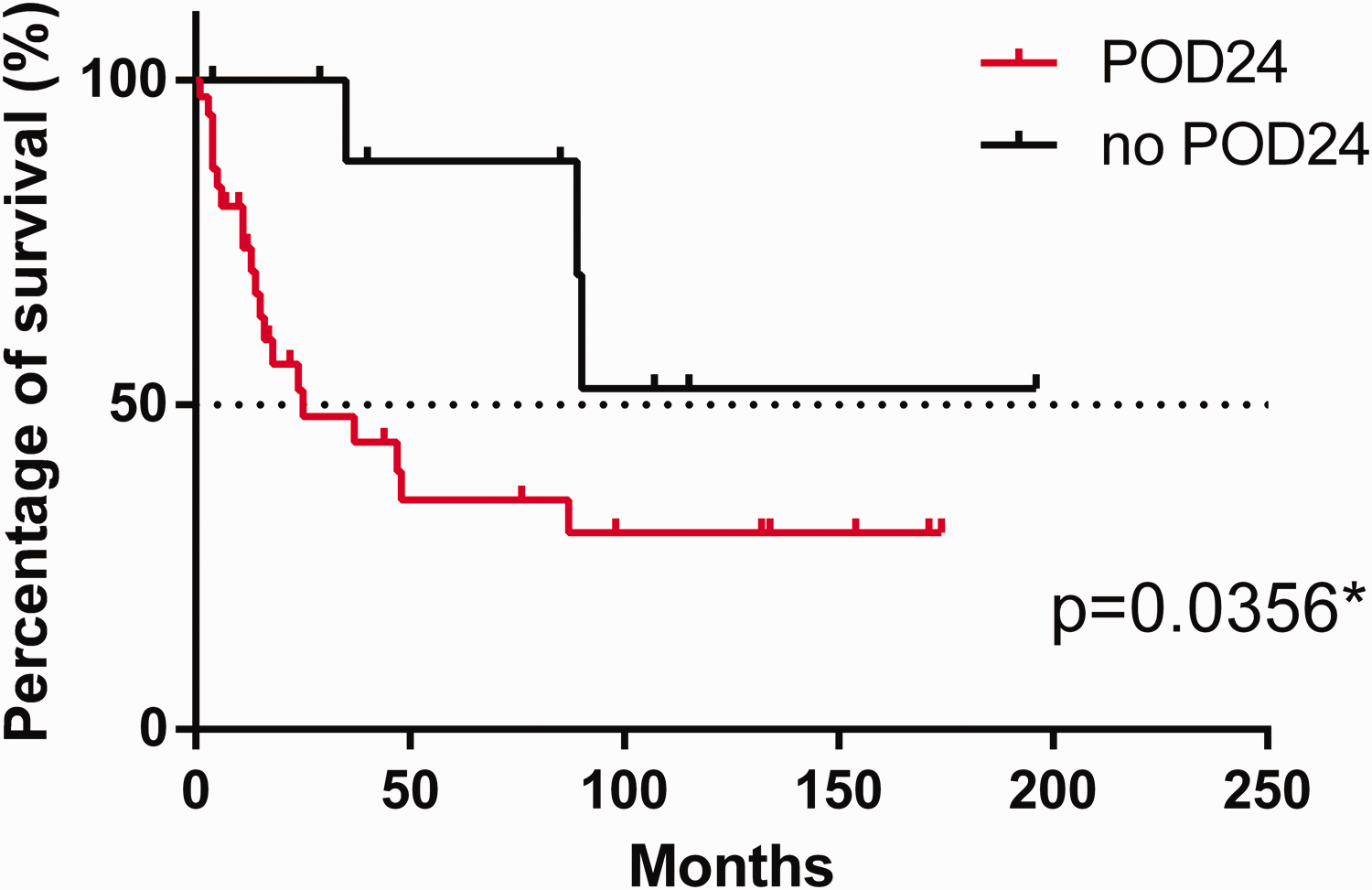
Follicular lymphoma patients who underwent autologous stem cell transplant after progressing within 24 months (POD24) of initiating first-line treatment had a significantly worse progression-free survival post-transplant.
Discussion
In addition to the use of conventional prognostic scores (FLIPI-1,-2, primary rituximab and maintenance prognostic index, m7-follicular lymphoma prognostic index, follicular lymphoma evaluation index), 11 the methods of characterizing first-line treatment of FL appear to include EOT (restaging PET/CT) and POD24. After histological transformation is ruled out, patients who are characterized with these methods can be consolidated with high-dose chemotherapy and ASCT after successful salvage therapy. Studies have demonstrated an enduring response after autologous transplants.12–14 Young, fit patients who experience a high tumor burden and early relapse should also be consolidated with ASCT, 1 which is considered a potential curative treatment option. 15 Early ASCT performed within a year of early treatment failure improved OS compared with conventional chemotherapy. 16 The use of ASCT was independently associated with improved OS in patients with POD24 who received rituximab-chemo. According to the literature, OS improved after ASCT in patients with early relapse. 6 However, our dataset did not support the same conclusion: the inferior post-transplant PFS in our patients with POD24 did not translate into inferior OS. Patients were successfully salvaged in the event of post-transplant relapse.
It should be noted that, according to the literature, OS improvement in late relapse has not been observed in patients who underwent ASCT compared with those who were not treated with ASCT. 17 ASCT in the first or second progression was an effective therapy independent of previous rituximab exposure. Those who maintain disease response for a specified period may have an outcome that is similar to that of the general population. 17 Our patient cohort reached a plateau at 98 months (i.e., slightly more than 8 years).
The Spanish GELTAMO group reported the long-term outcomes and prognostics of patients with FL who were transplanted a longer time ago (1999–2007). 18 The group revealed a median PFS of 9.7 years and a median OS of 21.3 years post-transplant. The group also reported the prognostic significance of POD24 for OS in the post-ASCT setting. Early progression within 24 months post-transplantation resulted in significantly worse OS. Sex, disease status (complete or partial remission), prior rituximab exposure, and time from diagnosis to ASCT affected PFS, whereas age, sex, Eastern Cooperative Oncology Group status, lactate dehydrogenase, disease status, prior rituximab use, stem cell source (bone marrow vs. peripheral blood), and time from diagnosis to ASCT affected OS. We observed that age and Ly/Mo ratio affected PFS, whereas only age affected OS. No significant survival difference was detected between the periods 2004 to 2010 and 2011 to 2022 because the principals of ASCT did not change within this time window.
The 12-year follow-up of the prospective randomized European Society for Blood and Marrow Transplantation LYM1 trial reported a 10-year PFS of 47% after ASCT, whereas the 10-year OS was 75%. 19 The study reported a 10-year OS of 60% in patients with previous POD24 compared with 85% in patients with no POD24. However, clinically and statistically significant OS differences were observed 24 months post-transplantation.
Our results were similar to those of a German workgroup that investigated a comparable number of post-transplant patients; specifically, the pre-transplant presence of POD24 did not impact PFS or OS, and other outcomes were also similar. 4 Our findings were also similar to those of a small Canadian study that examined post-transplant outcomes in patients with FL and in which the 5-year PFS and 5-year OS were 53.6% and 71.8%, respectively. 20
Pre-transplant factors have not been established for predicting outcomes. A molecular-based model did not identify patient subsets in which a significant OS benefit from frontline ASCT was observed. 21
A French study retrospectively investigated 414 patients with FL who underwent autologous stem cell treatment in four centers. Only 23% of patients relapsed following ASCT after a median follow-up of 8.8 years. The researchers observed that anemia, stage at relapse, and number of treatment lines before ASCT were prognostic for PFS, whereas relapse after 2 years post ASCT, histological transformation, beta 2-microglobulin, and high-risk FLIPI at relapse were additional prognostic factors for OS. Importantly, in contrast to our findings or those of other studies, the authors did not observe long-term disease control or a potential plateau. 22
Our results were very similar to those of a single-center Polish study. 23 In 30 transplanted patients with FL, the 10-year PFS was 33% and OS was 60%. The median OS was not met, and the median PFS was 4.8 years. The authors did not observe any effect of the FLIPI or disease status before transplant on PFS or OS.
Consistent with previous studies and in contrast to our results, the pre-transplant PET/CT result was prognostic of 59 patients with FL who were transplanted in Lymphoma Study Association centers in France between 2007 and 2013 and who relapsed after R-CHOP but were chemosensitive to salvage therapy. 24
ASCT has proven to be a more reliable and cost-effective treatment for patients with relapsed FL. With lower transplant-related mortality and similar OS compared with allo-SCT, ASCT is preferred as an initial transplant strategy. Allo-SCT is the best rescue strategy for those who fail to have ASCT or who relapse after ASCT. 25
The retrospective nature of our study and its small number of patients are limitations. However, we maintain that our experience with 49 patients who underwent transplants in a single institute may have significant value, especially considering that similar studies were equally small.
Potential treatment options for patients who relapse after ASCT are novel agents such as idelalisib (a phosphoinositide 3-kinase inhibitor), obinituzumab (cluster of differentiate [CD] 20 antibody), lenalidomide (an immunomodulatory drug), venetoclax (a B-cell lymphoma-2 inhibitor), ibrutinib (a bruton tyrosine kinase inhibitor), tazemetostat (an enhancer of zeste homolog 2 inhibitor that has also shown activity in EZH wild type FL), tafasitamab (an anti-CD 19 antibody), bispecific antibodies, and chimeric antigen receptor T cells.1,2,26 Polatuzumab vedotin (an anti-CD79 b antibody conjugated to mono-methyl auristatin), inotuzumab ozogamicin (an anti-CD 22 antibody conjugated to calicheamicin), and pidilizumab (an anti-programmed cell death protein 1 antibody) may also be options for patients who relapse after transplantation or in maintenance or bridging therapies. 3
Footnotes
Acknowledgement
The authors thank Katalin Hódosi for statistical support.
Author contributions
AK collected and analyzed the data and wrote the manuscript; EK collected and analyzed the data; ZS analyzed the data and critically analyzed and accepted the final manuscript; ÁI analyzed the data and approved the final manuscript; ÁJ analyzed the data, wrote the manuscript, and accepted the final manuscript. All authors read and approved the manuscript.
Data availability
The raw data are publicly available upon request. The corresponding author Dr. Ádám Jóna can be contacted at
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
