Abstract
Objective
This study aimed to explore the association between lactate dehydrogenase-to-albumin ratio and the severity of coronary artery disease in patients with acute myocardial infarction.
Methods
Patients with acute myocardial infarction were categorized into three groups based on the lactate dehydrogenase-to-albumin ratio tertiles. Demographic characteristics and Gensini scores were collected. Logistic regression and the receiver operating characteristic analyses were performed to evaluate the predictive ability of the lactate dehydrogenase-to-albumin ratio for a high Gensini score (>60). The area under the curve was calculated.
Results
A total of 489 individuals were retrospectively enrolled in this study. Multivariate logistic regression analysis demonstrated that the highest lactate dehydrogenase-to-albumin ratio was an independent risk factor for a high Gensini score (odds ratio, 3.45; 95% confidence interval: 1.37–8.71; p = 0.009). The receiver operating characteristic analysis showed that the lactate dehydrogenase-to-albumin ratio had good predictive ability for identifying a high Gensini score (area under the curve, 0.743; 95% confidence interval: 0.698–0.788).
Conclusions
Lactate dehydrogenase-to-albumin ratio may serve as a novel indicator for predicting the severity of coronary artery disease in patients with acute myocardial infarction.
Introduction
Acute myocardial infarction (AMI), a life-threatening cardiovascular condition caused by acute coronary artery occlusion, remains a major contributor to global morbidity and mortality. The age-standardized incidence rate of AMI is 43.2 cases per 100,000 population, with a mortality rate of 19.3 cases per 100,000 population. 1 Despite advances in diagnostic imaging, the assessment of coronary artery disease severity in patients with AMI before percutaneous coronary intervention remains a challenge in cardiology practice. Therefore, there is an urgent need for reliable biochemical markers with the potential to complement existing tools in predicting the severity of coronary lesions.
Lactate dehydrogenase (LDH) is a key enzyme involved in anaerobic glycolysis and is widely distributed in cardiac tissue. During AMI, necrotic cardiomyocytes release LDH into the peripheral circulation. LDH is recognized as a sensitive indicator of cardiac injury and a predictor of long-term cardiac dysfunction. 2 It also contributes to vascular endothelial dysfunction by mediating oxidative stress, metabolic imbalance, and inflammatory responses. 3 Albumin, the most abundant plasma protein synthesized by the liver, plays a pivotal role in maintaining plasma osmotic pressure, transporting substances, and participating in physiological processes. Recent studies have reported that albumin levels are negatively associated with the risk of coronary calcification regression, and that patients with hypoalbuminemia exhibit poor cardiovascular outcomes.4–6
Although LDH and albumin have been investigated independently, the LDH-to-albumin ratio (LAR) integrates these two distinct pathophysiological pathways. LAR provides a more comprehensive biomarker than either component alone, reflecting the balance between ongoing cellular injury and protective mechanisms. In patients with AMI, a high LAR signifies pronounced tissue damage concurrent with a compromised anti-inflammatory and antioxidant defense system. Recent studies have demonstrated that LAR is a stronger prognostic marker than its individual components in several conditions, including severe infection, 7 pulmonary embolism, 8 coronavirus disease 2019 (COVID-19), 9 and diffuse large B-cell lymphoma. 10 Given its established role in cardiovascular disease, we hypothesized that LAR is associated with the severity of coronary artery disease in patients with AMI. Therefore, this study aimed to explore the association between LAR and the severity of coronary artery disease, as assessed by the Gensini score, in patients with AMI.
Methods
Study population
A total of 662 consecutive patients with AMI admitted to the Cardiovascular Center, Beijing Tongren Hospital, from February 2022 to April 2023 were screened in this retrospective study. All enrolled patients with a diagnosis of AMI underwent coronary angiography and met the criteria outlined in the Fourth Global Definition of Myocardial Infarction. 11 The exclusion criteria were as follows: (a) active infections, defined as the presence of clinical signs or a diagnosis of acute infectious disease at admission; (b) history of malignancy; (c) hematologic disorders; (d) hepatic or renal dysfunction (defined as alanine aminotransferase (ALT) >three times of the upper limit or estimated glomerular filtration rate <30 mL/min/1.73 m2); and (e) missing data on LDH or albumin. After excluding 173 individuals who met the exclusion criteria, a total of 489 individuals were ultimately enrolled in this study. They were categorized into three groups according to LAR tertiles. This single-center, retrospective study was conducted in accordance with the ethical principles of the Declaration of Helsinki 1975, as revised in 2024. Ethical approval was obtained from the Ethics Committee of Beijing Tongren Hospital, Capital Medical University (TREC2022-KY081), and written informed consent was obtained from patients before enrollment. All patient data were deidentified to ensure anonymity and confidentiality. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 12
Data collection
Demographic characteristics (age and sex), comorbidities (hypertension, diabetes, and stroke), medications (aspirin, clopidogrel, angiotensin-converting enzyme inhibitors (ACEI)/angiotensin receptor blockers (ARB), beta blocker, and calcium channel blockers (CCB)), and potential risk factors for coronary artery disease (smoking and alcohol consumption) were obtained from hospital records. Venous blood samples were collected to test for high-sensitive cardiac troponin I (hs-cTnI), ALT, creatine kinase-MB (CK-MB), albumin, LDH, creatinine, uric acid, glucose, potassium, sodium, total cholesterol, triglyceride, low-density lipoprotein-cholesterol, high-density lipoprotein-cholesterol, and glycosylated hemoglobin immediately prior to the initiation of any reperfusion therapy (thrombolysis or percutaneous coronary intervention). All samples were analyzed promptly in the central laboratory of our hospital.
Gensini score
The severity of coronary artery disease was assessed using the Gensini score based on the percentage of stenosis in each major coronary artery and segment. 13 Each coronary lesion exhibiting at least 25% stenosis was assigned a severity score according to predefined criteria: 1 point for 1%–25% stenosis, 2 for 26%–50%, 4 for 51%–75%, 8 for 76%–90%, 16 for 91%–99%, and 32 for total occlusion. To account for the functional significance of different coronary segments, each lesion score was multiplied by a corresponding regional weighting factor. The total Gensini score was calculated as the sum of all weighted lesion scores, with higher values indicating more extensive and severe coronary artery disease. Gensini scores were calculated by two expert cardiologists. Patients were classified as having severe coronary artery disease if they had a Gensini score >60, as reported previously. 14
Statistical analysis
All non-normally distributed continuous variables were presented as median (interquartile range) and compared using one-way analysis of variance (ANOVA) or Kruskal–Wallis test. Categorical variables were presented as frequency (percentage). The Chi-squared or Fisher’s exact test was applied to evaluate the categorical variables. Based on the LAR tertiles on admission, patients were divided into three groups. Logistic regression models were used to estimate the odds ratio (OR) and 95% confidence interval (CI) for the association between LAR and a high Gensini score (>60). Model 1 was unadjusted. Model 2 was adjusted for age, sex, hypertension, diabetes, stroke, smoking, and alcohol consumption. Model 3 included variables from Model 2 and those with a p-value <0.05 in univariate regression analysis. Consequently, Model 3 was adjusted for sex, age, hypertension, diabetes, stroke, smoking, alcohol consumption, clopidogrel, statin, ACEI/ARB, glycosylated hemoglobin, hs-cTnI, CK-MB, ALT, uric acid, glucose, and sodium. Multicollinearity among continuous variables was assessed using the variance inflation factor (VIF), and no significant collinearity was detected (all VIF values <10). Restricted cubic spline (RCS) analysis, adjusted for variables included in Model 3, was used to examine the association between LAR and a high Gensini score. The receiver operating characteristic (ROC) analysis was performed to evaluate the predictive performance of LAR for a high Gensini score. The area under the curve (AUC) was calculated, and DeLong’s test was used to compare AUC differences among LAR, hs-cTnI, and CK-MB. Subgroup analyses were conducted based on hypertension, diabetes, stroke, smoking, and alcohol consumption to assess the predictive value of LAR across different patient subgroups. Analyses were performed using R software (version 4.2.2), and a two-sided p-value <0.05 was considered statistically significant.
Results
Individual characteristics
A total of 489 patients with AMI were enrolled according to the inclusion and exclusion criteria (Figure 1) and were stratified into three groups based on LAR tertiles at admission (T1, ≤5.509; T2, 5.509–11.491; and T3, >11.491; Table 1). The proportion of male patients was comparable in the three groups (T1, 77.9%; T2, 79.8%; and T3, 84.7%; p = 0.279). Patients in the T3 group were younger, had a lower prevalence of hypertension and diabetes, and exhibited higher levels of hs-cTnI, ALT, CK-MB, glucose, total cholesterol, and low-density lipoprotein-cholesterol along with higher Gensini score. The percentage of patients receiving ACEI/ARB and CCB was lower in the T3 group. No significant differences were observed between the groups in terms of smoking or alcohol consumption. Additional parameters, including creatinine, uric acid, high-density lipoprotein-cholesterol, glycosylated hemoglobin, and potassium, were comparable between the groups.

Flowchart of the study patients.
Patient demographics and baseline characteristics.

LAR tertiles: T1 (≤5.509), T2 (5.509–11.491), and T3 (>11.491).
LAR: lactate dehydrogenase-to-albumin ratio.
Association between LAR and a high Gensini score
When analyzed as a continuous variable, univariable logistic regression analysis showed that LAR was significantly associated with a high Gensini score (OR, 1.15; 95% CI: 1.11–1.19; p < 0.001; Table 2). Multivariate logistic regression analysis in Model 3 demonstrated that LAR remained an independent risk factor for a high Gensini score (OR, 1.21; 95% CI: 1.12–1.29; p < 0.001) in patients with AMI. When analyzed as a categorical variable, the highest tertile of LAR was associated with a high Gensini score (OR, 6.83; 95% CI: 4.19–11.13; p < 0.001) in Model 1. After adjustment for sex, age, hypertension, diabetes, stroke, smoking, alcohol consumption, clopidogrel, statin, ACEI/ARB, glycosylated hemoglobin, hs-cTnI, CK-MB, ALT, uric acid, glucose, and sodium, LAR in T3 was significantly correlated with a high Gensini score (OR, 3.45; 95% CI: 1.37–8.71; p = 0.009). The risk of a high Gensini score exhibited an upward tendency as the LAR tertiles increased, both in the unadjusted Model 1 and the fully adjusted Model 3.
Logistic analysis of LAR for a high Gensini score (>60).

LAR tertiles: T1 (≤5.509), T2 (5.509–11.491), and T3 (>11.491). Model 1: unadjusted model. Model 2: adjusted for sex, age, hypertension, diabetes, stroke, smoking, and alcohol consumption. Model 3: adjusted for sex, age, hypertension, diabetes, stroke, smoking, alcohol consumption, clopidogrel, statin, angiotensin-converting enzyme inhibitors or angiotensin receptor blockers, glycosylated hemoglobin, high-sensitive cardiac troponin I, creatine Kinase-MB, alanine aminotransferase, uric acid, glucose, and sodium.
CI: confidence interval; OR: odds ratio; LAR: lactate dehydrogenase-to-albumin ratio; Ref: reference.
RCS analysis
The RCS analysis was performed after adjustment for sex, age, hypertension, diabetes, stroke, smoking, alcohol consumption, clopidogrel, statin, ACEI/ARB, glycosylated hemoglobin, hs-cTnI, CK-MB, ALT, uric acid, glucose, and sodium. The RCS analysis for the severity of coronary artery disease demonstrated a linear relationship between LAR and a higher Gensini score (p for nonlinear = 0.409; Figure 2) in patients with AMI.
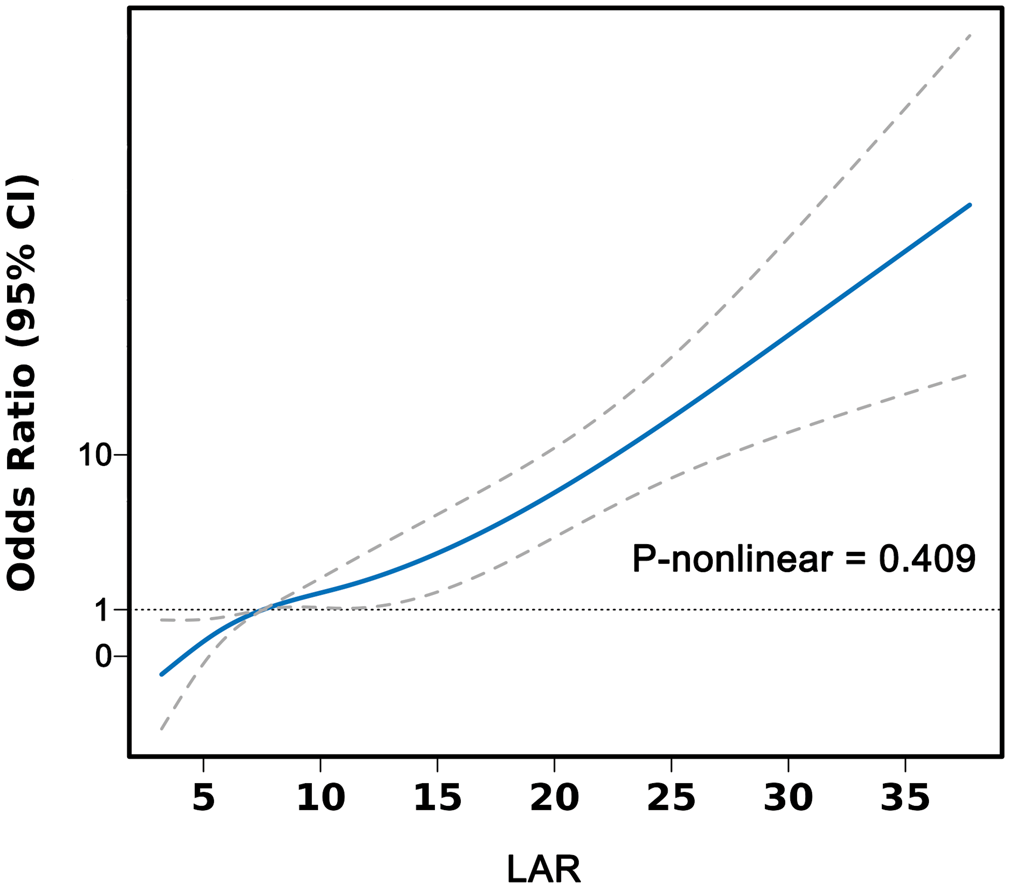
Restricted cubic spline analysis of LAR with a high Gensini score (>60). LAR: lactate dehydrogenase-to-albumin ratio.
ROC curve
The ROC analysis demonstrated that LAR had good discriminatory ability for identifying patients with AMI who had a high Gensini score (AUC, 0.743; 95% CI: 0.698–0.788; Figure 3). The AUC of LAR was significantly higher than those of hs-cTnI (AUC, 0.687; 95% CI: 0.639–0.736; DeLong’s test, p < 0.001) and CK-MB (AUC, 0.662; 95% CI: 0.612–0.713; DeLong’s test, p < 0.001). An LAR value of 9.29 was identified to be the best cutoff for detecting a high Gensini score, yielding a sensitivity of 63.2% and a specificity of 74.0% among patients with AMI undergoing coronary angiography.
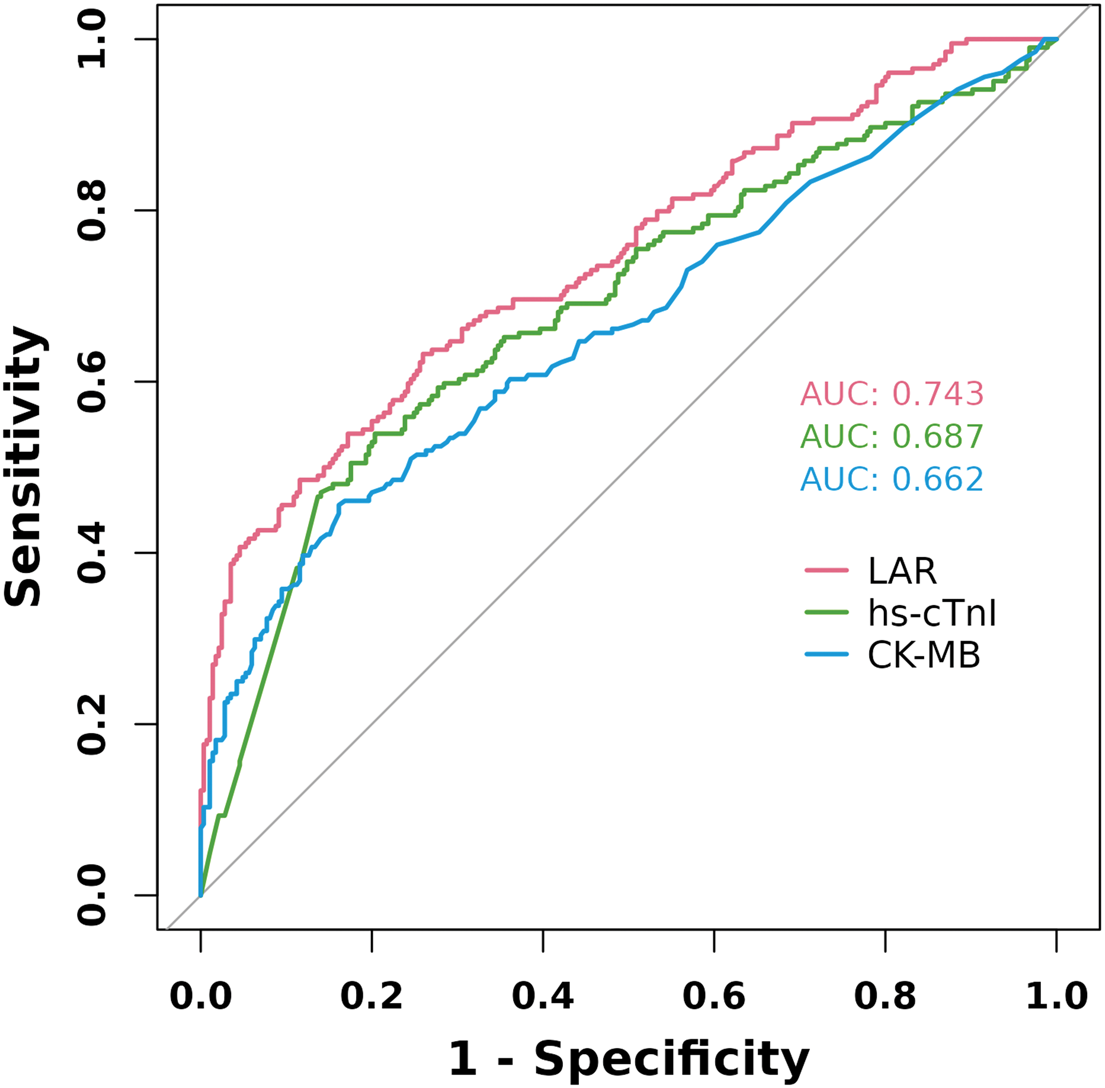
Receiver operating characteristic curve of LAR, CK-MB, and hs-cTnI for predicting a high Gensini score (>60). LAR: lactate dehydrogenase-to-albumin ratio; CK-MB: creatine kinase-MB; hs-cTnI: high-sensitive cardiac troponin I.
Subgroup analysis
Subgroup analyses stratified by hypertension, diabetes, stroke, smoking, and alcohol consumption were performed after adjusting for variables included in Model 3 to assess whether LAR remained an independent indicator across different individuals (Figure 4). The ORs for a high Gensini score were significant in subgroups of patients without a history of stroke or alcohol consumption. The association between LAR and a high Gensini score remained significant regardless of hypertension, diabetes, and smoking. Interaction analyses revealed no significant difference in the presence of hypertension, diabetes, stroke, or smoking. However, the relationship between LAR and a high Gensini score was influenced by alcohol consumption.

Forest plots of stratified analyses of LAR and a high Gensini score (>60). LAR: lactate dehydrogenase-to-albumin ratio.
Discussion
This retrospective study analyzed 489 patients with AMI who underwent coronary angiography and demonstrated that LAR was independently associated with a high Gensini score after adjustment for potential confounding factors. The RCS analysis of coronary artery disease severity revealed a linear relationship between LAR and a higher Gensini score. Moreover, this association remained consistent across subgroups stratified by hypertension, diabetes, and smoking, supporting the robustness of our conclusions. The ROC curve analysis demonstrated that LAR had predictive value for a high Gensini score, with an AUC higher than those of CK-MB and hs-cTnI. An LAR value of 9.29 was identified to be the best cutoff for detecting a high Gensini score, with a sensitivity of 63.2% and a specificity of 74.0% in patients with AMI. Therefore, LAR may serve as an easily accessible and useful biomarker for assessing the severity of coronary artery disease in patients with AMI.
LDH is an enzyme present in various organ systems, and its serum levels are elevated in multiple disorders. Previous studies have demonstrated the prognostic value of LDH in patients with tumors and metabolic syndrome.15,16 A recent study involving 893 patients with AMI reported that baseline LDH levels were positively associated with the development of cardiac insufficiency. Additionally, LDH was superior to troponin I (TnI) or creatine kinase (CK) in predicting long-term cardiac structure and function in these patients. 2 LDH, a key enzyme in anaerobic glycolysis, serves as a sensitive marker of myocardial injury, with serum levels rising in proportion to the extent of cardiac damage. It catalyzes the interconversion of lactate and pyruvate, with elevated levels indicating cellular hypoxia and oxidative stress. The blocking of the pyruvate–lactate axis helps balance glycolysis and oxidative phosphorylation, which protects against vascular endothelial dysfunction in hypoxic conditions. 3 Apart from being directly involved in the energy metabolism in cardiomyocytes, LDH is also related to inflammation during AMI. The balance between proinflammatory and anti-inflammatory immune cells is essential for cardiomyocyte survival in AMI. LDH-mediated aerobic glycolysis in activated T cells promotes interferon-gamma (IFN-γ) expression. Ablation of LDH in T cells protects against immunopathology caused by the excessive IFN-γ production. 17 These findings confirm the role of LDH in dysregulated immune system activation and the acceleration of adverse cardiac remodeling. 2
Hypoalbuminemia has been shown to be significantly associated with poor prognosis in patients with cardiac arrest, sepsis, and diabetes.18–20 Recent clinical studies have demonstrated a relationship between hypoalbuminemia and the severity of coronary artery disease. A study comprising 12,904 individuals who underwent coronary computed tomography angiography reported that lower albumin levels were identified as a risk factor for increased coronary calcification burden and adverse cardiac and cerebrovascular events. 4 Additionally, patients with decreased albumin levels exhibited poor prognosis and a higher incidence of cardiothromboembolic events. 21 Albumin exerts anti-inflammatory and antioxidant effects, contributing to prevent the progression of various cardiovascular diseases. 22 At the molecular level, its anti-inflammatory action involves the suppression of tumor necrosis factor alpha–mediated vascular cell adhesion molecule-1 expression and subsequent leukocyte–endothelial interactions, thereby interrupting early inflammatory cascades and atherosclerotic progression. 23 The antioxidant capacity of albumin primarily derives from its metal ion–chelating properties, enabling the neutralization of harmful hydroxyl radicals generated via transition metal–catalyzed reactions with hydrogen peroxide. 24 Furthermore, clinical investigations have revealed that hypoalbuminemia is associated with reduced thromboxane B2 suppression, highlighting the critical role of albumin in platelet inhibition. 20 The anticoagulant function of albumin further contributes to its comprehensive cardiovascular protective effects. 25
The LAR has demonstrated good predictive value for prognosis across various diseases. In a study of 581 patients with pulmonary embolism, increased LAR was associated with a lower 30-day survival rate. 8 Among patients with COVID-19, an LAR of 10.48 predicted mortality with a sensitivity of 73.5% and a specificity of 73.7%. 9 In patients with severe infection, LAR was independently associated with in-hospital mortality. 7 A recent study further revealed that LAR was associated with the prognosis of diffuse large B-cell lymphoma and demonstrated a better prognostic value than LDH or albumin alone. 10 However, the association between LAR and the severity of coronary artery disease in patients with AMI has not yet been comprehensively evaluated. In this context, our findings suggest that LAR serves as an easily accessible indicator reflecting the severity of coronary artery disease in patients with AMI. The superior predictive performance of LAR over traditional cardiac biomarkers such as hs-cTnI and CK-MB may be attributed to its ability to integrate two opposing pathophysiological pathways. Although hs-cTnI and CK-MB are highly specific markers for cardiomyocyte necrosis, they offer limited insight into the concomitant systemic inflammatory and oxidative stress responses. In contrast, elevated LAR reflects a state of heightened metabolic stress and cellular damage accompanied by a reduced systemic capacity to mitigate inflammation and oxidative injury. Such an imbalance may be particularly detrimental in the context of AMI. Clinically, the accessibility and low cost of LDH and albumin measurements make LAR a promising tool for rapid risk stratification upon hospital admission. Future research should explore whether LAR provides incremental predictive value when incorporated into established risk models that include clinical variables and other biomarkers.
Several limitations of this study should be acknowledged. First, as a single-center, retrospective study, the results may be influenced by selection bias, potentially limiting generalizability to broader populations. Additionally, the small sample size was relatively modest. Second, the observational nature of our analysis precludes definitive causal inferences between LAR and coronary artery disease severity. Although we adjusted for a range of potential confounders, residual confounding from unmeasured or imperfectly measured factors cannot be ruled out. Biomarkers of inflammation and liver function, such as C-reactive protein and aspartate aminotransferase, were not included in the analysis due to a high rate of missing data (>20%). Third, LAR was assessed only at admission, which may not reflect dynamic changes in biomarker levels over time. Finally, a key limitation of our study is the lack of data on in-hospital complications and short-term clinical outcomes. Therefore, the ability of LAR to predict clinical events remains undefined.
Conclusion
This study demonstrated that LAR may serve as a novel indicator for predicting the severity of coronary artery disease (Gensini score > 60) in patients with AMI, even after adjustment for confounding factors. The ROC curve demonstrated good discriminative ability, further supporting the potential role of LAR in AMI. Future multicenter prospective studies are warranted to validate the value of LAR in patients with AMI.
Footnotes
Acknowledgments
None.
Author contributions
Caixia Guo and Haosheng Wu conceptualized this study. Haosheng Wu, Yu Xin, and Xue Jiang performed the research. Haosheng Wu, Zhenyan Wu, and Yu Xin wrote the manuscript. Yu Xin analyzed the data. All the authors revised the manuscript.
Consent for publication
Not applicable.
Data availability
Data are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that they have no conflict of interest.
Funding
This work was supported by the National Natural Science Foundation of China (grant number 82171808 and 82200369), Beijing Natural Science Foundation (grant number 7232022), and the priming scientific research foundation for the junior researcher in Beijing Tongren Hospital, Capital Medical University (grant number 2021-YJJ-ZZL-001 and 2022-YJJ-ZZL-015).
