Abstract
Objective
To explore the potential of the transcription factor LMX1B and downstream gankyrin as prognostic biomarkers of glioma.
Methods
The expression levels of gankyrin and LMX1B were detected in 52 normal brain specimens and 339 glioma specimens. Correlations of gankyrin and LMX1B expression levels with pathological stages and clinical characteristics were statistically analyzed. Furthermore, the binding of LMX1B to the gankyrin promoter was evaluated using ALGGEN PROMO.
Results
Levels of LMX1B and gankyrin were significantly increased in tumor tissue, and were significantly associated with advanced glioma grade and poor survival. Compared with gankyrin- and LMX1B-negative glioma, the mean survival of patients with higher gankyrin and LMX1B expression was significantly reduced, from 83.46 to 18.87 months and from 63.79 to 18.29 months, respectively. Furthermore, LMX1B had a moderate positive correlation with gankyrin expression (Pearson’s
Conclusions
Increased expression of LMX1B and gankyrin has independent prognostic value in glioma patients. The transcription factor LMX1B may have an upstream role in the mechanism of action.
Introduction
Glioma is the most common type of primary brain tumor in the world, and most patients with glioma have a poor prognosis. In addition, gliomas have considerable impacts on patients’ physical, psychological, and social wellbeing. Although radiotherapy and adjuvant use of temozolomide are currently standard post-surgical treatments for patients with stage IV glioma (glioblastoma multiforme [GBM]), the survival of GBM patients is very poor. 1 The 5-year survival rate using this standard therapy is approximately 5%, and the median survival time is around 12 to 18 months.2–4 In recent years, targeted therapies have attracted increasing attention, and many studies are in clinical trial stages. To date, however, most of these trials have failed to achieve the expected results. The main challenges in developing new therapeutic strategies include the heterogeneity of gene expression in GBM and the complex interactions between multiple signaling pathways. 5 There is therefore an urgent need to identify other potential GBM biomarkers and clarify their possible mechanisms of action.
LIM homeobox transcription factor 1-beta (LMX1B), which belongs to the LIM-homeodomain (LIM-HD) protein family, plays a crucial role in co-regulating a subset of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) target genes. 6 LMX1B contains two LIM domains at the NH2-terminus and a central HD. The HD of LMX1B can recognize AT-rich elements (also known as FLAT elements), which contain the 5'-ATTA-3' (reverse 5'-TAAT-3') core sequence in the promoter or intron region.7,8 LMX1B can functionally cooperate with NF-κB to bind the corresponding recognition sites of target genes and recruit specific cofactors to activate or repress transcription. It has been recently reported that LMX1B is highly expressed in ovarian cancer cells, 9 laryngeal squamous cell carcinoma, 10 and radioresistant esophageal cancer cells. 11 However, the role of LMX1B in the survival outcome of glioma, and its possible mechanisms of action, remain unclear.
Gankyrin (also known as PMSD10) is one of the non-ATPase regulatory subunits in the assembly of the 26S proteasome, and is involved in proteasome-mediated protein unfolding and degradation.12,13 The ankyrin-repeat oncoprotein gankyrin regulates cell cycle and apoptosis balance by binding to mouse double minute 2 homolog (MDM2) or cyclin-dependent kinase 4 (CDK4), thereby enhancing the degradation of retinoblastoma 1 (RB1) and p53.14–16 In addition to playing a key role in the proteasome, gankyrin can also translocate into the nucleus and induce autophagy by promoting the binding of heat shock factor 1 (HSF1) to the autophagy related 7 (ATG7) promoter. 17 Moreover, gankyrin has recently been reported as highly expressed in a variety of malignancies, including gastric cancer, colorectal cancer, breast cancer, hepatocellular cancer, cholangiocarcinoma, ovarian cancer, cervical cancer, and endometrial cancer.13,18–26 However, the key role of gankyrin in gliomas, and its possible regulatory mechanisms, remain unclear.
To explore the roles of gankyrin and LMX1b in glioma, the expression of gankyrin and LMX1B were examined by immunohistochemistry (IHC) in 339 glioma specimens and 52 normal brain specimens. The associations of gankyrin and LMX1b with clinicopathological characteristics and overall survival were evaluated. We demonstrated that LMX1B-regulated gankyrin is a novel biomarker for tumor progression and prognosis in patients with glioma. Both gankyrin and LMX1B expression levels were positively associated with advanced tumor stages, Karnofsky performance status (KPS) scores, and overall survival of glioma patients. Furthermore, LMX1B may act as a transcription factor and cooperate with NF-κB to regulate gankyrin expression by binding to its promoter region.
Methods
Patients and specimens
Tissue specimens were collected from the ShengJing Hospital of China Medical University from January 2008 to January 2015. Clinical and pathological features were classified in accordance with the Cancer Staging Manual by the American Joint Committee on Cancer (seventh edition). The glioma pathological grades were diagnosed and scored by two experienced pathologists using the World Health Organization (WHO) classification. All glioma cases were newly diagnosed patients who had not previously received intracranial surgery, chemotherapy, or radiotherapy. All glioma patients were advised to receive adjuvant radiotherapy and chemotherapy in accordance with the contemporary versions of the National Comprehensive Cancer Network (NCCN) guidelines. Almost all patients underwent total resection followed by radiotherapy and chemotherapy. Patient follow-up was performed by phone or social media, or as outpatients. For the control group, normal brain tissue specimens that had been excised for non-tumor diseases, such as trauma or hypertensive cerebral hemorrhage, were used. This study was approved by the Ethics Committee of ShengJing Hospital of China Medical University, and written informed consent was obtained from each patient.
IHC staining and analysis
IHC staining was performed on formalin-fixed, paraffin-embedded tissue sections. Briefly, the paraffin-embedded specimens were sliced into consecutive sections of 4-µm thickness, mounted onto glass slides, and incubated at 65°C for 30 minutes. After being deparaffinized in xylene and rehydrated in decreasing concentrations of ethanol, antigen retrieval was performed by heating the sections in 0.01 M (pH 6.0) citrate buffer in the microwave for 10 minutes. To inhibit endogenous peroxidase activity, the sections were then incubated in 0.3% H2O2 for 10 minutes at room temperature. After washing with phosphate-buffered saline (PBS), the tissue sections were blocked with 10% normal goat serum (MXB Biotechnologies, Fuzhou, China) for 10 minutes and then incubated with primary antibodies against LMX1B (1:100; Abcam, Cambridge, USA) or gankyrin (1:100; Abcam) overnight at 4°C. After washing with PBS to remove unbound antibodies, the sections were incubated with biotinylated goat anti-rabbit IgG (1:500; MXB, Fuzhou, China) for 30 minutes at room temperature. After removing unbound secondary antibodies with PBS, the sections were incubated with streptavidin–peroxidase complex (MXB) for 10 minutes, stained with 3,3ʹ-diaminobenzidine tetrahydrochloride (DAB; Sigma, St. Louis, USA) for 10 minutes at room temperature, and counterstained with hematoxylin. The IHC images were obtained using a light microscope (Eclipse NI; Nikon, Tokyo, Japan).
The expression intensities of gankyrin and LMX1B in the IHC images ware independently assessed by two pathologists using an immunoreactivity score (IRS) system.27,28 This IRS system scored the percentage of positive cells (4: ≥80% of positive cells; 3: 51%–80% of positive cells; 2: 10%–50% of positive cells; 1: ≤ 10% of positive cells; and 0: no positive cells) and the staining intensity (3 = intense reaction; 2 = moderate reaction; 1 = mild reaction; and 0 = no color reaction). The final IRS score (range: 0–12) was calculated by multiplying the score of the percentage of positive cells (0–4) by that of the staining intensity (0–3). Finally, the expression intensities of gankyrin and LMX1B in IHC images were sorted into four categories: negative (IRS score = 0–1), low (IRS score = 2–3), medium (IRS score = 4–8), and high (IRS score = 9–12).
UALCAN dataset analysis and promoter analysis of gankyrin
The expression profiles of gankyrin and LOB domain-containing protein 1 (LDB1) were analyzed using the UALCAN database (http://ualcan.path.uab.edu/index.html), which contains cancer transcriptome data. Data from both normal and GBM cases were used, and gene expression was stratified by age, race, and sex. In addition, the transcriptional binding sites (TFBS) on the gankyrin promoter region were analyzed using ALGGEN PROMO (http://alggen.lsi.upc.es/home.html), which defines TFBS according to the TRANSFAC database. After analyzing 1700 bp upstream of the gankyrin promoter region, five consensus FLAT core binding sequences were identified, and three NF-κB binding sites were found in the vicinity of FLAT elements.
Statistical analysis
Statistical analyses were performed using SPSS Statistics for Windows, version 18.0 (SPSS Inc., Chicago, IL, USA). Relationships between clinicopathological factors and LMX1B or gankyrin expression were analyzed using the χ2 test and logistic regression analysis. Correlations between the stratified expression levels of gankyrin or LMX1B (low, medium, or high) and clinicopathological factors were analyzed using Spearman’s correlation coefficient. Survival curves were plotted using the Kaplan–Meier method, and significant differences between groups were compared using the log-rank test. Cox’s proportional hazards regression analysis was performed to identify which factors might have a significant influence on survival. Differences with
Results
Upregulation of gankyrin and LMX1B expression in brain tumors
To evaluate whether gankyrin was associated with brain tumorigenesis, the UALCAN database was used to examine the expression of gankyrin in GBM samples. Data from five normal and 156 GBM cases were analyzed. Compared with normal brain tissue, gankyrin expression was upregulated in GBM samples (Figure 1). Sex-, race-, and age-stratified gankyrin expression also indicated that gankyrin expression may play an important role in the prognosis of brain tumors. Thus, to further understand the role of gankyrin in brain tumorigenesis, we used IHC to analyze gankyrin expression levels in 391 brain specimens, which consisted of 52 normal brain specimens and 339 glioma specimens. Gankyrin expression was markedly higher in grade III and IV gliomas than in grade I and II gliomas (Figure 2a). Interestingly, higher expression of the transcription factor LMX1B was also related to higher tumor stage in glioma tissue (Figure 2b). Histograms of gankyrin and LMX1B expression by IHC, based on the IRS system, are shown in Figure 2c.

Expression profiles of gankyrin in glioblastoma multiforme (GBM) patients by UALCAN dataset analysis. (a) Gankyrin expression was upregulated in 156 GBM tissue specimens compared with five normal brain tissue specimens. Gankyrin expression stratified by sex, race, and age are shown in the results of (b), (c), and (d), respectively.

Gankyrin and LMX1B expression levels were associated with advanced tumor stage of glioma. Representative images of immunohistochemical staining of gankyrin (a) and LMX1B (b) in normal brain tissue (NBT) and glioma tumor tissue with different World Health Organization grades. Upper panel: magnification ×200. Lower panel magnification ×400. (c) Histogram of gankyrin and LMX1B expression levels based on the immunoreactivity score (IRS) system from immunohistochemistry experiments. * indicates a significant difference compared with NBT, # indicates a significant difference compared with grade I tissue, ζ indicates a significant difference compared with grade II tissue, and ξ indicates a significant difference compared with grade III tissue. *, #, ζ, and ξ represent
Gankyrin and LMX1B expression levels are associated with different tumor grades of glioma
Next, we examined the associations between clinicopathological factors and gankyrin or LMX1B expression. The expression levels of both gankyrin and LMX1B were determined using IHC staining and quantified using the IRS scoring system. Table 1 shows the relationship between gankyrin expression and clinicopathological factors. Although gankyrin expression was not correlated with age, sex, or comorbidities in patients with glioma, the KPS values in patients with gankyrin-negative glioma tumors were mostly higher than 80 (
Relationship between gankyrin expression and clinicopathological factors.

1. Cases with IHC staining score more above 1+ were defined as positive expression. 2. Pathological staging was performed according to the WHO classification system. 3. Abbreviation: IHD, Ischemic Heart Disease; COPD, Chronic Obstructive Pulmonary Disease.
Relationship between LMX1B expression and clinicopathological factors.
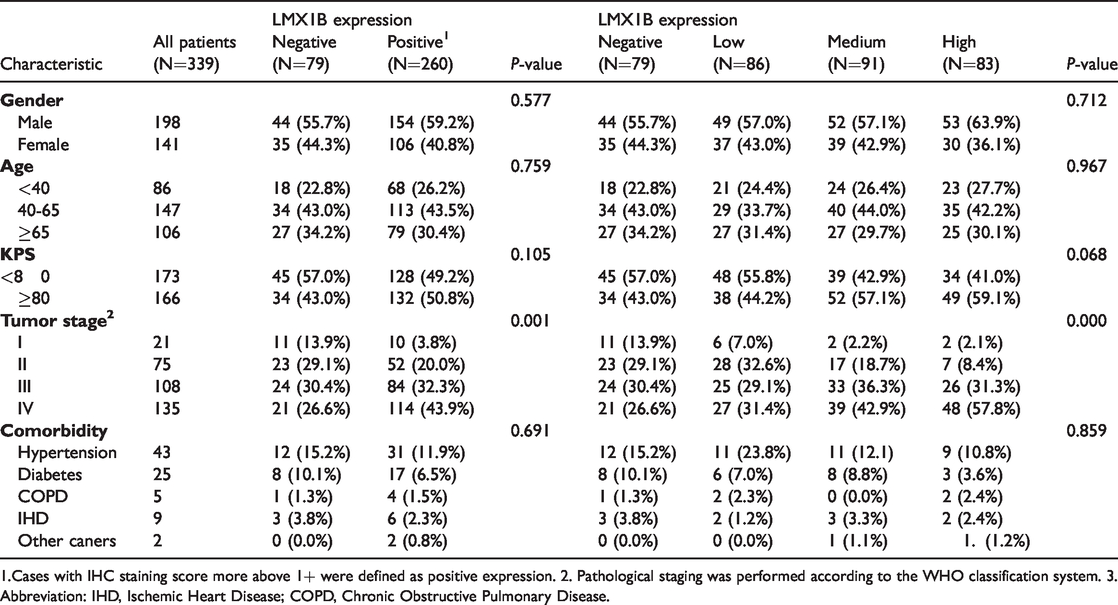
1.Cases with IHC staining score more above 1+ were defined as positive expression. 2. Pathological staging was performed according to the WHO classification system. 3. Abbreviation: IHD, Ischemic Heart Disease; COPD, Chronic Obstructive Pulmonary Disease.
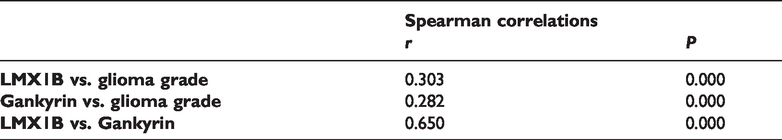
Spearman’s correlation analysis of the correlation between gankyrin, LMX1B, and glioma grade.

Association of poor survival with gankyrin expression, LMX1B expression, and clinicopathological characteristics in glioma patients.

Gankyrin as the hypothetical target of the transcription factor LMX1B
Because LMX1B has characteristics of a transcription factor, it is likely that LMX1B acts on the gankyrin promoter in glioma, which results in both gankyrin and LMX1B being related to advanced stages of glioma. Thus, the promoter region of gankyrin was analyzed. As expected, five consensus LMX1B core binding sequences were identified upstream of the gankyrin promoter region. Because LMX1B regulates gene expression in cooperation with NF-κB and E47, we further examined whether NF-κB and E47 were distributed near these LMX1B binding sites. Three NF-κB and multiple E47 binding sites were located in the vicinity of the LMX1B consensus site (Figure 3), suggesting that LMX1B may cooperate with NF-κB and E47 to regulate gankyrin expression in glioma tumors.

Gankyrin as the potential target of transcription factor LMX1B. (a) Bioinformatic analysis of 1700 bp upstream of the gankyrin promoter region, using PROMO/TRANSFAC. In addition to identifying five LMX1B (FLAT element) binding sites in the gankyrin promoter, there were also three NF-κB and seven E47 binding sites located in the vicinity of the LMX1B binding sites.
Higher gankyrin and LMX1B expression is associated with worse prognosis in glioma patients
We next examined whether gankyrin and LMX1B expression was associated with survival outcomes in patients with gliomas. Table 4 presents the results of the associations between poor survival and clinicopathological characteristics, gankyrin expression, and LMX1B expression. Poor survival was associated with older age (hazard ratio [HR] = 1.83 for 40–65 years and HR = 4.61 for ≥65 years,
A Kaplan–Meier survival analysis was then conducted to appraise the prognostic value of gankyrin and LMX1B in glioma patients. Poor overall survival in glioma patients was significantly associated with gankyrin expression, LMX1B expression, KPS score, and tumor grade (Table 5). The mean overall survival of gankyrin-negative and gankyrin-positive glioma patients was 76.26 and 38.56 months, respectively. In addition, the mean overall survival of LMX1B-negative and LMX1B-positive glioma patients was 73.65 and 38.13 months, respectively. Moreover, the association between advanced glioma (stage IV) and gankyrin and LMX1B expression levels were analyzed. As shown in Table 6, higher gankyrin and LMX1B expression levels were associated with worse overall survival in advanced glioma patients. Compared with gankyrin-negative patients (mean overall survival = 83.46 months), the mean overall survivals of advanced glioma patients with low, median, and high gankyrin expression were significantly reduced to 40.62, 20.48, and 18.97 months, respectively (
Associations between gankyrin, LMX1B, clinicopathological factors, and overall survival.

Overall survival according to gankyrin and LMX1B expression levels.
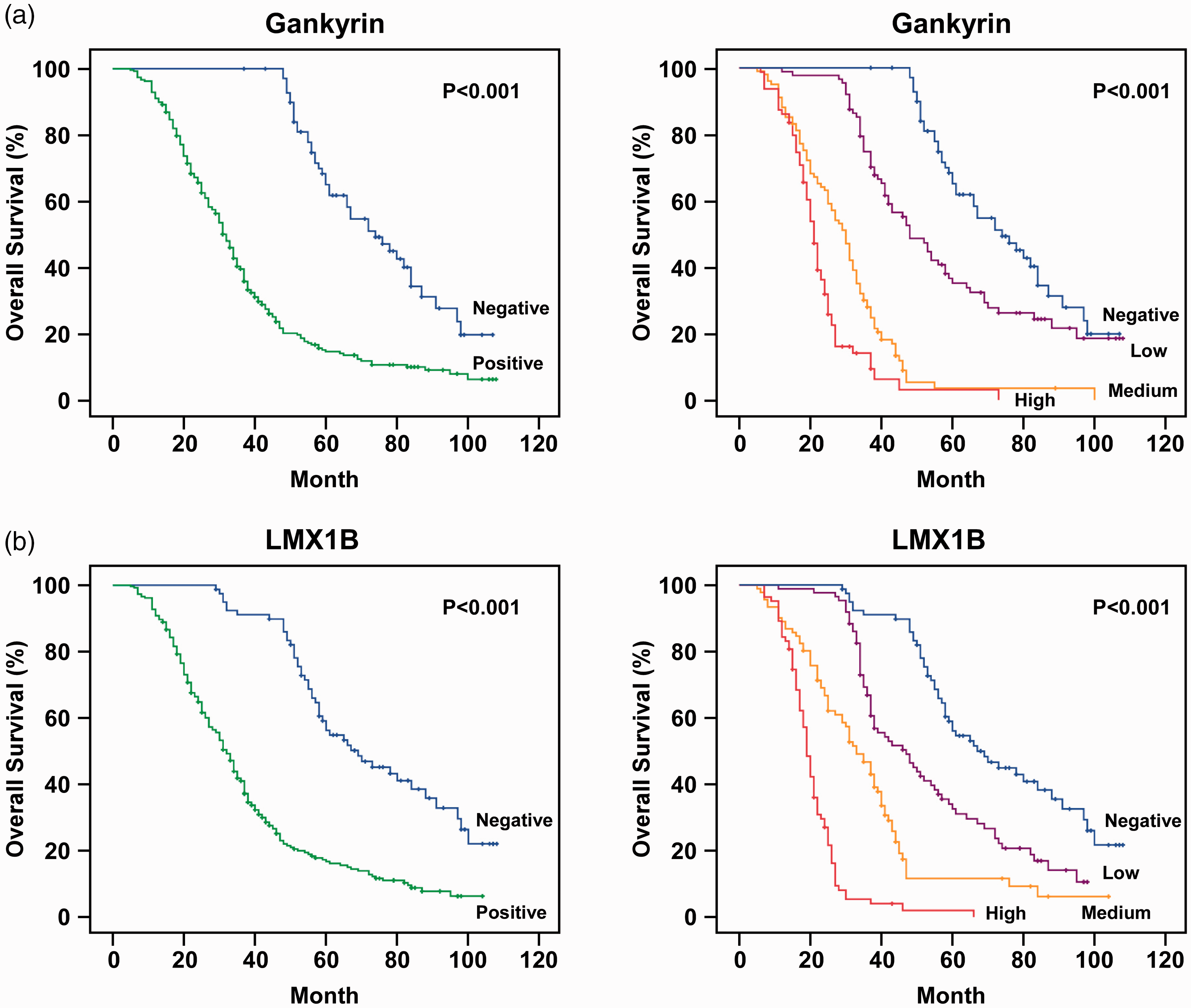
Higher gankyrin and LMX1B expression levels were associated with worse survival in patients with glioma. (a) Kaplan–Meier survival analysis of 339 glioma patients stratified for gankyrin expression. Higher gankyrin expression was significantly associated with worse survival. (b) Survival analysis stratified by LMX1B expression in 339 glioma patients. Higher LMX1B expression was significantly associated with worse survival.
Discussion
In the current study, we demonstrated for the first time that gankyrin and its transcription factor LMX1B are both upregulated in glioma and are associated with poor prognosis in glioma patients. The possible mechanism of action may be that LMX1B acts as a transcription factor in cooperation with NF-κB and E47, binding to the gankyrin promoter and regulating gankyrin expression in glioma patients. Thus, it appears that both gankyrin and LMX1B are independent risk factors for survival prognosis in glioma patients.
In the present study, IHC analysis revealed that both gankyrin and LMX1B were highly expressed in glioma tissues and were associated with tumor grade (Figure 2). As an indispensable chaperone for the assembly of the 26S proteasome, gankyrin can act as a nuclear–cytoplasmic shuttling protein to accelerate NF-κB1–RELA nuclear export, 29 or as a transcription factor to induce autophagy by translocating into the nucleus and cooperating with the transcription factor HSF1 to bind the ATG7 promoter. 17 Autophagy plays a vital role in the malignancy, senescence, radioresistance, and chemoresistance of glioma.30–32 It is therefore likely that LMX1B cooperates with NF-κB and E47 to regulate gankyrin expression in glioma patients, and upregulated gankyrin subsequently promotes the malignancy and poor prognosis of glioma by regulating autophagic flux. This hypothesis warrants further investigation.
The transcription factor LMX1B participates in protein–protein interactions and binds to FLAT elements on the promoter regions of target genes. LMX1B is able to synergistically cooperate with E47 and NF-κB to activate insulin, interleukin (IL)-6, and IL-8 genes by binding to FLAT elements.6,33,34 Consistent with this idea, the gankyrin promoter contains three NF-κB and multiple E47 binding sites (Figure 3), suggesting that LMX1B, NF-κB, and E47 transcription factors functionally cooperate to regulate gankyrin expression in glioma. However, several corepressors, such as LDB1 and E3 ubiquitin-protein ligase RLIM, have been identified to negatively regulate LMX1B function. LDB1 overexpression significantly inhibits activation of downstream gene expression via LMX1B alone, and synergistically via LMX1B and E47. 35 RLIM, a negative transcription repressor of LMX1B, can recruit the SIN3A/histone deacetylase corepressor complex to inhibit and target LIM-HD transcription factors for degradation. 36 To better understand whether LDB1 and RLIM corepressors are negatively associated with gankyrin expression patterns, we analyzed LDB1 and RLIM corepressor expression in patients with brain tumors using the UALCAN dataset. Interestingly, the expression of LDB1 (Figure 5) and RLIM (Figure 6) in GBM patients was exactly the opposite of that of gankyrin. Compared with normal brain tissue, LDB1 and RLIM were both downregulated in GBM tissue. Future research should focus on the detailed interplay among LDB1, RLIM, NF-κB, E47, and LMX1B in gankyrin activation during brain tumorigenesis.
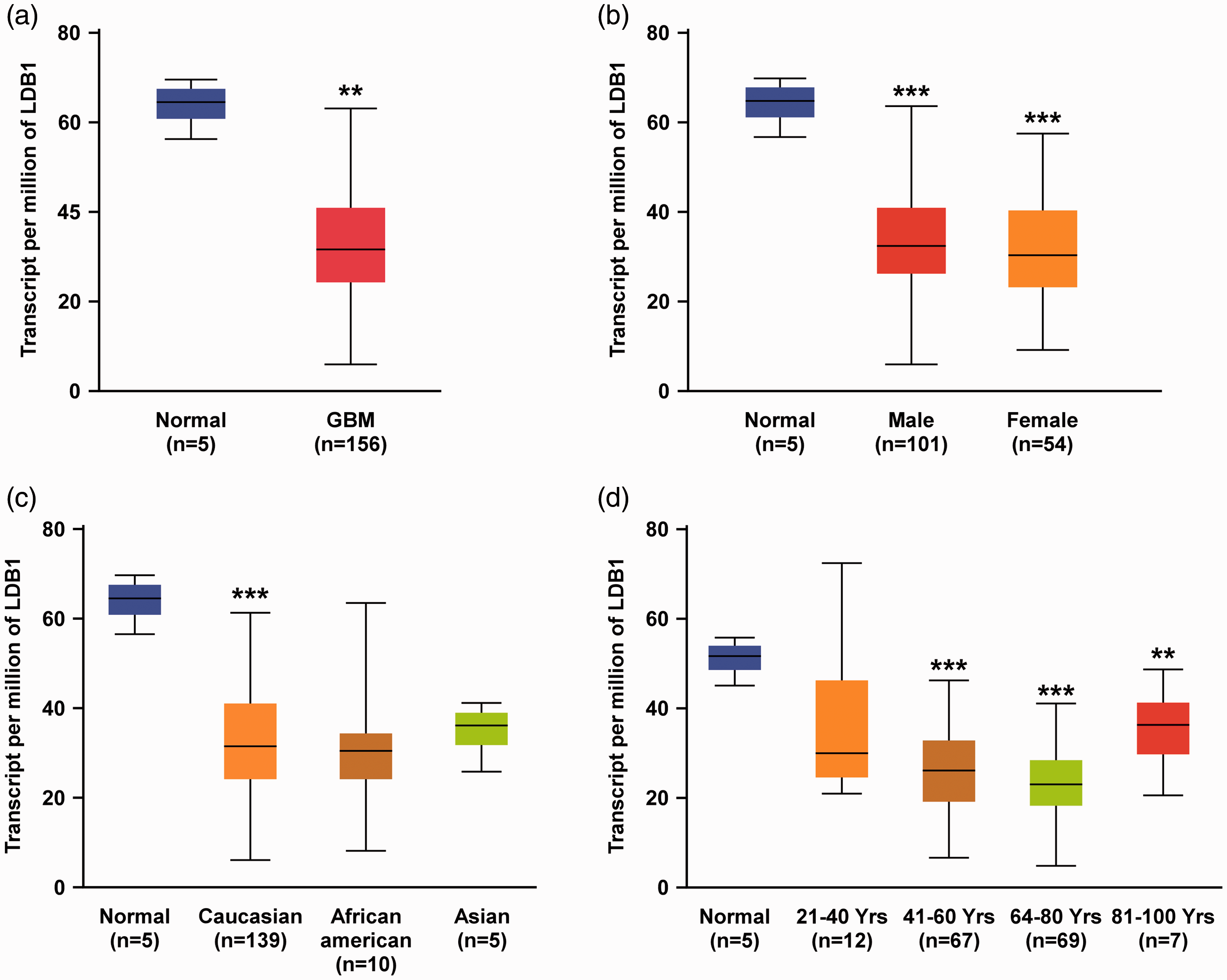
Expression profiles of LDB1 in glioblastoma multiforme (GBM) patients by UALCAN dataset analysis. (a) LDB1 expression was upregulated in 156 GBM tissue specimens compared with five normal brain tissue specimens. LDB1 expression levels stratified by sex, race, and age are shown in the analysis results of (b), (c), and (d), respectively. Differences were statistically significant at *

Expression profiles of RLIM in glioblastoma multiforme (GBM) patients by UALCAN dataset analysis. (a) RLIM expression was upregulated in 156 GBM tissue specimens compared with five normal brain tissue specimens. RLIM expression levels stratified by sex, race, and age are shown in the analysis results of (b), (c), and (d), respectively. Differences were statistically significant at *
In the current study, we demonstrated that LMX1B and gankyrin expression levels were significantly higher in advanced glioma tissue than in normal brain tissue, indicating that both LMX1B and gankyrin may play important roles in brain tumorigenesis. In addition, the expression levels of LMX1B and gankyrin were significantly and positively correlated with WHO grade. Because the WHO grade is a recognized marker of glioma proliferation and invasion, we speculated that LMX1B and gankyrin may exacerbate the malignant behavior of glioma cells. Consistent with this speculation, the Kaplan–Meier survival analysis supported the important roles of LMX1B and gankyrin in glioma; specifically, patients with higher LMX1B or gankyrin expression levels had lower overall survival rates. In addition, the expression levels of LMX1B and gankyrin in patients with KPS < 80 were significantly higher than those with KPS ≥80, supporting the idea that LMX1B and gankyrin may lead to poor clinical outcomes. These findings are also consistent with the findings of LMIMB and gankyrin expression in other cancers.9,13,18–26 Together, these findings indicate the clinical value of LMX1B and gankyrin as potential biomarkers or therapeutic targets in cancer, and especially for patients with advanced glioma.
In conclusion, LMX1B and gankyrin were both upregulated in glioma tissue, and their expression levels were positively associated with WHO grades, KPS scores, and overall survival. These findings indicate that LMX1B and gankyrin are potential prognostic biomarkers in glioma. Furthermore, the expression levels of LMX1B and gankyrin were positively correlated, and there were multiple binding sites of LMX1B and its co-transcription factors in the upstream promoter of gankyrin. Thus, LMX1B may act as a transcription factor in gliomas to upregulate gankyrin expression. The present study also suggests the value of LMX1B and gankyrin as potential biomarkers and therapeutic targets for glioma in the future. The exact regulatory mechanisms between LMX1B and gankyrin should be elucidated in further studies.
Footnotes
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
