Abstract
Posterior reversible encephalopathy syndrome (PRES) is a rare clinical disease, which has been seen in patients with systemic lupus erythematosus (SLE). Its main manifestations are seizure, headache and other neurological symptoms. While the condition is reversible, if not treated in time, there can be risks of cerebral haemorrhage. We report here the case of a young patient with SLE who developed PRES after receiving the immunosuppressant, mycophenolate mofetil. Neurological symptoms, signs, or changes in a patient’s condition that cannot be explained by lupus, should alert physicians to the possibility of the drug causing PRES, and prompt discontinuation should ensue.
Keywords
Background
Posterior reversible encephalopathy syndrome (PRES) is a clinical-radiology syndrome characterized by acute cerebral endothelial disease accompanied by continuous disruption of the blood-brain barrier, with seizures, headache, and visual impairment as the main symptoms. 1 Although rare, PRES has been reported in patients with SLE. 2 Several risk factors are associated with the development of PRES, including immunosuppressant drugs.2–4 We report here a case of PRES that developed in a patient with SLE after receiving mycophenolate mofetil.
Case report
A 16-year-old female patient, presented to our hospital because she had experienced repeated head neck pain, oedema, and foamy urine for the past 20 days. On examination, her weight was 52 kg, height 168 cm, vital signs were stable, blood pressure (BP) 125/68 mmHg, and she had a normal state of consciousness. While she had nephrotic facial features, pale lips, and concave swelling in both lower limbs, physical examination of her heart, lungs, abdomen, and nervous system showed no abnormalities. Laboratory tests showed reduced white blood cell (WBC) count, haemoglobin (Hb), platelets, complement C3 and C4 and elevated creatinine, cholesterol, and triglycerides (Table 1). Several inflammatory indicators were elevated as were several urinary parameters. A bone marrow biopsy showed megaloblastic anaemia with left shift of granulosa hyperplasia. Urinary ultrasound confirmed normal size of both kidneys.
Laboratory Results.
ANA, Antinuclear antibody; ANCA, Anti-neutrophil cytoplasm antibodies; CL-Ab, Cardiolipin antibodies; GP1, Glycoprotein-1; HPF, high power field; LPF, lower power field; MPO, myeloperoxidase; PR3, anti-proteinase 3; RBC, red blood cells; WBC, white blood cells.
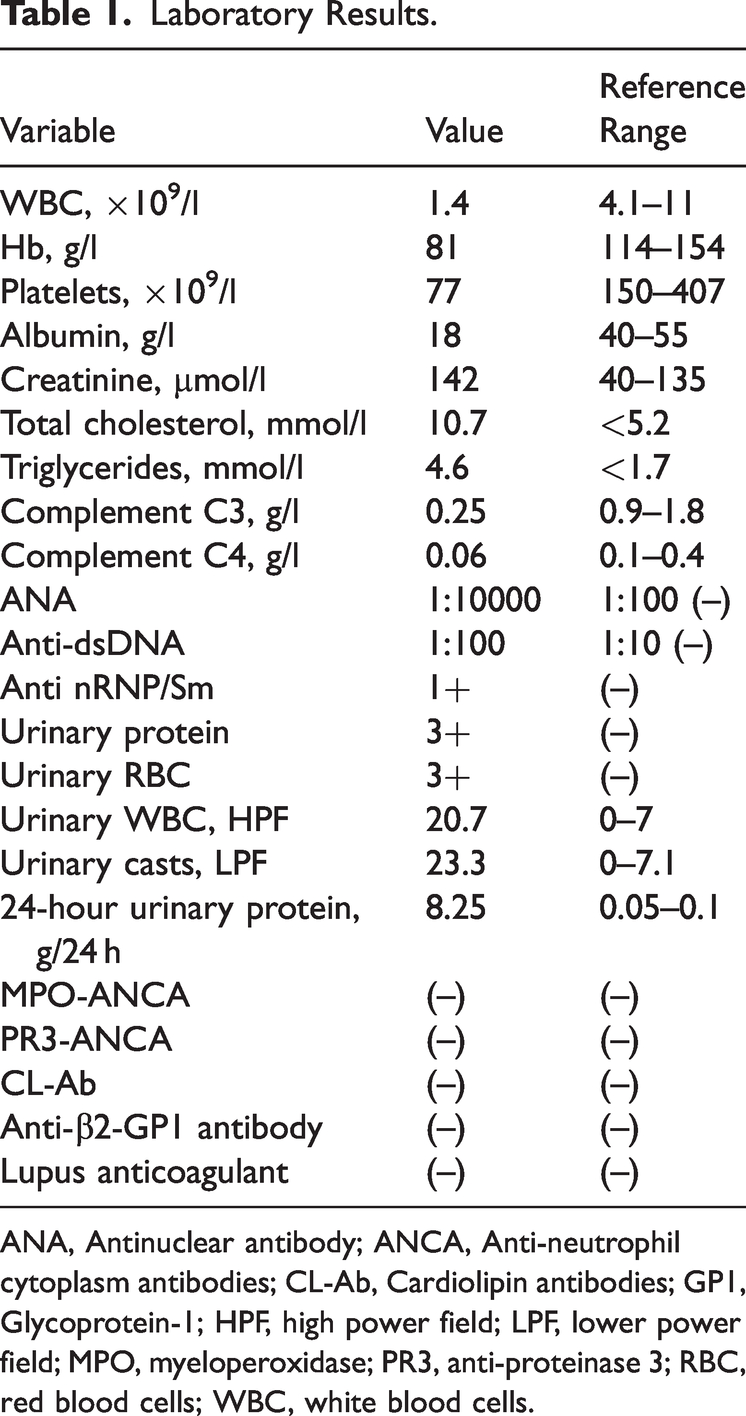
The first brain magnetic resonance image (MRI) scan showed that the two lateral ventricles, third and fourth ventricles and anterior pontine cistern were slightly widened, with mild traffic hydrocephalus (Figure 1). However, the patient’s renal pathology showed class IV and V lupus nephritis and acute renal tubulointerstitial injury. Her SLE Disease Activity Index without neurologic descriptors (SLEDAI-N) score was 18. Following a diagnosis of SLE, the patient was treated with immunosuppressive therapy that included IV methylprednisolone 40 mg per day and oral mycophenolate mofetil 750 mg twice a day for 20 days.

The first brain magnetic resonance image (MRI) (June 30, 2021) showed that the two lateral ventricles, third and fourth ventricles and anterior pontine cistern were slightly widened, with mild traffic hydrocephalus. Figure 1a and Figure 1b show that there were no abnormal signals in the brain parenchyma in T2-weighted images.
On the 20th day after treatment, the patient had a generalized tetanic convulsion, accompanied by foaming at the mouth, trismus, eyes turned up, and unconsciousness. Her BP was 135/84 mmHg. A lumbar puncture was performed, and the patient’s cerebrospinal fluid (CSF) pressure was 245 mmH2O. Routine cytological, and pathogenic examinations (i.e., bacteria, fungi, tuberculosis, TORCH) of the CSF fluid were negative. A second brain MRI scan with magnetic resonance venography (MRV) was taken and showed multiple abnormal signals in the bilateral frontal, parietal, temporal and occipital cortices, as well as in the subcortical white matter and the right side of the pons (Figure 2). These features suggested reversible posterior brain injury or osmotic demyelinating syndrome. However, the combination of the patient’s imaging results with her convulsions suggested lupus encephalopathy. Following treatment with methylprednisolone (500 mg/day) for three days, combined with intravenous immunoglobulin (IVIG) (20 g/day) for five days, intrathecal dexamethasone 10 mg and methotrexate 10 mg and IV mannitol to reduce intracranial pressure, the patient became conscious and her convulsions stopped. However, her systemic oedema worsened, her urine volume decreased, and her serum creatinine increased to 175 µmol/L. Plasma exchange in combination with Continuous Renal Replacement Therapy (CRRT) was initiated.
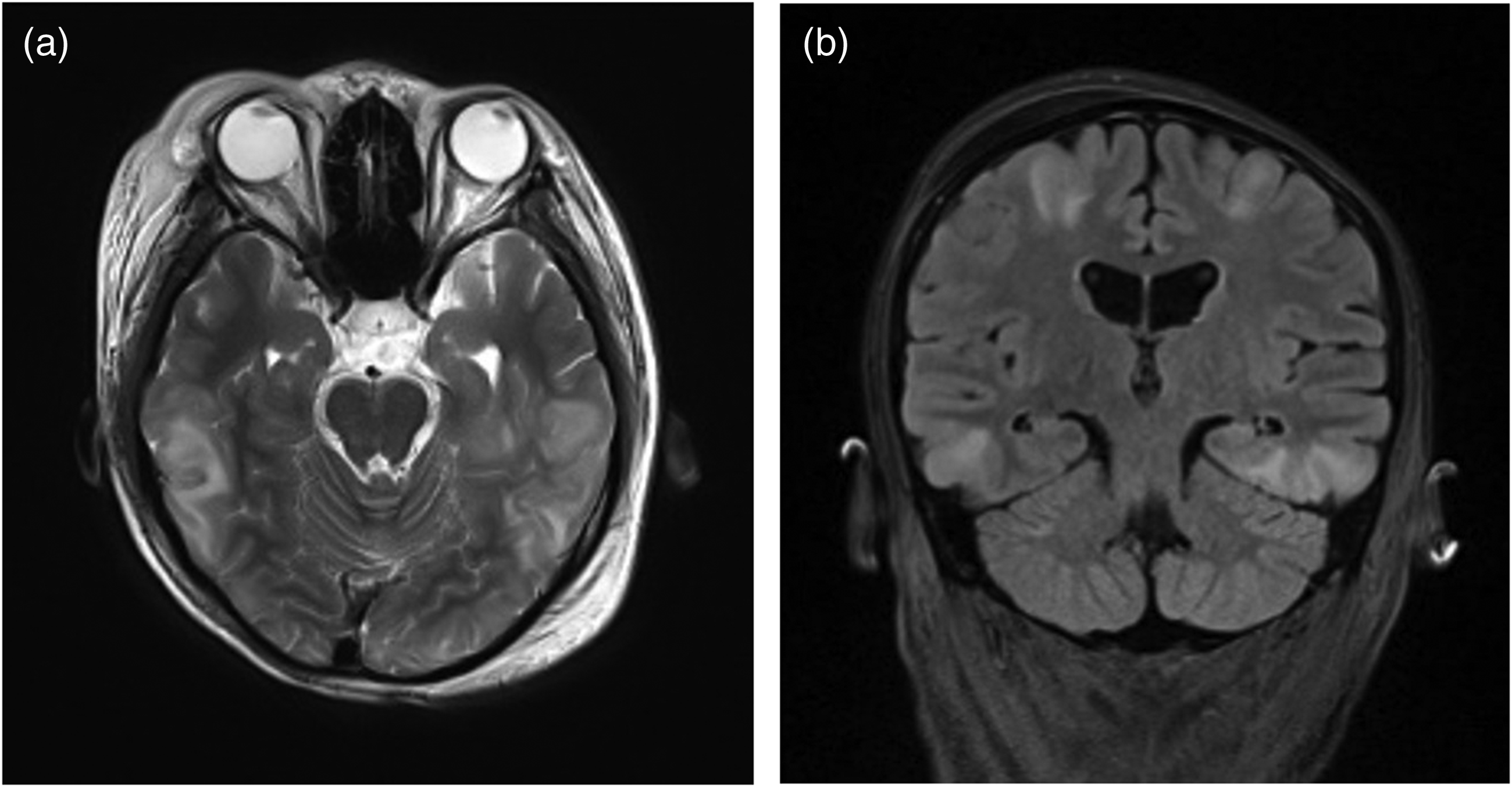
A second brain magnetic resonance image (MRI) with magnetic resonance venography (MRV) was taken 20 days later (July 20, 2021) and showed multiple abnormal signals in the bilateral frontal, parietal, temporal, and occipital cortices, as well as in the subcortical white matter and the right side of the pons. These features suggested reversible posterior brain injury or osmotic demyelinating syndrome. Figure 2a shows multiple abnormal signals newly detected in the temporal lobe in T2-weighted image. Figure 2b shows multiple abnormal signals newly detected in the parietal and occipital cortices in a fluid-attenuated inversion recovery (FLAIR) sequence.
A week later, the patient's vital signs were stable, her BP was 131/80 mmHg, her urine volume was 2000 ml/d, creatinine had decreased to 88 µmol/l, but although she had no convulsions, and normal limb movements, she still complained of dizziness. A lumbar puncture was taken and showed that CSF pressure had decreased to 150 mmH20. A third brain MRI scan showed evidence of a cerebral haemorrhage and hematoma (Figure 3). We considered that treatment with mycophenolate mofetil may have caused PRES and so stopped its administration and replaced it with IV 800 mg of cyclophosphamide once per month, 40 mg of methylprednisolone once a day and 480 mg of belimumab once per month. After using cyclophosphamide for eight months, it was replaced with belizumab 480 mg per month for the next 13 months. At the same time, the dosage of methylprednisolone was slowly reduced from 40 mg per day to 2 mg once every other day.
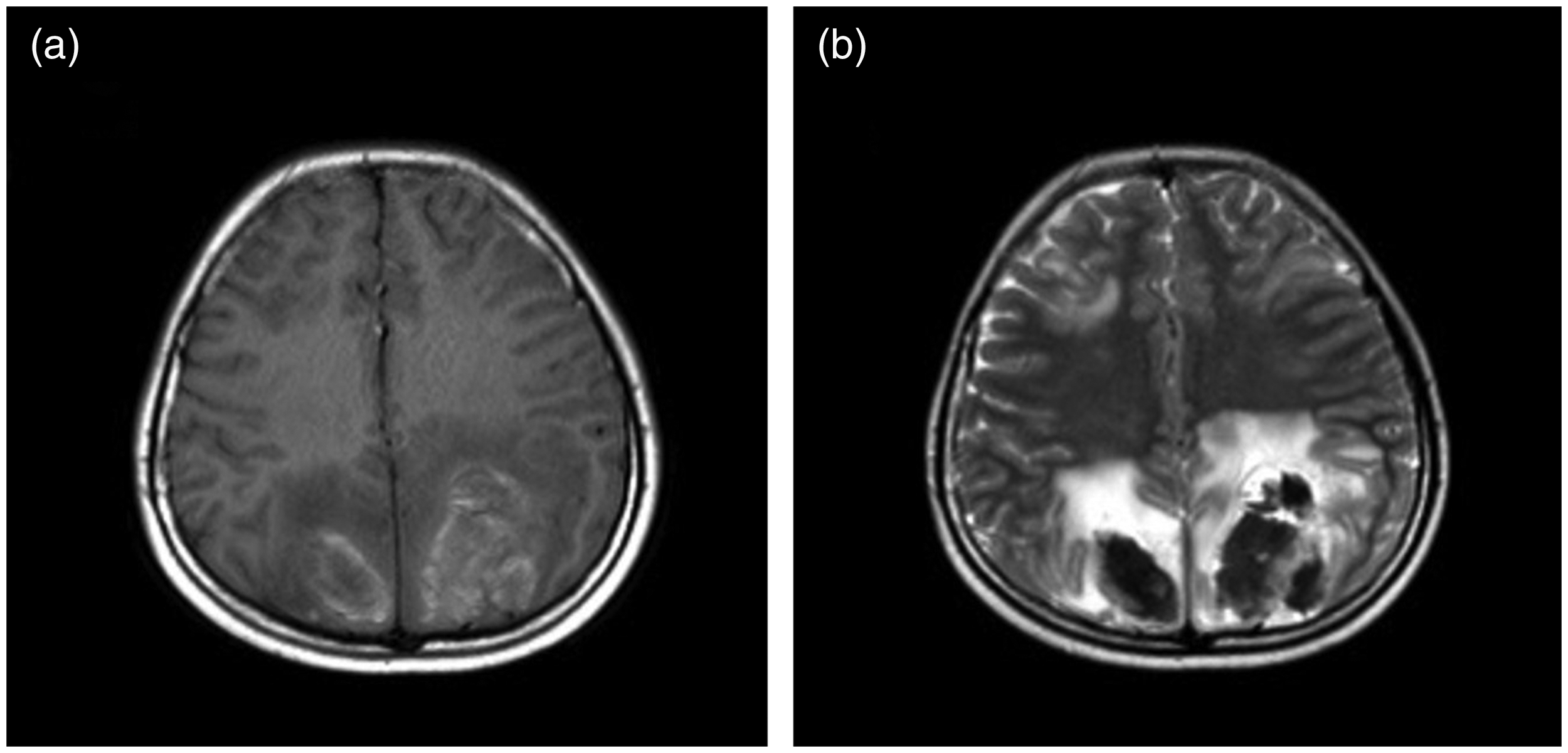
A third brain magnetic resonance image (MRI) taken seven days later (July 27, 2021) showed bilateral parietooccipital, frontal, and temporal lobes with newly added abnormal signals that suggested cerebral haemorrhage and partial hematoma formation. Figure 3a show cerebral haemorrhage and partial hematoma formation in bilateral parietooccipital, frontal, and temporal lobes in a T1-weighted image. Figure 3b show cerebral haemorrhage and partial hematoma formation in bilateral parietooccipital, frontal, and temporal lobes in a T2-weighted image.
At the two-year follow-up, the patient reported no lupus flare-ups nor any neurological sequelae. Her renal function and routine blood tests were within normal range (Complement C3, 1.24 g/l; C4, 0.29 g/l; antinuclear antibody (ANA), 1:3200; anti-dsDNA, negative). A fourth brain MRI taken at this time, showed abnormal signals in both parietal and occipital lobes, caused by the hematoma formation, but the scan was similar to that taken two years previously with a reduced T1-weighted image signal (Figure 4).

A fourth brain MRI taken at two-year follow-up (March 27, 2022) showed abnormal signals in both parietal and occipital lobes, caused by the haematoma formation, but the scan was similar to that taken two years previously, with a reduced T1-weighted image signal. Figure 4a show that the haematoma signals of parietal and occipital lobes were reduced in a T1-weighted image. Figure 4b show that the haematoma signals of parietal and occipital lobes were reduced in a T2-weighted image.
The reporting of this study conforms to CARE guidelines. 5 Written informed consent was obtained from the patient to publish her anonymised data and the case report did not require ethics committee approval.
Discussion
The neurological disorder PRES is a clinical-radiology syndrome, initially described in 1996, and characterized by acute brain endothelial disease with continuous destruction of the blood–brain barrier, vascular oedema and occasionally cytotoxic oedema.1,4 Clinically, its typical characteristics are acute attacks of nervous system symptoms (e.g., seizures, headaches, visual impairment, altered mental status, hemiplegia, aphasia, dysarthria, ataxia, dizziness) and various other focal neurological signs. 6 In addition, subarachnoid haemorrhage, parenchymal hematoma and small volume haemorrhages <5 mm have also been reported in 15%–19% of cases. 7 In a case control study involving 7059 patients with SLE, the prevalence rate of PRES was 0.43% and most patients had a younger age at onset compared with controls. 2 Risk factors of PRES include young age, kidney involvement, hypertension history, dyslipidaemia, lymphopenia, SLEDAI-N score >12, eclampsia, heart failure and WBC count >9 × 109/l.2,3 However, it is unclear if the association between SLE and PRES is related to the autoimmune disease itself, kidney injury associated with SLE or the immunosuppressant drugs (e.g., tacrolimus, cyclosporin, or mycophenolate mofetil ) frequently used in these patients. 2 In the case presented here, the patient with SLE was young and had risk factors of kidney involvement, hyperlipidaemia, SLEDAI-N score >18 and was receiving mycophenolate mofetil. We considered that mycophenolate mofetil had caused PRES and so discontinued treatment.
A brief literature search identified two other studies that had reported a possible association between mycophenolate mofetil and the development of PRES in patients with SLE (Table 2).8,9 In one study, a 29-year-old female patient with stage IV lupus nephritis developed a seizure one week after taking mycophenolate mofetil. Examination of her brain MRI showed bilateral subcortical oedema in the posterior hemisphere. Following a diagnosis of PRES, the drug was immediately discontinued and replaced with cyclophosphamide. 8 A second brain MRI eight days later showed that the subcortical oedema had subsided, and the patient's mental state had improved, with no permanent neurological sequelae. In the second study, a 22-year-old female patient with stage IV lupus nephritis developed headache and seizure five days after starting treatment with mycophenolate mofetil. 9 A brain MRI examination showed abnormal signal intensity involving the parietal and occipital regions in T2-weighted, diffuse weighted image (DWI), apparent diffusion coefficient (ADC) and fluid-attenuated inversion recovery (FLAIR) images consistent with the diagnosis of PRES. Twelve days after stopping mycophenolate mofetil, a follow-up MRI confirmed resolution of the brain oedema, and no severe lupus activity was reported during a 6-month follow-up.
Previous case reports of mycophenolate mofetil induced PRES in patients with SLE.

CR: complete resolution; F, female; H: headaches; Hyp: hypertension; LN: lupus nephritis; PRES, posterior reversible encephalopathy syndrome; S: seizures; SLE: Systemic lupus erythematosus.
Other reports suggest that mycophenolate mofetil is a risk factor for PRES in other autoimmune disorders. For example, one study involving a 7-year-old boy with Evans syndrome who had been treated with different immunosuppressive treatments, including corticosteroids, cyclosporine and rituximab, had improved and so corticosteroids were tapered off. 10 During this period he received mycophenolate mofetil and two months later developed seizures. Diagnosis of PRES was made following a brain MRI. Cyclosporine and mycophenolate mofetil were immediately stopped and replaced with azathioprine. A repeat brain MRI one month later was completely normal. The patient had no further neurological symptoms during a 6-month follow-up period.
The underlying mechanise that cause PRES are unclear. It is thought that damage to the vascular endothelium by toxins and hypertension, causes dysfunction of the cerebral vasoconstriction regulation, and leads to a relative vasodilation and hyperperfusion of cerebral arterioles. 9 This hyperperfusion may lead to damage of the blood–brain barrier, allowing fluid, macromolecules and possible red blood cells to leak into the brain parenchyma, resulting in PRES. 11 In the setting of PRES induced by mycophenolate mofetil, the pathogenic mechanisms are also unclear. However, the drug inhibits the expression of vascular cell adhesion molecule 1 (VCAM-1) and intercellular adhesion molecule-1 (ICAM-)1, inhibits the remodelling of vascular wall, and leads to smooth muscle cell proliferation damage and reduction in connective tissue components secretion, which may lead to PRES. 9
In conclusion, this report describes a case of a young patient with SLE who developed PRES after receiving the immunosuppressant, mycophenolate mofetil. Although rare, it is important to make physicians aware of this potential side effect of mycophenolate mofetil therapy in the treatment SLE. Neurological symptoms, signs, or changes in the patient’s condition that cannot be explained by lupus, should alert the management team to the possibility of the drug causing PRES, and prompt discontinuation should ensue.
