Abstract
CBP/p300 interacting transactivator with Glu/Asp-rich carboxy-terminal domain 1 (CITED1) is a transcriptional activator belonging to the non-DNA-binding transcription co-regulator family. It regulates diverse pathways, including the transforming growth factor/bone morphogenetic protein/SMAD, estrogen, Wnt-β-catenin, and androgen-AR signaling pathways, by binding to CBP/p300 co-activators through its conserved transactivation domain CR2. CITED1 plays an important role in embryonic development and a certain regulatory role in the occurrence and development of various tumors. In this article, the biological characteristics, expression regulation, participating signaling pathways, and potential roles of CITED1 in the clinical diagnosis and treatment of tumors are reviewed.
Keywords
Introduction
CBP/p300 interacting transactivator with Glu/Asp-rich carboxy-terminal domain 1 (CITED1) was originally named melanocyte-specific gene 1 (MSG1) because it was believed to be expressed only in melanocytes. 1 Subsequently, the extra-melanocytic expression of MSG1 in mammary epithelium, testes, the brain, embryonic tissues, and some tumors was detected, and its biological characteristics were elucidated.1–5 Therefore, “MSG1” was renamed “CITED1.”
Biological characteristics of CITED1
The CITED1 gene, located on chromosome Xq13.1, is highly conserved with three exons and two introns, and its 5ʹ end contains a TATA box and potential binding sites for multiple transcription factors (USF, Brn-3, Brn-2, TFE3, Oct-1, AP-2, and Sp1). 6 The promoter fragment activates transcription of the CITED1 gene, which encodes a 27-kDa nuclear protein. CITED1 shares a highly conserved transcriptional activation region (CR2) with other members of the CITED family, including CITED2 (MRG1 or p35srj), CITED3, and CITED4.7,8 CR2 (amino acids 145–193), a C-terminal acidic domain containing six aspartic acid and six glutamic acid residues, enhances the transcriptional activation activity of CITED1 by binding to the co-activators CBP/p300.1,7 Heat shock cognate protein 70, hypoxia-inducible factor-1, β-catenin, and microphthalmia-associated transcription factor (MITF) compete with CITED1 for binding to CBP/p300, thereby repressing related transcription.8–11 Moreover, phosphorylation of five serine residues of CITED1 in M-phase (Ser16, Ser63, Ser67, Ser71, and Ser137) also interferes with the binding of CITED1 to CBP/p300, thus reducing the ability of CITED1 to activate transcription. 12
Because CITED1 lacks DNA-binding activity, it serves as a mediator to stabilize and augment the interaction between CBP/p300 and certain DNA-binding proteins, and then they translocate to the nucleus as complexes, where they bind to the promoters of target genes and enhance corresponding transcription. Among these binding partners, CITED1 binds to the transcription factor SMAD4 through its N-terminal SMAD4-interacting domain (amino acids 30–60), enhancing transforming growth factor β (TGF-β)/bone morphogenetic protein (BMP)-induced transcription depending on SMAD hetero-oligomerization.8,13,14 CITED1 binds to estrogen receptor α (ERα) through its N-terminal CR2 region (amino acids 157–158), and this binding is independent of the interaction region with CBP/p300, permitting the coactivation of estrogen-dependent transcription (Figure 1). 15 CITED1 additionally binds to TOX high mobility group box family member 3 (TOX3) through CR2, thereby significantly enhancing estrogen response element-dependent transcription and playing an anti-apoptotic role. 16 The subcellular localization of CITED1 is characterized by cellular heterogeneity. In HEK and MCF-7 cells, the centralized distribution of CITED1 in the cytoplasm was confirmed to be associated with chromosomal region maintenance 1 (CRM1)-dependent nuclear export signals (108LMSLVVELGL117) on CR2, whereas in osteoblasts, the parathyroid hormone-induced nuclear translocation of CITED1 was confirmed to be associated with phosphorylation at Ser79 and activation of protein kinase C (PKC).12,17

The biological characteristics of CITED1. (1) CR2 enhances the transcriptional activity of CITED1 by binding to the co-activators CBP/p300. (2) Because CITED1 lacks DNA-binding activity, it serves as a mediator to stabilize and augment the interaction between CBP/p300 and some DNA-binding proteins. CITED1, CBP/p300 interacting transactivator with Glu/Asp-rich carboxy-terminal domain 1.
CITED1 is involved in the process of embryonic development, including the development of the metanephric mesenchyme, ureter, placenta, and brain.4,5,14 The loss of CITED1 often leads to abnormal embryonic development. 18 In addition, CITED1 plays a certain regulatory role in the occurrence and development of various tumors. To provide a valuable theoretical basis for further study of CITED1, this article reviews the research progress on CITED1 in these tumors and its potential function in clinical tumor diagnosis and treatment.
Expression regulation of CITED1
Currently, there are few studies on the regulatory mechanism of CITED1 expression. Existing studies suggest that certain regulatory factors can regulate the expression of CITED1 at the transcriptional or post-transcriptional level. In melanocytes and melanoma, the PKC activators 12-O-tetradecanoyl phorbol-13-acetate/endothelin-1 (ET-1) and receptor tyrosine kinase activator fibroblast growth factor-2 (FGF-2) regulate the expression of CITED1 at the transcriptional and post-transcriptional levels, respectively. 19 Studies suggest that under UV-B irradiation, epidermal keratinocytes can secrete ET-1 and FGF-2, which act on melanocytes in a paracrine manner, leading to increased CITED1 expression. 19
BRAF activating mutations are also involved in the regulation of CITED1 expression, but they play different roles in different tumor types. BRAF activating mutations are common in melanoma and papillary thyroid carcinoma (PTC). Studies revealed that in melanoma, BRAF activating mutations downregulate CITED1 expression by enhancing the expression and function of the transcription factor nuclear receptor subfamily 4 group A member 1/2. 10 However, in PTC, the BRAF V600E mutation induces high expression of CITED1 mRNA via hypomethylation of CpGs in the CITED1 promoter (especially CpG18 and CpG24). 20
In addition, miR-26, the Runx1/CBFβ complex, and c-Myc were found to be involved in the regulation of CITED1 expression in tumors, but the specific regulatory mechanisms remain to be further explored.21–23
The main signaling pathways and role of CITED1 in tumors
TGF-β/BMP/SMAD signaling pathway
TGF-β plays a dual role in tumorigenesis and development. When tumor cells successfully evade the growth-inhibiting effect of TGF-β, TGF-β–mediated oncogenic functions, such as the induction of epithelial-to-mesenchymal transition (EMT), can be used to promote tumor cell survival, proliferation, invasion, and metastasis. In the process of tumor cell growth and metastasis, various cells in the tumor microenvironment often release a large amount of TGF-β, which directly promotes the malignant transformation of tumor cells. Studies illustrated that CITED1 is highly expressed in the invasive front of melanoma. CITED1 enhances the transcription of the TGF-β/SMAD signaling pathway-related target genes JAK1, leukemia inhibitory factor, interleukin 11 (IL11), myosin phosphatase rho interacting protein, and rho guanine nucleotide exchange factor 5; promotes cooperation between Rho/ROCK and JAK1/STAT3 signaling; and provides the high levels of actomyosin contractility for melanoma cells to migrate in the rounded ‘‘amoeboid’’ mode (Figure 2).24,25 In colorectal cancer (CRC), CITED1, through an association with TGF-β/SMAD signaling, was found to be one of the genes leading to the dedifferentiation of CRC invasive front cells, and the metastatic ability of CRC invasive front cells without the epithelial phenotype was enhanced. 26 In addition, CITED1 overexpression induces the differentiation of embryonic stem cells into trophoblast cells by activating the BMP signaling pathway, leading to teratoma formation (Figure 3). 27

CITED1 promotes amoeboid melanoma migration. CITED1 enhances the transcription of TGF-β/SMAD signaling pathway target genes (JAK1, LIF, IL11, M-RIP, and ARHGEF5), promotes the cooperation between Rho/ROCK and JAK1/STAT3 signaling, and provides high levels of actomyosin contractility for melanoma cells to migrate in the rounded ‘‘amoeboid’’ mode. CITED1, CBP/p300 interacting transactivator with Glu/Asp-rich carboxy-terminal domain 1; LIF, leukemia inhibitory factor; IL11, interleukin 11; M-RIP, myosin phosphatase rho interacting protein; ARHGEF5, rho guanine nucleotide exchange factor 5.

CITED1 induces teratoma formation. CITED1 overexpression induces the differentiation of ESCs into trophoblast cells by activating BMP signaling pathway, leading to the formation of teratoma. CITED1, CBP/p300 interacting transactivator with Glu/Asp-rich carboxy-terminal domain 1; ESCs, embryonic stem cells; BMP, bone morphogenetic protein.
Estrogen signaling pathway
CITED1 activates the transcription of amphiregulin (AREG) via the estrogen signaling pathway during adolescent breast development, thus promoting the growth of mammary epithelium and the formation of mammary ducts.2,28 In addition, AREG has been identified as a downstream effector of estrogen in ER+ breast cancer, and its expression is necessary for the growth of MCF-7 xenografts. 29 CITED1 can also coactivate the estrogen-dependent ER-mediated transcription of TGF-α, promoting the growth of MCF-7 cells in an autocrine manner. The effect is more apparent when the estrogen concentration is low because CITED1 enhances the sensitivity of MCF-7 cells to estrogen. 15 However, the latest literature confirms that MCF-7 cells overexpressing CITED1 exhibit selective amplification of AREG but not TGF-α, and the maintenance of specific ERα/CITED1-mediated transcription is a good prognostic marker in patients with anti-endocrine–treated ER+/LN- breast cancer.28,30 Therefore, the role of CITED1 in regulating TGF-α transcription in MCF-7 cells remains controversial and further exploration is needed. In addition, the EGR2/CITED1 transcription factor complex can bind to the ErbB2 promoter and increase the expression of human epithelial growth factor receptor 2 (HER2). 31 HER2 overexpression is related to the occurrence of breast cancer and poor prognosis. Therefore, the mechanism and clinical role of CITED1 might depend on the molecular subtype of breast cancer (Figure 4).
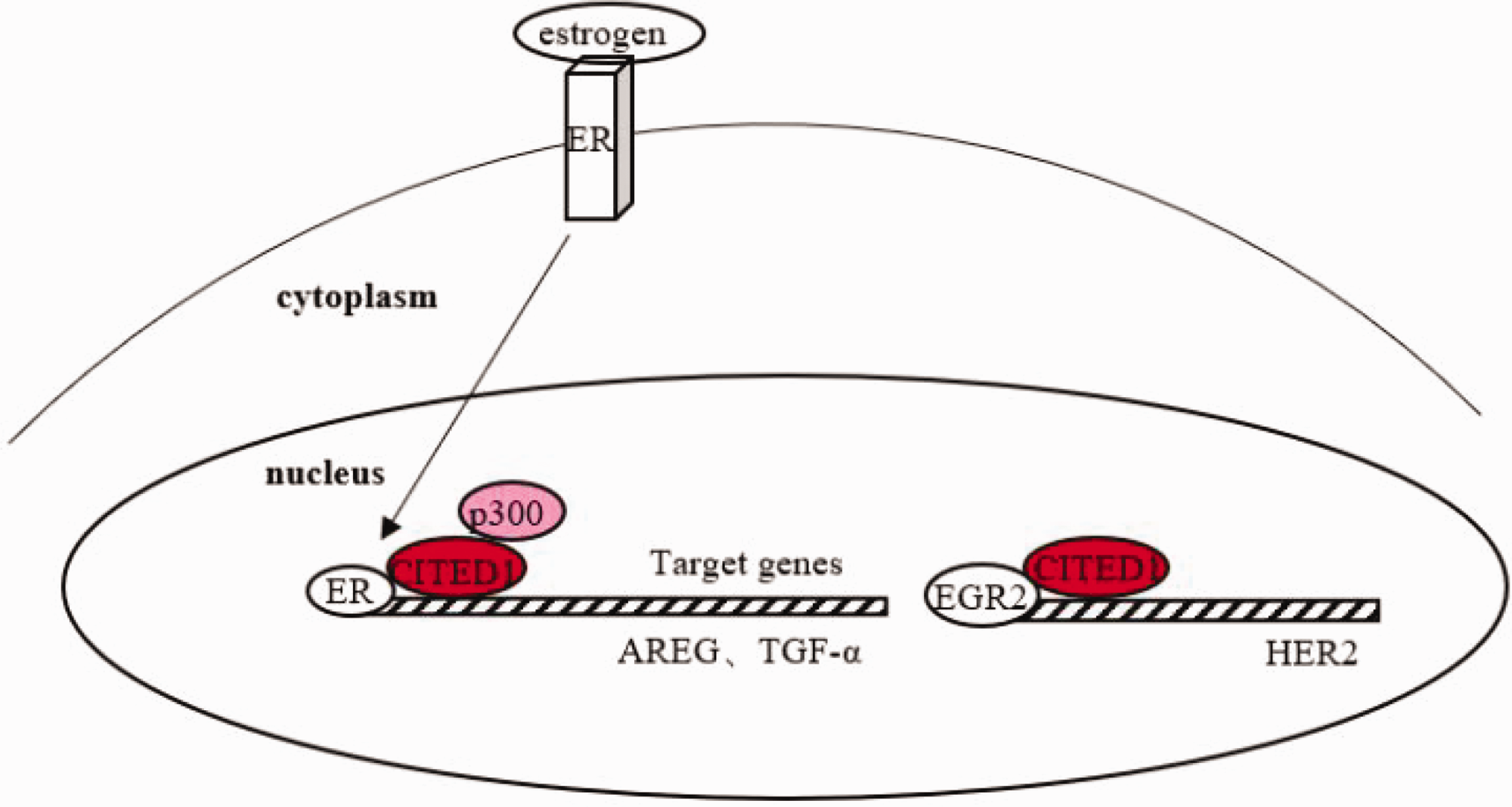
The role of CITED1 in breast cancer. (1) CITED1 can selectively coactivate the estrogen-dependent ER-mediated transcription of AREG and TGF-α. (2) The EGR2/CITED1 transcription factor complex can bind to the ErbB2 promoter and enhance the expression of HER2. CITED1, CBP/p300 interacting transactivator with Glu/Asp-rich carboxy-terminal domain 1; ER, estrogen receptor, AREG, amphiregulin; TGF-α, transforming growth factor α.
Wnt/β-catenin signaling pathway
The Wnt/β-catenin signaling pathway is involved in maintaining the balance between proliferation and differentiation in a variety of tumors, and this role is related to the transcription coactivator CBP/p300.32–34 Specifically, the binding of CBP to β-catenin activates transcription of cell proliferation-related genes, whereas the binding of p300 to β-catenin mediates transcription of cell differentiation-related genes. 35 CBP/P300 is the main protein interacting with CITED1. Thus, CITED1 might act as a coregulator controlling the balance between CBP/β-catenin–mediated transcription (maintaining stem/progenitor cell proliferation) and p300/β-catenin-mediated transcription (initiating cell differentiation).
During vertebrate development, CITED1 is highly expressed in the progenitor cells of embryonic tissues, but its expression is downregulated and ultimately lost when they differentiate into mature tissues. Because of the persistent presence of undifferentiated components, embryonic tumors usually have high CITED1 expression. 36 CITED1 is expressed in both the cytoplasm and nucleus of Wilms tumor (WT) blastemal cells. CITED1 in the cytoplasm confers stemness to cancer cells by inhibiting the Wnt-β/catenin signaling pathway, whereas CITED1 in the nucleus is more tumorigenic.37,38 In hepatoblastoma, CITED1 induces cell proliferation by upregulating the Wnt signaling pathway inhibitors kringle-containing transmembrane protein 1 and CXXC finger protein 4. 39
CITED1 is highly expressed in adenomatous polyposis coli (APC)-deficient CRC mouse models and human tissue samples, and knockdown of CITED1 extended the lifespan of APC-deficient CRC mice. As a member of the β-catenin degradation complex, APC negatively regulates β-catenin. Loss of APC can induce activation of the Wnt-β/catenin signaling pathway and enhance c-Myc-dependent CITED1 transcription, whereas negative feedback of upregulation of CITED1 expression inhibits hyperactivation of the Wnt signaling pathway and maintains the activity of the Wnt signaling pathway at the level promoting tumor development (Figure 5). 23 In addition, CITED1 also promotes tumor cell proliferation by inhibiting the Wnt/β-catenin signaling pathway in PTC. 40

The mechanism of CITED1 in APC-deficient CRC. Loss of APC can induce activation of the Wnt-β/catenin signaling pathway and enhance c-Myc-dependent CITED1 transcription, whereas negative feedback via CITED1 upregulation inhibits hyperactivation of the Wnt signaling pathway and maintains the activity of this pathway at the level promoting tumor development. CITED1, CBP/p300 interacting transactivator with Glu/Asp-rich carboxy-terminal domain 1; APC, adenomatous polyposis coli; CRC, colorectal cancer.
Androgen/androgen receptor (AR) signaling pathway
The androgen/AR signaling pathway plays an important role in all stages of prostate cancer occurrence and development. CITED1 enhances the transcriptional activity of AR. 15 Prior research found that CITED1 expression is not increased in human prostate cancer tissue samples, but it is highly expressed in PIN-like tissue samples (a precursor to prostate cancer) of mice carrying the AR E231G mutation. Knockdown of CITED1 can reduce the viability and proliferation of LNCaP prostate cancer cells. 41 Thus, it is speculated that CITED1 plays a role in promoting the survival of premalignant cells during prostate cancer initiation, but its sustained overexpression is not necessary for tumor progression.
Potential role of CITED1 in clinical tumor diagnosis and treatment
CITED1 expression is higher in melanoma, PTC, WT, and CRC than in normal tissues, and its expression is closely related to tumor stage, metastasis, and prognosis.
In melanoma, high CITED1 expression is considered a biomarker distinguishing melanoma from benign nevus. 42 The BRAF V600E mutational load is negatively correlated with melanoma progression. 43 Therefore, a low mutational load of BRAF V600E (<5%) combined with the synergistic high expression of CITED1 can be used as an indicator of poor prognosis of melanoma. CITED1 has also been proven to negatively regulate MITF, specifically repressing MITF to maintain its level in a range compatible with tumorigenesis, suggesting that CITED1 can be a therapeutic target for genetic manipulation. Successful implementation of such a strategy would result in the cell-specific enhancement of MITF expression and potentially induce cyclin-dependent kinase inhibitor 1A (CDKN1A)/p21 and CDKN1C/p57-dependent cell growth arrest, thereby increasing the sensitivity of melanoma to chemotherapy. 11
In PTC, previous studies confirmed that Hector Battifora mesothelial antigen-1 (HBME-1) is the most sensitive (92.8%) and specific (86.4%) marker distinguishing PTC from benign thyroid nodules, and negative staining for cytokeratin 19 (CK19) is helpful in excluding PTC. As CITED1 is preferentially expressed in PTC but not in follicular carcinoma and normal thyroid tissue, it is recommended to combine CITED1 with HBME-1 and CK19 to attain high sensitivity and specificity for differential diagnosis.44–48 In addition, high CITED1 expression is associated with clinical stage (P = 0.003) and lymph node metastasis (P = 0.006) in PTC. 49 Studies revealed that CITED1 promotes the proliferation of PTC cells via the regulation of p21 and p27, 50 and CITED1 silencing might inhibit the progression of PTC cells by inhibiting the PI3K/AKT pathway. 51 All of these findings indicate that CITED1 can be used as an indicator of diagnosis and prognosis in PTC, and it might be a potential therapeutic target in the treatment of PTC.
The level of CITED1 in WT blastema is positively correlated with tumor stage, and it is expected to be a prognostic indicator of WT. 36 CITED1, as a marker of nephrogenic progenitor cells with cancer stem cell characteristics in WT, might represent a target for novel therapy that inhibits cell proliferation and promotes terminal differentiation.36,38,52 Other scholars proposed that dual immunofluorescence staining of CITED1 and epithelial cell markers (such as cytokeratin) can improve the recognition ability of blastemal WT in histologically high-risk groups and thus guide treatment. 53
In the diagnosis and treatment of CRC, the detection rate of early CRC has increased greatly with the improvement of endoscopic equipment and techniques, and the presence of lymph node metastasis has become a major concern to determine the treatment strategy. In T1 CRC, high CITED1 expression is an independent risk factor for lymph node metastasis (P = 0.010). 54 Therefore, CITED1 can be used as a potential marker to predict CRC metastasis and prognosis and to guide treatment.
Of course, the mechanism of CITED1 in the aforementioned tumors remains unknown, and further research is needed. Meanwhile, additional research is also needed to determine whether CITED1 plays a role in other tumors and whether it has potential diagnostic and therapeutic functions. For example, high-throughput sequencing of a cancer-associated fibroblast-induced EMT model of lung cancer cells revealed that CITED1 was upregulated in the early EMT stage of A549 cells (3–24 hours). 55 In addition, Kaplan–Meier plotter database analysis demonstrated that CITED1 expression was negatively correlated with progression-free survival (hazard ratio [HR] = 1.54, P < 0.001) and overall survival (HR = 1.17, P = 0.024) in lung cancer. We believe that with increasing research on CITED1, its tumor-promoting effect will be clarified.
Association of CITED1 with beige fat
Beige fat is produced by white fat in response to certain inducible factors through a process called fat/adipose tissue “browning” or “beiging.”56,57 Similarly as brown fat, beige fat is a thermogenic adipose tissue. However, unlike brown fat, which degrades and disappears with age, beige fat persists in the dorsal spine region of the neck, above the clavicle, and around the aorta in adults, and it can be metabolized to produce heat after stimulation such as cold stimulation, conferring therapeutic effects on obesity and type 2 diabetes. 58 As a newfound marker of beige fat, the focus of recent research on CITED1 has shifted to the regulation of beige fat formation, obesity, and type 2 diabetes. Cyanidin-3-glucoside, IL4, and peroxisome proliferator-activated receptor γ upregulate CITED1 expression to induce beige fat formation.59–61 Chronic inflammation promotes tumor development and progression, such as that induced by Helicobacter pylori infection in gastric cancer and human papillomavirus infection in cervical cancer. 62 Studies indicated that prolonged exposure of adipose tissue macrophages to interferon-γ can increase the expression of interferon-stimulated genes such as CITED1, resulting in adipose tissue “browning” and enhanced pro-inflammatory activity.63,64 However, it has not been confirmed whether excess lactate released during adjacent adipose tissue “browning” can promote tumor progression. 65 It is believed that with further research, the relationships of CITED1 with tumor development and progression through beige fat will be elucidated.
Conclusion and prospects
CITED1, as a transcriptional activator, performs transcriptional regulation through multiple signaling pathways including TGF-β/BMP/SMAD, estrogen, Wnt-β/catenin, and androgen/AR. Studies on CITED1 in tumors to date have mainly focused on melanoma, PTC, breast cancer, WT, and CRC. CITED1 plays important roles in tumor proliferation, invasion, and metastasis. Meanwhile, CITED1 is expected to become a potential target for tumor diagnosis and treatment in the future. First, in terms of diagnosis, CITED1 can be used as a diagnostic marker for solid tumors. Through immunohistochemical staining of histopathological section, CITED1 can be used in combination with other markers such as BRAF V600E, and HBME-1 to improve the specificity and sensitivity of tumor diagnosis. Second, as a marker of early tumor metastasis, we can predict potential metastases that have not yet been uncovered by imaging by detecting CITED1 expression in ctDNA and exosomes. Finally, as a marker of advanced tumor metastasis, the severity of metastasis can be assessed by detecting CITED1 levels in metastases and peripheral blood. In terms of treatment, CITED1 can play an anti-tumor role as a therapeutic target. CITED1 is a mediator in multiple signaling pathways, and it is expected to become a new target after drug resistance in upstream and downstream targets. CITED1 is also closely related to tumor metastasis, and this could permit new strategies for targeted therapy for tumor metastasis. In terms of prognosis assessment, the relationship between CITED1 expression and patient survival can be explored through the follow-up of patients in different tumor stages, and we believe that there will be unexpected results.
Footnotes
Author contributions
Wenting Chen, Jianing Jiang, and Jinqi Gao contributed equally to this work. All authors read and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by grants from the National Natural Science Foundation of China (#81803109), the Doctor Study-up Foundation of Liaoning Province (2019-BS-010), the High-level Talent Innovation Support Plan of Dalian (2021RQ018), and the Research Project of Dalian Medical Science (2211033).
