Abstract
Porphyromonas gingivalis, a gram-negative oral anaerobe among more than more than 500 bacterial species that colonizing the oral cavity, is involved in the pathogenesis and prototypic polybacterial consortium of periodontitis. It is mainly found in oral infections and rarely present in other organ diseases. Here, we describe a 43-year-old man with underlying diabetes who developed hematogenous disseminated severe pneumonia after P. gingivalis had invaded the blood. Next-generation sequencing of early alveolar lavage fluid and blood samples confirmed the diagnosis. The patient’s lung infection improved after targeted antimicrobial treatment. He was successfully weaned from ventilatory support and transferred to the general ward. This case illustrates bacterial entry into the bloodstream of a patient with diabetes who had periodontal disease but did not maintain oral hygiene, leading to severe pneumonia. Periodontal disease is often ignored by the public, and it is difficult for critical care physicians to link severe pneumonia with periodontal disease. Thus, this case represents an important warning to critical care clinicians.
Keywords
Introduction
Porphyromonas gingivalis, a non-glycolytic gram-negative anaerobic bacillus, has been extensively characterized as an important periodontal pathogen. Its surface fimbriae play a key role in the host immune response and the pathogenesis of periodontitis. 1 Although P. gingivalis can colonize host epithelium without causing obvious disease, multiple studies involving various host cell types have revealed that it is highly pathogenic and exhibits diverse virulence factors, which can disrupt host defense mechanisms and disrupt structural components of periodontal tissue. 2 Thus far, P. gingivalis has been associated with rheumatoid arthritis, dementia, cardiovascular disease, ulcerative colitis, and some cancers.3,4 These reports indicate that its effects are not limited to periodontal disease. Moreover, P. gingivalis can cause lesions in other organs through various pathways. These extraoral manifestations are relatively rare and often overlooked. Importantly, there have been no reports of severe pneumonia caused by P. gingivalis. Here, we describe a patient who developed hematogenous disseminated severe pneumonia after P. gingivalis had invaded the blood.
Case presentation
A 43-year-old man was admitted to the hospital with a 1-week history of back pain, chest tightness, shortness of breath, and mild limitation of movement; there was no apparent cause. His symptoms had worsened 1 day before admission, and he began to experience dizziness and weakness. The pain was temporarily alleviated by taking analgesic medications. The patient presented to a local hospital for treatment. Cervicothoracic magnetic resonance imaging revealed no abnormalities. The patient’s symptoms of back pain, chest tightness, and shortness of breath continued to worsen; therefore, he was transferred to the emergency department of our hospital. Routine blood test results in the emergency department were: blood glucose, 9.6 mmol/L (high); potassium, 2.69 mmol/L (low); high-sensitivity C-reactive protein, 279.49 mg/L (extremely high); white blood cell count, 15.4 × 109/L (high); and total platelet count, 18 × 109/L (extremely low). Concurrent blood gas analysis revealed the following findings: pH, 7.4 (normal); partial pressure of carbon dioxide, 34.6 mmHg (slightly low); partial pressure of oxygen, 58.7 mmHg (low); base excess, 0.4 mmol/L (normal); oxygen saturation, 91.3% (low); and lactate, 2.0 mmol/L (normal). In the emergency department, non-contrast enhanced chest computed tomography (CT) and CT pulmonary angiography showed multiple zones of inflammation in both lungs, small nodules in both lungs, and inflammatory granulomas (Figure 1a). CT angiography showed no abnormalities. The patient was admitted to the intensive care unit after he had received symptomatic treatment in the emergency department. A review of the patient’s medical history revealed that he had been diagnosed with hypertension 4 years prior; under long-term oral medication, his blood pressure was well-controlled. The patient had been diagnosed with diabetes mellitus 1 month prior to admission; oral metformin was not effectively controlling blood glucose levels. During hospitalization, the patient’s cardiac ultrasound, myocardial markers, troponin, electrocardiogram, and abdominal ultrasound findings improved. Considering the lack of obvious abnormalities, a diagnosis of infective endocarditis was excluded; moreover, the liver and spleen were not involved. On the first day of hospital admission (in the intensive care unit), the patient received ventilatory support using a non-invasive ventilator. Chest imaging showed scattered nodules in both lungs, which were distinct from the patchy shadows and interstitial exudation features in patients with conventional bacterial pneumonia and viral pneumonia; therefore, diagnoses of fungal infection and Staphylococcus aureus infection could not be excluded. In terms of antimicrobial therapies, voriconazole was used to target possible fungal etiologies, vancomycin was used to target possible bacterial etiologies involving cocci, and ceftriaxone was used to target possible bacterial etiologies involving bacilli. The disease rapidly progressed, and the patient became dependent on non-invasive ventilatory support because of severe respiratory distress. On the second day of hospital admission, a tracheal cannula was connected to the non-invasive ventilator to provide additional ventilatory support. After the airway had been opened, fiberoptic bronchoscopy was performed to collect bronchoalveolar lavage fluid samples; these fluid samples and concurrent blood samples were subjected to metagenomic next-generation sequencing (mNGS). Fiberoptic bronchoscopy showed a smooth bronchial wall without obvious hyperemia or hydrops. On the third day of hospital admission, the patient began to experience high fever. His oxygenation index worsened, and the partial pressure of oxygen did not exceed 60 mmHg (low) when pure oxygen was supplied by the ventilator. The patient’s blood pressure decreased, and his platelet count substantially decreased to 8 × 109/L (extremely low). Repeat CT (Figure 1b) showed that chest lesions had progressed, such that obvious fusion was evident. There were no abnormalities on brain or maxillofacial CT. The results of mNGS showed that P. gingivalis was present in blood and bronchoalveolar lavage fluid; the quantity of bacterial DNA was higher in blood. Therefore, the patient was diagnosed with severe pneumonia caused by hematogenous dissemination of P. gingivalis to the lungs. Further assays showed no evidence of viruses or Mycobacterium tuberculosis. Considering that P. gingivalis is an anaerobic gram-negative bacterium with a clear mechanism of pathogenesis, the antimicrobial therapy regimen was adjusted to levonidazole phosphate to target its anaerobe features and imipenem to target its bacillus features. Notably, Klebsiella pneumoniae and members of Enterobacteriaceae are common in patients with diabetes patients; therefore, imipenem was used to target bacillus features, rather than anaerobe features. No fungal spores were detected in sputum smear samples, and there were no abnormalities in the β-1,3-D-glucan and galactomannan tests. Voriconazole was discontinued because there was no evidence of fungal infection. Additional discussion with the patient revealed no history of obvious gingival swelling or pain. A dentist was subsequently consulted; retraction with an oral appliance revealed a small amount of pus in the dentition, along with periodontitis involving the right molars. The patient was tentatively diagnosed with pericoronitis. Based on guidance from the Department of Stomatology, intensive oral care was provided; chlorhexidine and metronidazole were administered during drainage. The patient underwent intermittent ventilation in the prone position, as well as simultaneous bronchoscopic sputum suction. Subsequently, his oxygenation index improved. A repeat routine blood test showed a gradual decrease in white blood cell count and progressive increase in the platelet count. The patient displayed stable breathing; thus, the tracheal cannula and ventilator were removed after intermittent spontaneous breathing exercises on the ninth day of hospital admission. Repeat dynamic chest CT showed obvious resolution of pulmonary inflammation (Figure 1c). Subsequent chest CT showed bilateral, small, sponge-like changes in some pulmonary nodules. Changes on chest CT during the course of disease are depicted in Figure 1a–f. Trends in white blood cell count, procalcitonin level, and oxygenation index during hospitalization are shown in Figure 2a–d. After intensive treatment, the patient’s condition had stabilized. Repeat chest CT on the 12th day of hospital admission (Figure 1d) revealed further resolution of pulmonary inflammation. The patient was transferred to the general ward for further treatment by the Department of Respiratory Medicine. Repeat chest CT on the 21st day (Figure 1e) and 28th day (Figure 1f) of hospital admission showed that pulmonary lesions had largely disappeared. The patient’s length of stay was approximately 30 days. He was discharged from the hospital after his disease condition had considerably improved. After discharge, he no longer experienced chest tightness, shortness of breath, or back pain. The patient and his family were satisfied with the diagnosis and treatment processes, and they actively cooperated with the treatment.
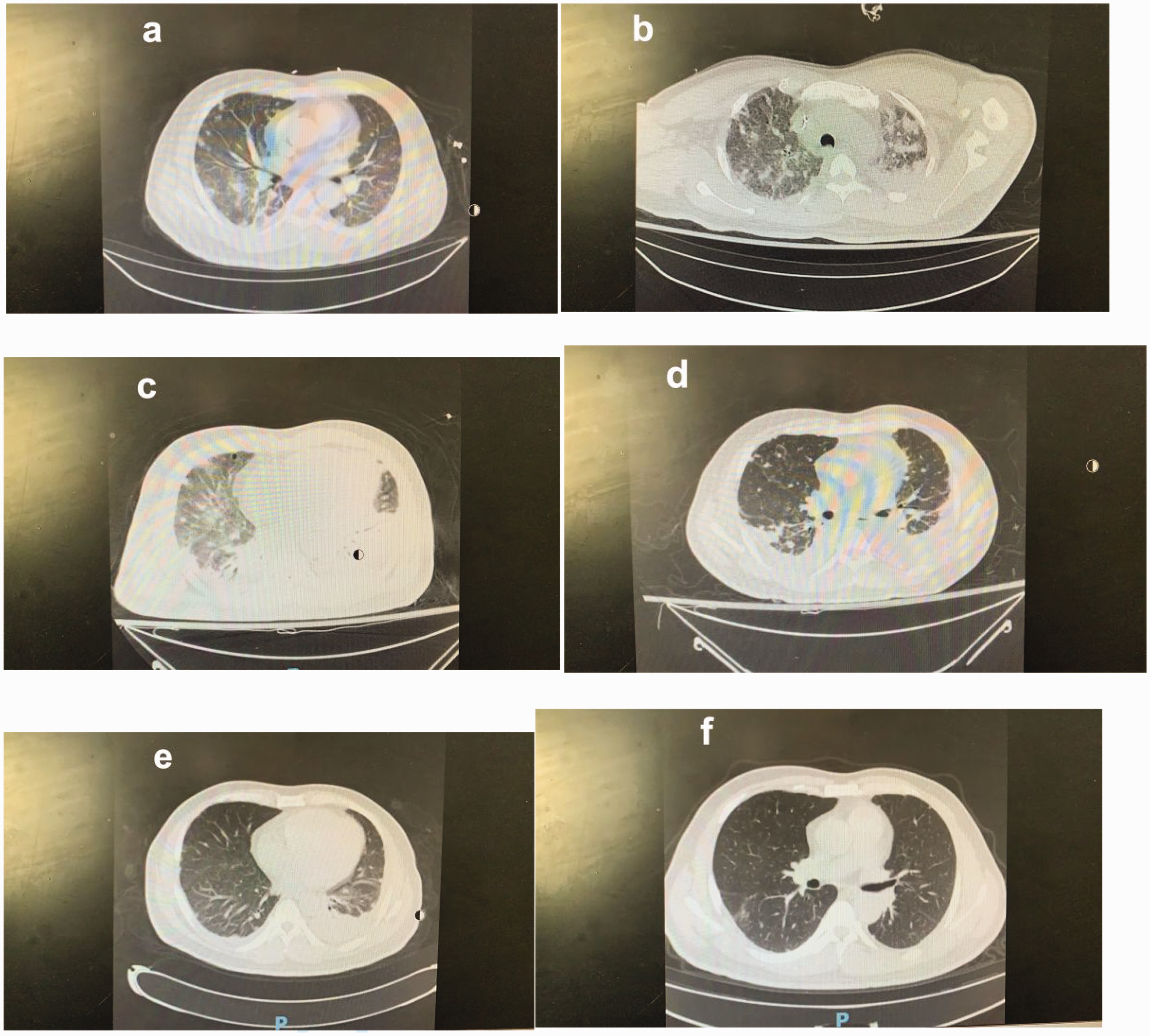
(a) In the emergency department, chest computed tomography (admission day 1) revealed scattered nodular changes in both lungs. (b) On admission day 3, the patient’s condition substantially worsened; he developed high fever and shock. (c) After treatment on admission day 8, the oxygenation index considerably improved. Tracheal intubation was discontinued on the next day (admission day 9), and persistent imaging factors were considered and (d) Admission day 12. (e) Admission day 21 and (f) Admission day 28.

(a) White blood cell count (×109/L) gradually decreased to the normal range during treatment. (b) Platelet count (×109/L) gradually increased to the normal range during treatment. (c) Procalcitonin level (µg/L) gradually decreased to the normal range during treatment and (d) Oxygenation index (unitless) gradually increased to the normal range during treatment.
Discussion
During growth, P. gingivalis obtains metabolic energy from protein decomposition products, heme, and vitamin K. 5 P. gingivalis bacteremia is uncommon. When bacteremia occurs in healthy people, it generally constitutes a transient, clinically benign infection that does not cause further sequelae. However, when immune response mechanisms fail or become overwhelmed, bacteremia can progress to a blood infection with various clinical manifestations. 6 Thus far, very few cases of spleen abscess, liver abscess, post-chest wall abscess empyema, and P. gingivalis bacteremia-induced brain abscess have been reported.7–10 There is evidence that P. gingivalis can cause bronchial and pharyngeal epithelial cells to produce proinflammatory cytokines in the absence of acute infection, leading to aspiration pneumonia. 11 However, there has been no evidence that P. gingivalis causes hematogenous disseminated severe pneumonia. There is extensive evidence that oral microbiota and community-acquired pneumonia pathogens are common, in contrast to the conventional clinical perspective; thus, such pathogens are often ignored. Standard bacterial culture conditions generally are insufficient for detection in these unusual cases; our patient was successfully diagnosed through mNGS of early clinical samples. His condition rapidly progressed; oxygenation index worsening was accompanied by clinically significant deterioration on repeat chest CT. Moreover, the lung lesions comprised scattered nodular changes involving the inner, middle, and outer bands, with gradual fusion; these findings considerably differ from aspiration pneumonia-related lung abscesses that often constitute a single type relevant to anatomical structure and body position, mostly involving the right lung. Typical X-ray signs for aspiration pneumonia-related lung abscesses include large, dense, and fuzzy inflammatory infiltrating shadows with indistinct edges and a large purulent cavity. Initially, we suspected hematogenous severe pneumonia. The patient had underlying diabetes mellitus and immune impairment, which initially led us to suspect a liver abscess-related hematogenous disseminated lung abscess. To investigate this, we conducted simultaneous abdominal CT and ultrasound examinations. However, the findings ruled out our initial hypothesis. Thus, we immediately collected alveolar lavage fluid for mNGS analysis. Furthermore, we considered the characteristics of typical hematogenous lung abscesses, which are predominantly caused by gram-positive cocci such as S. aureus and Streptococcus spp. 12 Early clinical manifestations include obvious symptoms of sepsis (e.g., chills and fever). Our patient exhibited a small volume of mildly purulent and bloody sputum. Radiographic findings comprised multiple small areas of inflammatory shadows on the periphery of both lungs, along with spherical and elliptical dense nodules displaying neat edges of varying size. Some of these patches and nodules evolved into liquid abscesses or tension cavities. Post-inflammatory absorption can lead to focal fibrosis or the formation of small air sacs. Although these absorption manifestations were not evident in our patient, considering his rapid deterioration, we initially adopted a treatment plan targeting S. aureus and Streptococcus pneumoniae. Surprisingly, the mNGS results indicated that both blood and alveolar lavage fluid contained P. gingivalis, which provided guidance for targeted treatment. From a broader treatment perspective, the absence of early etiological insights often results in a clinical focus on cocci-induced infection. However, failure to promptly manage pulmonary inflammation can lead to a progressive increase in dead space, providing an anaerobic environment that accelerates pathogen growth. This situation can become a vicious cycle with a high risk of mortality. A key difference between this case and typical cases of hematogenous lung abscess was the absence of obvious sepsis symptoms during early stages of disease; this observation may provide valuable insights for clinical diagnosis and treatment. Fortunately, early mNGS analysis identified the pathogen associated with severe pneumonia and bacteremia; prompt initiation of appropriate antimicrobial agents inhibited bacterial growth. Additionally, we used fiberoptic bronchoscopy to aspirate sputum and implemented prone position ventilation; these interventions effectively reduced dead space in the lungs and hindered pathogen survival. Accordingly, we were able to prevent disease progression and ensure patient safety. Currently, treatments for P. gingivalis infection mainly involve conventional antimicrobial agents; however, there are increasing concerns regarding drug resistance, drug toxicity, and long treatment cycles. In one study, 24.6%, 21.3%, and 24.6% of P. gingivalis isolates were resistant to amoxicillin, azithromycin, and metronidazole, respectively. 13 In this case, a combination treatment involving levonidazole phosphate and imipenem rapidly improved the patient’s condition, enabling successful weaning from mechanical ventilation and transfer to the general ward. Thus, we infer that levonidazole plus imipenem is an effective treatment for P. gingivalis-induced hematogenous disseminated severe pneumonia. However, there is an aspect of treatment that warrants further consideration. In the management of gram-negative bacilli, it may be more prudent to begin with lower-level antimicrobial agents (e.g., second- and third-generation cephalosporins), rather than imipenem. This approach may help to limit impacts on antibiotic resistance.
Conclusion
Overall, effective pathogen identification is essential for treatment. In the early stages of the patient’s illness, we were unaware that the primary cause was an oral pathogen. This case highlights the clinical significance of severe pneumonia arising from oral pathogens. Our report should be carefully considered because it offers valuable insights that can assist future diagnoses of infections involving uncommon bacterial pathogens. Especially when severe pneumonia in patients with immune abnormalities (e.g., diabetes, tumor, or autoimmune disease) cannot be explained by common pathogenic bacteria, there is a need to consider infection by oral pathogens. The prompt implementation of guidance from mNGS analysis was the key to successful treatment in this case. This outcome underscores the utility of mNGS analysis in pathogen identification. 14 Therefore, when an infection involving multiple microorganisms is suspected, mNGS analysis should be prioritized.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231213760 - Supplemental material for Porphyromonas gingivalis-induced hematogenous disseminated severe pneumonia: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605231213760 for Porphyromonas gingivalis-induced hematogenous disseminated severe pneumonia: a case report by Zhou Yuanjun, Cai Yan, Zhang Qingyan and Xiong Feng in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
Identifying information was removed prior to publication; therefore, written patient consent was not required to publish the case. This report was written in compliance with the CARE guidelines.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
