Abstract
Objective
We evaluated the pharmacoeconomics of amlodipine combined with benazepril and hydrochlorothiazide combined with benazepril in the treatment of hypertension using a Markov model to provide an evidence-based reference for clinical drug use.
Methods
In this retrospective study, we constructed two types of Markov model using data from the ACCOMPLISH (Avoiding Cardiovascular Events through Combination Therapy in Patients Living with Systolic Hypertension) trial to dynamically simulate the development of hypertension. The models were subjected to rollback analysis and cohort analysis to obtain the cost and effectiveness of the two drug regimens in preventing stroke and myocardial infarction in hypertensive patients. We conducted sensitivity analysis to determine the stability of the results.
Results
The cost-effectiveness of amlodipine combined with benazepril was 66,196.97 RMB with 6.59 QALYs and that of hydrochlorothiazide combined with benazepril was 74,588.50 RMB with 6.46 QALYs. The incremental cost-effectiveness ratio of hydrochlorothiazide + benazepril was −64,550.23 compared with amlodipine + benazepril. The amlodipine + benazepril regimen was therefore more cost-effective than hydrochlorothiazide combined with benazepril. The sensitivity analysis results showed that the model was robust.
Conclusion
Compared with the hydrochlorothiazide + benazepril treatment regimen, the amlodipine + benazepril regimen showed greater economic benefits.
Introduction
Hypertension is a clinical syndrome characterized by increased systemic arterial blood pressure (systolic blood pressure ≥140 mmHg, diastolic blood pressure ≥90 mmHg), which may cause functional or organic damage to the heart, brain, kidney, and other organs. Hypertension is the most common chronic disease and the most common risk factor for cardiovascular and cerebrovascular diseases globally, such as coronary heart disease and stroke. 1 According to relevant surveys, the global prevalence of hypertension is as high as 26%, making it one of the primary diseases endangering human health. 1 The China Hypertension Survey found that the crude prevalence of hypertension among Chinese individuals aged ≥18 years was 27.9%; the prevalence of hypertension among young people was 5.1% and that among individuals over 75 years old was 59.8%. It is estimated that 245 million Chinese adults aged ≥18 years have hypertension. 2 According to expert forecasts, hypertension is expected to affect more than 1.5 billion people worldwide by 2025, and the number of deaths related to hypertension is expected to increase by 49% each year. 3
Stroke and myocardial infarction are fatal cardiovascular diseases caused by hypertension. The incidence of myocardial infarction in patients with hypertension is significantly increased, and 61% of stroke cases are caused by hypertension. 4 Evidence shows that good control of blood pressure is beneficial for reducing the risk of stroke; 5 therefore, blood pressure control through drug therapy is particularly important for stroke prevention.
At present, treatment and control of hypertension are mainly achieved with drugs. The most commonly used antihypertensive drugs include calcium antagonists and angiotensin-converting enzyme inhibitors. Amlodipine is widely used in the treatment of cardiovascular diseases such as hypertension, angina pectoris, and heart failure. This drug can dilate peripheral arterioles, reduce peripheral resistance (afterload), and effectively reduce myocardial energy consumption and oxygen demand. Additionally, amlodipine can dilate the coronary arteries and coronary arterioles in normal and ischemic areas and increase myocardial oxygen supply in patients with coronary spasm (variant angina pectoris). 6 Hydrochlorothiazide is a diuretic. In treating older patients with isolated systolic hypertension using hydrochlorothiazide, the diuretic effect can eliminate sodium in the body, thereby reducing the hypertensive volume load and improving the patient's hypertension symptoms. 7 Benazepril can inhibit the conversion of angiotensin I to angiotensin II, inhibit the degradation of bradykinin, reduce vascular resistance, and reduce blood pressure. 8 Previous studies have shown that the combination of amlodipine and benazepril has an additive effect in reducing left ventricular hypertrophy and arterial stiffness. 9 Furthermore, combination therapy with benazepril and amlodipine has been shown to be more effective than benazepril and hydrochlorothiazide in reducing cardiovascular events in high-risk patients with stage 2 hypertension and similar blood pressure. 10
Hypertensive patients require long-term treatment with medication, so both the efficacy and cost of antihypertensive drugs should be considered to obtain the best curative effect with the lowest cost and thereby reduce the economic burden on the patient. Previous meta-analyses have shown that compared with the combination hydrochlorothiazide+benazepril,11,12 amlodipine + benazepril is more useful in reducing the cardiovascular risk in hypertensive patients. In this study, we used a Markov model of hypertension based on data from the ACCOMPLISH (Avoiding Cardiovascular Events through Combination Therapy in Patients Living with Systolic Hypertension) trial 13 to explore the effects and economic benefits of amlodipine and hydrochlorothiazide in the prevention of stroke and myocardial infarction in hypertensive patients. We aimed to provide a basis for the selection of optimized drug treatment regimens and pharmacoeconomic evaluation of interventions for Chinese hypertensive patients with poor prognosis as well as to provide a methodological reference.
Methods
Study participants
We searched Medline and PubMed databases using different combinations of the keywords hypertension, stroke, myocardial infarction, angiotensin receptor blockage, calcium channel blockers, diuretics, and randomized controlled trial, as well as their corresponding Medical Subject Headings. We restricted the search to publications in English and to clinical trials published from database inception to December 2020.
The inclusion criteria were patients with high blood pressure and a high risk of cardiovascular events with a history of coronary events, myocardial infarction, revascularization, stroke, impaired kidney function, peripheral artery disease, left ventricular hypertrophy, or diabetes.
Exclusion criteria were secondary hypertension, serious acute or chronic systemic diseases, history of malignancy, animal experiments, non-randomized controlled trials, inability to extract the data or incomplete data, and duplicate publications. We also excluded participants with any other concomitant illness, physical impairment, or mental condition that could interfere with the effective conduct of the study during its likely duration. After completing the database search and screening, a large sample was finally selected as the data source in this study.
The participants in the present study were derived from the study population of the ACCOMPLISH trial. 13 The ACCOMPLISH study was a multicenter, double-blind clinical trial with participants from five countries (United States, Sweden, Norway, Denmark, and Finland) representing 548 centers. The study compared morbidity and mortality related to cardiovascular causes with use of two different combination therapies as initial trial interventions for hypertensive patients at high risk of cardiovascular events. Outcomes in the group receiving the amlodipine + benazepril regimen were compared with those in the group receiving hydrochlorothiazide + benazepril. The baseline characteristics of the included patients are shown in Table 1.
Baseline characteristics of patients in the two groups.
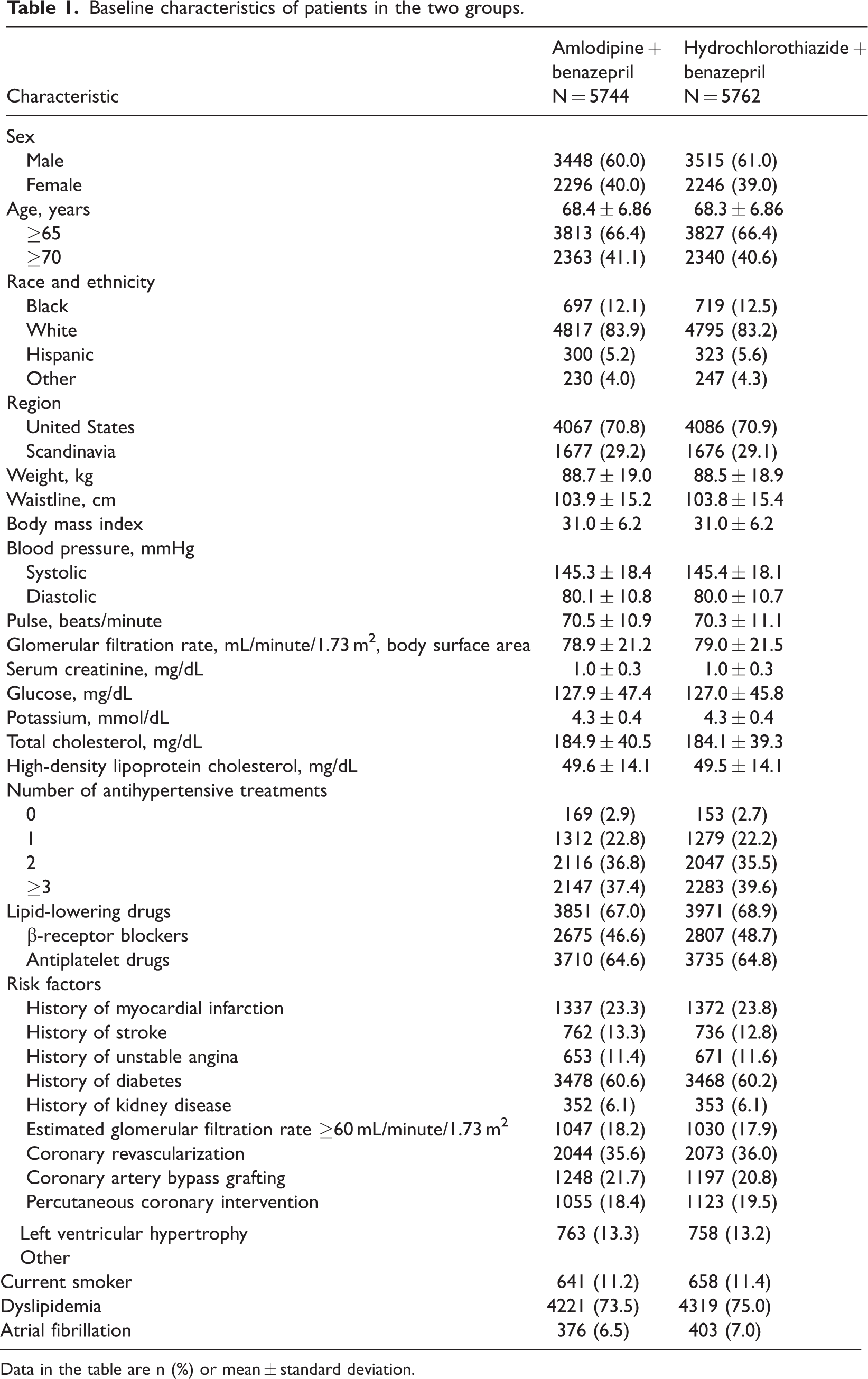
Data in the table are n (%) or mean ± standard deviation.
Markov model
The principle of the Markov model is that a disease is summarized as a state corresponding to its development process according to its impact on health. The disease is simulated according to the probability of transition between states in a specific time, and the resource consumption and health outcome of each state were calculated through multiple cycles to evaluate the final outcome and resource consumption of disease development. 14 The Markov model can well simulate the development of diseases, especially chronic diseases such as hypertension and diabetes. In this study, we adopted a retrospective research approach and constructed a Markov model of hypertension to compare the cost and effectiveness of the combination of benazepril and amlodipine with the combination of benazepril and hydrochlorothiazide in preventing stroke and myocardial infarction among hypertensive patients.
Disease process and cycle
According to the natural history of hypertension, in reference to Markov models for the economic evaluation of hypertension in the existing literature and with availability of the transition probability in the model, the obtained Markov model included the following four health states: hypertensive event-free, nonfatal stroke, nonfatal myocardial infarction, or death, with death as the absorbing state15–18 (Figure 1). These four states cannot exist at the same time in the same patient during one period, which conforms to the principle of the Markov model state setting.

State transition diagram of the hypertension Markov model.
Because hypertension is a chronic disease and it can be many years before related cardiovascular diseases appear, the Markov model cycle in this study was set to 1 year. Moreover, because the research participants were older (average age 68 years), the Markov model was set to 10 cycles for the loop. The Markov model was created using TreeAge Pro 2021 (TreeAge Software LLC, Williamstown, MA, USA), and we performed multiplication analysis, Markov cohort analysis, and sensitivity analysis.
Determination of model parameters
Markov model assumptions
Owing to the complexity of clinical problems in practice, to simplify use of the Markov model, we established the following assumptions: (1) the probability of occurrence of various events in the two patient groups at the end of the trial is the same; (2) the health status transition probability in each cycle is the same as the probability of clinical trial events; (3) because the cost of adverse drug reactions is low, the analysis does not include the occurrence of adverse reactions, nor does it include the reduction in cost or effect size owing to adverse reactions.
Treatment costs
The cost of the hypertensive event-free state was the average annual direct medical cost of the two treatments. According to statistics on the price of current effective drugs in various dosage forms and specifications, obtained from the provincial drug recruitment website, the daily average median price was used as the daily cost of each drug. The treatment costs of nonfatal stroke and myocardial infarction were obtained from published studies using domestic surveys 19 and were converted to 2021 costs according to the Chinese consumer price index. The costs of the two treatments are shown in Table 2.
Status cost of the two schemes.

Health utility value
The key health outcome variable used in this study was quality-adjusted life years (QALYs). According to the Markov model, utility is assigned to the patient’s disease state in each cycle, the time length of each state is calculated cumulatively, and the required QALYs are finally weighted. Because this study did not measure the utility values of various health states required by the model, the health utility values of this model were derived from published studies of the same or similar populations.14,19 The health utility values for each state are shown in Table 3.
Health utility value of each state.

State transition probability
Because there are currently no large-scale clinical trials in China examining the same treatment regimens as those assessed in the present study, we referred to the state transfer probability. 13 The result was obtained using the formula r = −[ln(1−P1)]/t1 and P2 = 1−exp(−rt2), where r is the instantaneous occurrence rate, P1 is the probability of an event occurring within a period of observation, P2 is the transfer rate under one cycle, t1 is the length of the observation period, and t2 is the duration of a single cycle. 20 The calculation results are shown in Tables 4–6.
Incidence of events related to amlodipine and hydrochlorothiazide groups (%).

Annual transfer probability (%) of events related to amlodipine and hydrochlorothiazide treatments.

Results of state transition probability calculation (%).

P, probability; H, hypertension; E, hypertensive event-free; M, nonfatal myocardial infarction; S, nonfatal stroke; D, death.
Evaluation indicators
In this study, QALYs were used as the evaluation index. Incremental cost-effectiveness ratio (ICER) analysis was used as the pharmacoeconomic evaluation standard for the two treatment regimens; there is no such standard in China at present. According to the World Health Organization recommendation, an ICER<1 times gross domestic product (GDP) per capita is extremely cost effective; 1 times GDP per capita <ICER<3 times GDP per capita is cost effective; and ICER>3 times GDP per capita is not cost effective. We selected 1 times GDP per capita (80,976 RMB) in 2021 as the threshold standard in this study. The data were obtained from the 2021 China Statistical Yearbook.
Univariate sensitivity analysis
To investigate the stability of the model, a single-factor sensitivity analysis was performed on the drug cost, discount rate, cost of each state, utility value of each state, and conversion rate of each state for the two treatments. Each cost value was calculated according to the baseline ±25%, the utility value was calculated according to the baseline ±10%, the conversion rate was calculated according to the baseline ±10%, and the discount rate fluctuated from 0% to 7%.
Probabilistic sensitivity analysis
We conducted probabilistic sensitivity analysis via second-order Monte Carlo simulations (simulation 1000 times), with costs following a normal distribution and all probability and rate values a beta distribution. The results of the analysis are depicted using cost-effectiveness acceptability curves and incremental cost-effectiveness scatterplots.
Ethical considerations
All the data used in this study were sourced from published papers, and no identifying patient information was used. This study was exempted by the review committee of the First Affiliated Hospital of Jinan University. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 21
Results
In the present study, we included a total of 11,506 participants in the ACCOMPLISH trial. The average age of participants was 68.4 years, of which 6963 (60.5%) were men. There were no significant differences in baseline characteristics between patients in the two treatment groups, amlodipine +benazepril and hydrochlorothiazide +benazepril (Table 1). 13
Rollback analysis
Multiplication analysis was performed for the amlodipine + benazepril group and the hydrochlorothiazide + benazepril group. The analysis showed that the cost-effectiveness of amlodipine + benazepril was 66,196.97 RMB with 6.59 QALYs gained and that of hydrochlorothiazide + benazepril was 74,588.50 RMB with 6.46 QALYs gained. Compared with the ICER of the amlodipine + benazepril regimen, that of the hydrochlorothiazide +benazepril regimen was −64,550.23. Therefore, the amlodipine + benazepril treatment regimen was more cost-effective than the hydrochlorothiazide + benazepril regimen (Table 7).
Cost-effectiveness analysis of the two treatments.

ICER, Incremental cost-effectiveness ratio; QALY, quality-adjusted life years.
Cohort analysis
Cohort simulations were performed using the Markov models of the two treatment regimens. When the 10th cycle was reached, 38.0% of patients in the model amlodipine treatment cohort died, 4.5% had nonfatal stroke, and 11.6% had nonfatal myocardial infarction. In the simulated hydrochlorothiazide-treated cohort, 41.0% of patients died, 5.1% had nonfatal stroke, and 13.8% of patients had nonfatal myocardial infarction. The results are shown in Figures 2 and 3.

Probability distribution of each state in the Markov model of amlodipine treatment.

Probability distribution of each state in the Markov model of hydrochlorothiazide treatment.
Univariate sensitivity analysis
The model parameters were recalculated at a discount rate of 5%, and the results were consistent with the original model. Single-factor analysis was performed with 80,976 RMB as the threshold, and a tornado map was drawn. Figure 4 shows the factors with a greater impact on the variation range of the ICER. The results of the hurricane plot showed that within the scope of sensitivity analysis, the results of pharmacoeconomic evaluation were not affected. The evaluation results of the Markov model were most sensitive to changes in the transition probability from hypertensive event-free to death for the two treatment regimens.

Hurricane map of single-factor sensitivity analysis. ICER, incremental cost-effectiveness ratio; EV, economic value.
Probabilistic sensitivity analysis
The Monte Carlo sampling method was used to simulate 1000 random samplings. We found that when λ increased from 0, the amlodipine+benazepril group always showed an economic advantage. The cost-effectiveness acceptability curve is shown in Figure 5. When λ = 80769, the probability that amlodipine + benazepril regimen was cost effective was close to 100%. Additionally, Figure 6 depicts the incremental cost-effectiveness scatterplot for 1000 simulations. As seen in the figure, the 95% confidence interval for 1000 simulations was below the threshold standard line, reflecting the economic cost of the amlodipine + benazepril regimen. Therefore, the results of probabilistic sensitivity analysis indicated that the economic advantages of amlodipine + benazepril treatment were relatively stable.
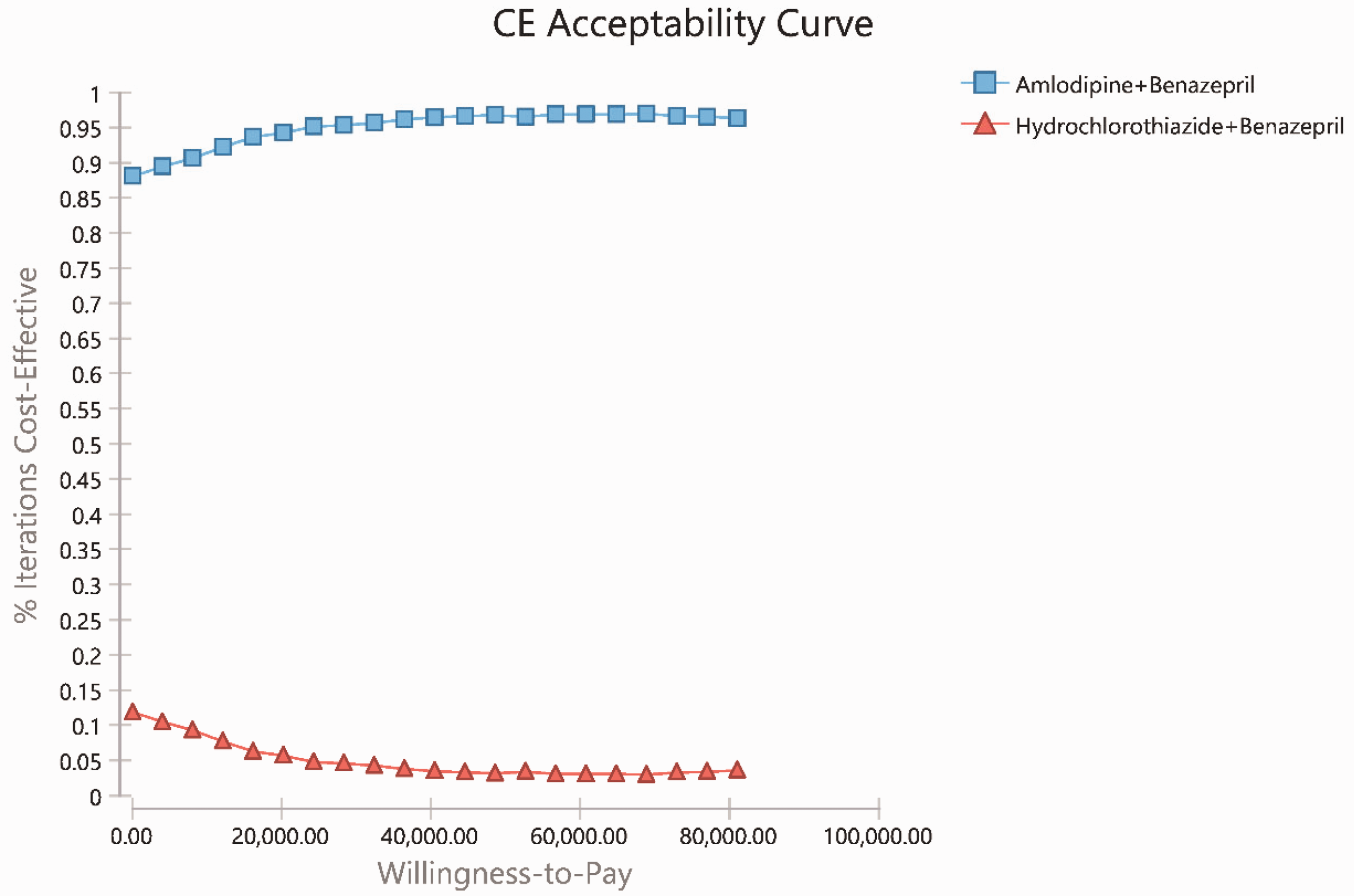
Cost-effectiveness acceptability curve.

Incremental cost-effectiveness scatterplot. WTP, willingness to pay.
Discussion
Hypertension is a leading chronic disease threatening human health and a major risk factor for cardiovascular disease. 22 According to reports, 23 2 million people die as a result of hypertension every year, with 71% of stroke deaths and 53% of deaths from coronary atherosclerotic heart disease being related to hypertension. Good blood pressure control could avert 350,000 to 600,000 deaths between 2016 and 2030. 24 In 2015, the per capita medical expenses of hypertensive patients in China totaled USD 1123.88 per year, and the per capita medical expenses of patients with four kinds of essential hypertension complications were USD 1587.16 per year. 25
For most patients, the antihypertensive effect of antihypertensive drugs is of secondary importance; their main purpose is to reduce the incidence of cardiovascular events and mortality. At present, the drugs commonly used to treat hypertension mainly include calcium antagonists, angiotensin-converting enzyme inhibitors, angiotensin receptor antagonists, diuretics, and beta-blockers. Previous studies have shown that amlodipine+benazepril combination therapy is superior to hydrochlorothiazide+benazepril therapy in reducing the risk of cardiovascular events and death among high-risk patients with hypertension. However, no pharmacoeconomic evaluations of these two treatment regimens have been carried out in China. In the present study, we used a Markov model based on published clinical reports from the ACCOMPLISH trial of hypertension treatment, combined with information on the clinical treatment of hypertension in China and expert advice. The aim of this study was to identify more cost-effective treatment options for hypertension among high-risk patients.
The study results showed that the cumulative cost of the amlodipine+benazepril regimen for the treatment of hypertension was 66,196.97 RMB, with 6.59 QALYs gained; the cumulative cost of the hydrochlorothiazide+benazepril regimen was 74,588.50 RMB, with 6.46 QALYs gained. The incremental cost-effectiveness of the amlodipine group compared with the hydrochlorothiazide group was −64,550.23, indicating that amlodipine+benazepril treatment was more economically beneficial; the death rate was also lower with this regimen. Sensitivity analysis showed that the economic advantage of the amlodipine + benazepril regimen was very robust.
The main limitations of this study are as follows. First, parameters such as efficacy and disease state transition probability were obtained from the limited published literature; extrapolation of the data should be further explored. Second, health utility value is a key parameter in cost–benefit analysis; however, we found no studies on the health utility value for the Chinese population. Therefore, performing utility measurements based on the Chinese population is recommended. Third, we only considered the cost of medicines in direct medical costs and did not consider other costs in this study. Finally, we used models to predict the long-term effects of drugs on treatment costs and patient quality of life. In the prediction process, we simplified complex disease processes to facilitate the analysis; this simplification may introduce certain biases. Therefore, further in-depth and optimized research is needed. However, pharmacoeconomic evaluation mainly focuses on the difference between the cost and the effect of the study object. In the case of similar events in the two treatment regimens, the difference in cost was small and therefore has little impact on the study results. Additionally, we conducted sensitivity analysis on the cost and utility values of the two treatments, separately. The results showed that there was no impact on the study conclusions.
Conclusions
In a comparison, amlodipine+benazepril combination treatment was found to be more economical than treatment with hydrochlorothiazide+benazepril. However, owing to several limitations in this study, further clinical trials based on the Chinese population are warranted to further clarify the findings of this study and identify the development of any complications during the follow-up period.
Footnotes
Acknowledgements
We would like to thank the Guangdong Provincial Key Laboratory of Traditional Chinese Medicine Informatization for the support of this project.
Author contributions
AF and LW conceptualized the research aims, planned the analyses, and guided the literature review.
AF and MZ extracted the data from the PubMed database.
MZ and LL participated in data analysis and interpretation.
JL and AF wrote the first draft of the paper and the other authors provided comments.
All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This study was supported by Guangdong Provincial Key Laboratory of Traditional Chinese Medicine Informatization (no. 2021B1212040007).
