Abstract
Posterior reversible encephalopathy syndrome is a rare neurological disorder, the diagnosis of which is based on the combination of clinical and radiological findings. It can be associated with many patient-related conditions such as autoimmune disorders or can be provoked by toxins or medication. We report the case of a 70-year-old patient, known for International Federation of Gynecology and Obstetrics stage IVB, high-grade serous ovarian carcinoma, who was diagnosed with a posterior reversible encephalopathy syndrome while on bevacizumab and olaparib maintenance treatment.
Introduction
Posterior reversible encephalopathy syndrome (PRES) is a neurological disorder, related to the inability of posterior circulation to autoregulate and resulting in vasogenic cerebral edema. 1 It was first described in 1996 and has been increasingly recognized thereafter, probably because of the improvement in radiological methods. It is associated with a variation of symptoms, including headache, visual disturbances, altered mental status and seizures. 2 It can be secondary to different conditions such as severe hypertension, eclampsia, cytotoxic drugs or autoimmune disorders. 1 The mechanism of the syndrome is not completely understood; however, it is proposed that results from endothelial dysfunction due to vasospasm, circulating toxins or hypertension induced central hypoperfusion.1,3
Angiogenesis-inhibiting drugs are known to provoke endothelial damage, which can cause PRES. Vascular endothelial growth factor (VEGF) is mitogen and anti-apoptotic for vascular endothelial cells and is a regulator of angiogenesis. 4 The role of angiogenesis being very important in carcinogenesis, many anti-angiogenic agents have been developed, targeting different VEGF receptors (VEGFR-1, VEGFR-2 and VEGFR-3). Bevacizumab, an anti-VEGF monoclonal antibody, has been approved for the treatment of different cancers and is very commonly used in the treatment of epithelial ovarian carcinoma.5–7 It can be associated with some clinically significant side effects, including gastrointestinal perforations and fistulae, wound healing complications, hypertension, hemorrhage, arterial or venous thromboembolic events, renal injury and proteinuria.6,7
The combination of bevacizumab with olaparib has been approved as maintenance treatment for patients with BRCA mutation and/or homologous recombination deficiency (HRD) newly diagnosed high-grade serous ovarian cancer (HGSOC) responding to platinum-based chemotherapy. 7 Bevacizumab monotherapy can be associated in 23%–26% of patients,5–7 with grade 1–3 hypertension irrespective of the dosage, whereas olaparib monotherapy can be associated with hypotension. 8 Despite these findings, the registration trial of bevacizumab–olaparib combination reported an all-grade hypertension occurrence of 46%, with 19% being of grade 3 or more. 7
In our report, we present the case of a PRES in a normotensive patient while on bevacizumab and olaparib maintenance treatment. To our knowledge, there is no other similar case presented in this clinical setting.
Case report
A 70-year-old patient was diagnosed with International Federation of Gynecology and Obstetrics (FIGO) stage IVB HGSOC, after evaluation for persistent abdominal pain, for 3 months. She underwent exploratory laparoscopy with biopsies and mediastinal lymph node sampling based on enlarged lymph nodes on positron emission tomography–computed tomography (PET-CT). She was treated with three cycles of neoadjuvant, platinum-based chemotherapy (3-weekly carboplatinum AUC6 and weekly paclitaxel 80 mg/m2), followed by interval debulking. Chemotherapy was pursued for six cycles, and because of HRD, bevacizumab and olaparib maintenance therapy was initiated for a total of 15 months and 24 months, respectively, according to the PAOLA-1 trial. 7
The maintenance treatment was well tolerated, with no side effects related to bevacizumab, notably hypertension (Figure 1), proteinuria or thromboembolic events. Olaparib-related grade 1 anemia was noted, according to Common Terminology Criteria for Adverse Events (CTCAE), version 5.0, 9 not necessitating dose adaption.
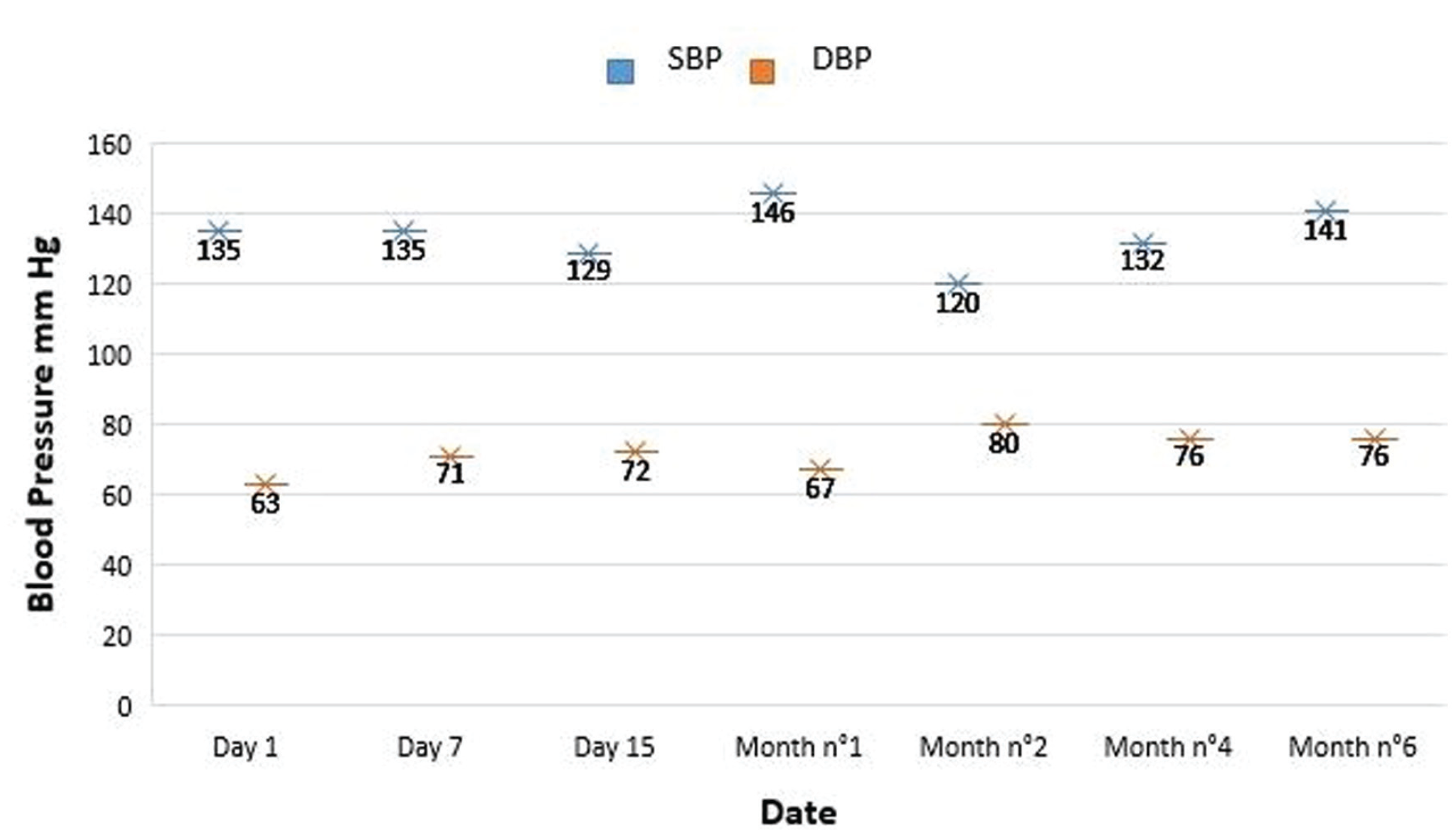
Blood pressure values during bevacizumab treatment.
Sixteen days after the 10th bevacizumab cycle, the patient was admitted to the emergency department for urinary incontinence, grade 2 asthenia, and grade 2 confusion. At the medical evaluation, her Glasgow Coma Score (GCS) 10 was 14 because of acute confusion. The rest of the physical examination was normal. According to her family, there was no seizure, alcohol or drug consumption. The patient was not known to have any previous neurological disorder.
Laboratory examinations were in the normal range, as were the urine culture, blood culture, electrocardiogram and chest X-ray. A cerebral computed tomogram (CT) showed no sign of hemorrhage, neither any abnormality regarding the posterior artery circulation nor stenosis or dilation compared with the anterior circulation. The lumbar puncture was negative for infectious, inflammatory and neoplastic diseases. Empiric antibiotic treatment was initiated (ceftriaxone) and a brain magnetic resonance imaging (MRI) was performed. Axial fluid-attenuated inversion recovery (FLAIR) images showed high signal intensities involving the cortex and the subcortical white matter in the posterior parietal and occipital regions bilaterally without restricted diffusion, consistent with vasogenic edema and highly suggestive of PRES (Figure 2). Differential diagnosis included PRES, reversible cerebral vasoconstriction syndrome (RCVS) 11 or less likely infectious meningitis or meningeal carcinomatosis. Cerebral CT angiography (CTA) did not show any abnormality regarding the size of the posterior circulation arteries, which ruled out RCVS.
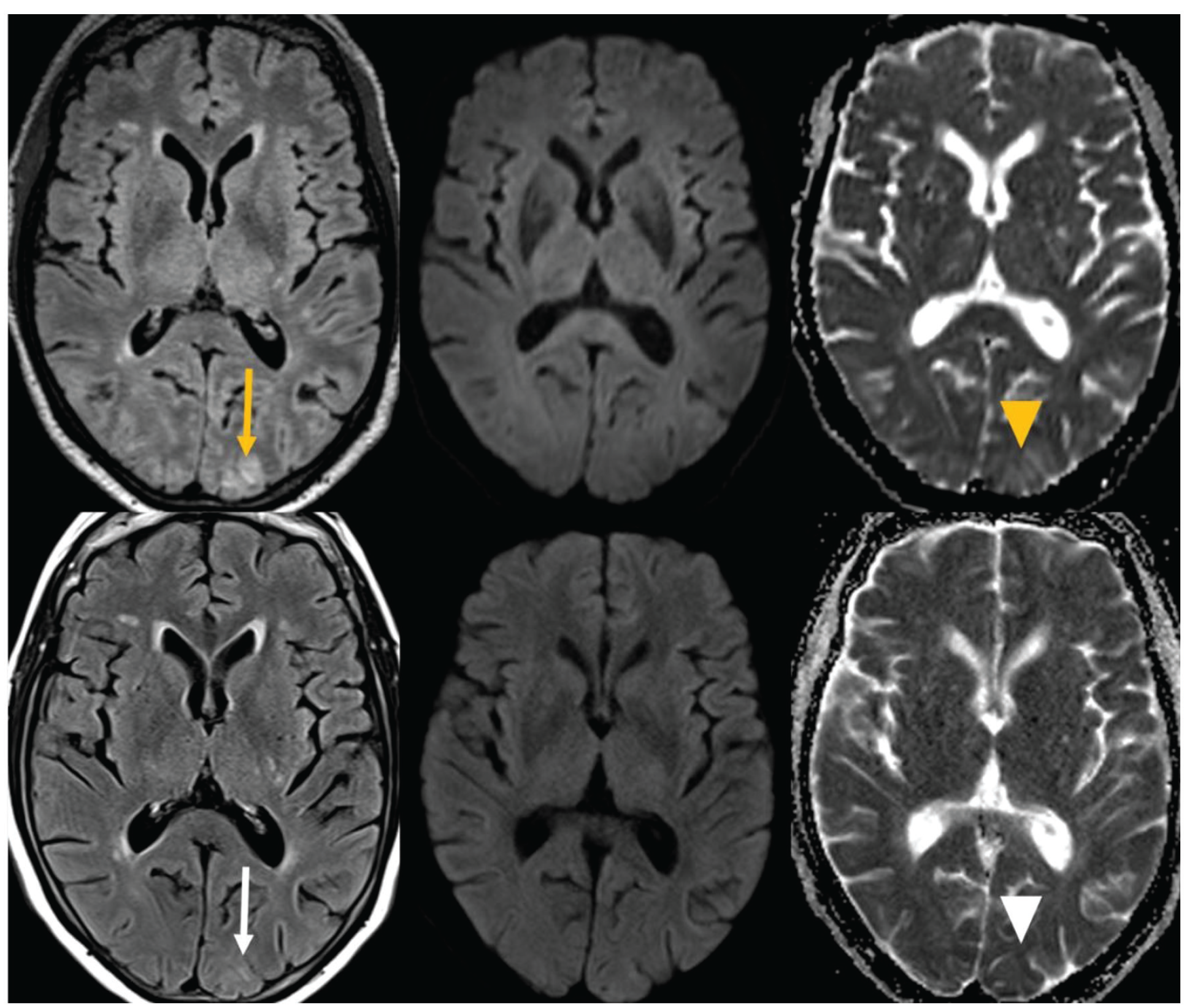
Initial and follow-up MRI.
The MRI detected exclusively vasogenic edema of posterior circulation with neither malignant nor infectious findings. The complete reversibility of the clinical findings confirmed the diagnosis of PRES. The patient’s time to recovery matched the 20-day plasmatic half-life of bevacizumab. During the hospitalization, the patient presented no headache, seizures or visual impairment, and the confusion resolved spontaneously. Thus, PRES was attributed to bevacizumab, which was permanently interrupted while olaparib treatment was pursued.
Almost 2 years since the interruption of bevacizumab, the patient presents complete metabolic and radiologic response, with no evidence of PRES recurrence.
Discussion
The pathophysiology of PRES is not completely elucidated to this day; however, it is proposed that the underlying mechanism is the result of endothelial dysfunction due to vasospasm, circulating toxins or hypertension that leads to cerebral hypoperfusion.1,3 Different conditions have been associated with PRES, varying from pre-existent medical conditions such as autoimmune disorders to cytotoxic and immunosuppressive medications. 1
The diagnosis of PRES can be challenging with non-specific symptoms at presentation and clinical findings mimicking other acute neurological conditions.2,3 PRES can be associated in up to 87% of patients with seizures; 1 encephalopathy develops in 28%-94% of patients 1 and visual symptoms in up to 39%. 12 Clinical symptoms usually resolve after a few days (mean of 5.3 days according to a Mayo clinic report). 12 Underlying conditions include hypertension, renal disease or malignancy. 12 The key to the diagnosis is clinical suspicion.
Several antineoplastic agents, both cytotoxic and targeted agents, are reported to be potentially PRES-inducing. 1 There is no common mechanism described for the antineoplastic agent–induced PRES. In some of the reports found in the literature, the underlying mechanism is unclear, with authors suggesting PRES to be associated with endothelial toxicity related to capillary leak, resulting in vasogenic edema. This was the case of a cisplatin-induced PRES, described by Ito et al. 13 Interestingly, PRES has also been associated with gemcitabine, 14 an antimetabolite with uncommon neurologic toxicities.
On the other hand, the association seems more well-established for anti-angiogenic agents. The importance of angiogenesis in carcinogenesis and tumor proliferation and invasion has been extensively studied. VEGF’s cancer excretion and its role as a mitogen and anti-apoptotic factor for vascular endothelial cells 15 has triggered the development of various VEGF inhibitors including bevacizumab. These molecules reduce neo-angiogenesis and impair tumor growth. Bevacizumab is an anti-VEGF humanized monoclonal antibody used for almost 20 years for the treatment of different solid tumors.
Based on the results of ICON7 and GOG 218, bevacizumab was approved as a frontline maintenance therapy for advanced-stage epithelial ovarian cancer.5,6 In the era of Poly (ADP-ribose) polymerase (PARP) inhibitors (PARPi), the combination of bevacizumab with olaparib was studied as the first-line maintenance treatment by the registered PAOLA-1 trial. 7
Bevacizumab can be related to some clinically significant side effects, including gastrointestinal perforations (0.3%–3%) and fistulae (<1%–1.8%), surgery and wound healing complications (up to 15%), hemorrhage (grades 3–5 from 0.4% to 7%), arterial or venous thromboembolic events (grade 3–5, 5%), hypertension (5%–18%), and renal injury and proteinuria (grade 3, from 0.7% to 7%). PRES was reported in less than 0.5% of patients in different studies, independently of the cancer type treated. Concerning ovarian cancer treatment complications, 16 PRES was reported in 0.2% in GOG 218, 5 0% in ICON7, 6 0.6% in AURELIA, 17 1% in OCEANS 18 and 0% in PAOLA-1. 7 Unfortunately, the details of the associated clinical conditions of these patients are lacking.
When our patient presented the acute confusion, we performed a comprehensive review of the literature. We used the following research engines: PubMed, Google, and Google Scholar. The research key words used were “primary HGSOC,” “Posterior reversible encephalopathy syndrome (PRES),” “bevacizumab” and “olaparib.” The results of the review are summarized in Table 1.19–25
Cross et al. 19 reported two cases presenting with PRES while on bevacizumab. The patients already had hypertension and the bevacizumab treatment was complicated with new-onset grade 3 and grade 2 proteinuria, respectively.
Abbas et al. 20 presented the case of a young patient who developed a generalized tonic–clonic seizure after bevacizumab administration. There was no electrophysiological evidence of epileptic disorder.
Another publication 21 is of a patient with metastatic ovarian cancer, having received multiple chemotherapy regimens and who presented seizures 16 h after the first administration of bevacizumab
The case of a patient with ovarian cancer and presenting with PRES 21 days after bevacizumab administration was published. 22 This patient also had hypertension and visual symptoms.
The fifth patient 23 was diagnosed with PRES while on cisplatin/gemcitabine combination chemotherapy. Paraneoplastic panel was positive for voltage-gated potassium channel antibodies.
Another case 24 describes a patient diagnosed with ovarian cancer, who presented with PRES before having started any oncological systemic treatment. In this case, the trigger point was inaugural hypertension.
Finally, a research performed by Berton et al. 25 aimed to report the real-world experience of bevacizumab as first-line maintenance treatment for ovarian cancer. In all, 468 patients were eligible, 6 of whom developed PRES. There was no further information on patients’ characteristics and comorbidities.
What differs our case from the cases summarized in Table 1 is that our patient had no other bevacizumab-related side effects at the time of PRES diagnosis and that our patient was on bevacizumab–olaparib combination treatment.
To our knowledge, there is no other case of PRES occurring during bevacizumab–olaparib combination treatment, reported in the literature. This could be explained by the fact that this is a novel association with few patients exposed to the combination of bevacizumab–olaparib. Another hypothesis, that could be interesting to explore, is the potentially protective effect of olaparib-induced hypotension 8 counterbalancing the hypertensive effect of bevacizumab.
Conclusion
Prognosis of PRES after bevacizumab is usually favorable, and it has been proposed to rename it as BRES (benign reversible encephalopathy syndrome). As PRES treatment is symptomatic, a high degree of clinical awareness is important for physicians to detect patients who are on treatments associated with PRES. Optimal management is a matter of debate with some reports describing successful bevacizumab re-challenge without PRES recurrence, but we advise caution. Further exploration of the rate of PRES cases on bevacizumb–olaparib combination and the correlation with the patient’s underlying medical conditions and characteristics is needed.
Footnotes
Acknowledgements
We thank the patient for their agreement to use their file information.
Authors’ contributions
Dr Liapi Aikaterini elaborated, drafted, and reviewed the manuscript and analyzed the patient’s data. Drs Atat Chirine and Dunet Vincent drafted and reviewed the manuscript. Dr Sarivalasis Apostolos elaborated, drafted, and analyzed the patient’s data; reviewed the manuscript; and coordinated publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.

