Abstract
Non-tuberculous mycobacterial spondylitis is a rare spinal infection, especially among patients without acquired immunodeficiency syndrome or other immune impairments. Because of its rarity and non-specific clinical manifestations, diagnosis is often delayed or missed. Here, we present a case of Mycobacterium chelonae spondylitis in an immunocompetent patient and review the relevant literature.
Keywords
Introduction
Non-tuberculous mycobacterial (NTM) spondylitis is a rare spinal infection. Disseminated NTM infection occurs mainly in immunodeficient patients and rarely in those with normal immune function. 1 The purpose of this report is to describe a case of Mycobacterium chelonae spondylitis caused by bloodstream dissemination in an immunocompetent patient. The study was approved by the Ethics Committee of the Dingli Clinical Institute of Wenzhou Medical University (Wenzhou Central Hospital) (approval number L 2021-01-050) and was published with written informed consent of the patient. The reporting of this study conforms to CARE guidelines. 2
Case report
In February 2020, a healthy 48-year-old woman employed in housework began to complain of low back pain that worsened in June and July. She was not taking any medications and had no history of conditions such as autoimmune diseases, diabetes, pulmonary disease, tumors, trauma, or acupuncture. Initially, her low back pain occurred after carrying heavy objects and was paroxysmal. At that time, a local doctor performed lumbar computed tomography (CT). The results were normal and the patient was diagnosed with lumbar muscle strain and treated with analgesics. Her pain persisted. When her low back pain worsened with no improvement in a supine position, magnetic resonance imaging was performed and showed multiple nodular abnormal signals in the thoracolumbar spine and accessories, lumbar 3-sacral 1 disc degeneration, and swelling (Figure 1). Positron emission CT revealed bone lesions with increased fluorodeoxyglucose (FDG) uptake in the sternum, left clavicle, multiple ribs, and multiple vertebral bodies of the spine and pelvis along with increased density and some bone destruction. The maximum standardized uptake value (SUV) was approximately 7.0 (Figure 2a). Multiple hilar and mediastinal lymph nodes showed increased FDG uptake. The maximum SUV was approximately 7.3 (Figure 2b).

Magnetic resonance imaging scan (T2-weighted image) showing multiple nodular abnormal signals of the thoracolumbar spine and accessories (see blue arrows), lumbar 3-sacral 1 disc degeneration, and swelling.

(a) Positron emission computed tomography (PECT) showing bone lesions in the sternum, left clavicle, multiple ribs, and multiple vertebral bodies of the spine and pelvis with increased fludeoxyglucose (FDG) uptake, increased density, and some bone destruction (see arrows). The maximum standardized uptake value (SUV) was about 7.0. (b) PECT findings of multiple hilar and mediastinal lymph nodes showing increased FGD uptake (see blue arrows). The maximum SUV was about 7.3.
Clinicians initially considered these lesions to represent multiple bone metastases from a distant cancer. However, bone marrow biopsy showed extremely reduced bone marrow hyperplasia, with no immature cells, megakaryocytes, or tumor cells. In August, the patient was transferred to another hospital. Bone radionuclide scanning showed abnormal radioactivity concentration in the sternum, right 5-7 anterior ribs, multiple thoracic vertebrae, and multiple lumbar vertebrae (Figure 3). Clinicians still considered bone metastases as the most probable diagnosis. The patient visited our hospital for further examination.

Bone scintigraphy showing sites of abnormal radioactivity concentration in the sternum, right 5-7 anterior ribs, multiple thoracic vertebrae, and lumbar vertebrae.
Upon admission, her vital signs were normal. There was no enlargement of the superficial lymph nodes. The results of cardiopulmonary and abdominal examinations were normal. Normal lordotic curvature of the lumbar spine and lumbar percussion pain was observed. There was no paravertebral tenderness. Straight leg raise tests were bilaterally negative, muscle strength and sensation in both legs were normal, and bilateral knee reflexes were brisk.
Initial laboratory data, including liver and kidney function, coagulation function, routine urine analysis, blood parameters, Epstein–Barr virus antibody titers, fungal D-glucan, Aspergillus galactomannans, levels of tumor markers, immunoglobulin G4, procalcitonin, blood cytomegalovirus DNA, anti-cyclic citrullinated peptide antibody, antinuclear antibody, and anti-neutrophil cytoplasmic antibody were all normal. Human immunodeficiency virus (HIV) and hepatitis virus tests were negative. Her white blood cell count was 9.8 × 109/L (normal range: 4–9 × 109/L), her platelet count was 509 × 109/L (normal range: 100–500 × 109/L), her erythrocyte sedimentation rate was 103 mm/hour (normal range: 0–20 mm/hour), and her C-reactive protein level was 107.2 mg/L (normal range: 0–8 mg/L). Her κ light chain level was 16.60 g/L (normal range: 5.98–13.29 g/L), her λ light chain level was 13.40 g/L (normal range: 2.80–6.65 g/L), and her κ/λ ratio was 1.24 (normal range: 1.47–2.95). Sputum mycobacterial cultures were negative. We neglected to check the patient's blood culture, mainly because she had no fever.
Three-dimensional lumbar reconstruction CT showed lumbar curvature, local disconnection of the right L3 lamina, increased density of the L3–L5 vertebral bodies, and depression of the upper edge of the L3 vertebral body (Figure 4). Three-dimensional CT reconstruction of the thoracic vertebrae showed increased density in the T1, T3, T4, T10, and T11 vertebrae and depression of the upper edges of the T1 and T3 vertebrae (Figure 5). There were a few fibrous foci in both lungs on thoracic CT, and the mediastinal lymph nodes were mildly enlarged. With the consent of the patient, a surgeon performed vertebral bone marrow biopsies of the T11 and T4 vertebrae. Pathology showed no tumor cells but partial braided bone tissue, bone marrow fibrosis, plasma cell infiltration, and multinucleated giant cell reactions (Figure 6a). Immunohistochemical staining was positive for CD38 and CD138 but negative for p53, myeloperoxidase, cyclin D1, CD3, and CD20; the κ/λ ratio was 1, with a Ki-67 index of <1%. Acid fast, periodic acid-Schiff (PAS), and silver hexamine staining were all negative. The bone marrow biopsy was negative following acid-fast staining (Figure 6b), hexamine silver staining (Figure 6c), and PAS staining (Figure 6d). Finally, blood metagenomic next-generation sequencing (mNGS) revealed the presence of M. chelonae, suggesting disseminated M. chelonae infection and M. chelonae spondylitis.

Three-dimensional reconstruction of lumbar CT shows local non-union of the right lamina of L3 (blue arrow) and increased density of the L3–5 vertebral bodies with depression of the upper edge of the vertebral body (black arrow).
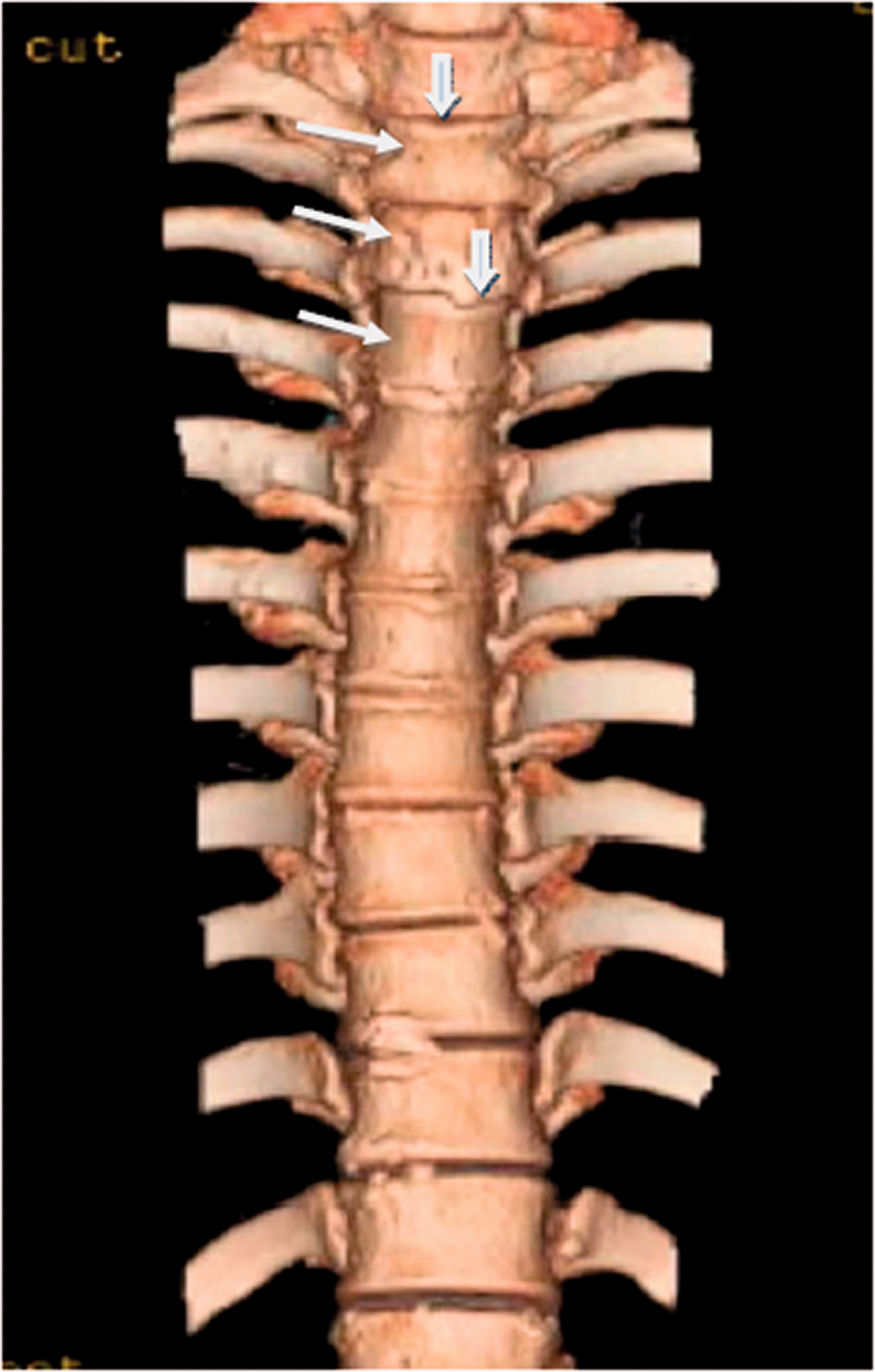
Three-dimensional thoracic computed tomography reconstruction showing increased density in the T1, T2, T3, T10, and T11 vertebrae (white arrow) and depression of the upper edges of the T1 and T3 vertebrae (blue arrow).

(a) Hematoxylin and eosin-stained 4 × 10 vertebral body biopsy showing some woven bone tissue (black arrow), bone marrow fibrosis (blue arrow), focal plasma cell infiltration (green arrow), and a multinucleated giant cell reaction. (b) Acid-fast staining of bone marrow biopsy was negative. (c) Hexamine silver staining of bone marrow biopsy was negative. (d) Periodic acid-Schiff staining of bone marrow biopsy was negative.
After consideration, the patient agreed to receive anti-NTM therapy (300 mg rifampicin once daily (qd), 500 mg levofloxacin qd by intravenous drip, 750 mg ethambutol od in the morning, and 400 mg amikacin qd by intravenous drip. One week later, her symptoms of low back pain were relieved. This effective treatment further confirmed the diagnosis of M. chelonae spondylitis. After communication with the patient, this anti-NTM treatment regimen was further adjusted for 1 month. Levofloxacin was stopped and switched to moxifloxacin (400 mg qd by intravenous drip). The amikacin dose was increased to 600 mg qd by intravenous drip, and linezolid injection 600 mg qd by intravenous drip was started. Two weeks later, the patient experienced no adverse reactions and she was discharged for outpatient treatment. The post-discharge medication plan was 300 mg rifabutin capsules qd perorally (po), 500 mg clarithromycin sustained-release tablets twice daily po, 400 mg moxifloxacin tablets qd po, 400 mg amikacin qd intramuscularly (im), and 600 mg linezolid tablets qd po.
One month later, the patient's low back pain was almost completely relieved, but her liver function was decreased. After 2 weeks of drug withdrawal, her liver function returned to normal. Her treatment regimen was adjusted to 500 mg azithromycin tablets qd po, 600 mg linezolid tablets qd po, and 400 mg amikacin qd im. Afterwards, the patient did not experience any adverse effects. After continuous treatment for 5 months, the patient had no low back pain, and improvement was apparent on spinal CT (Figure 7). At present, the patient is still receiving the anti-NTM treatment regimen described above.
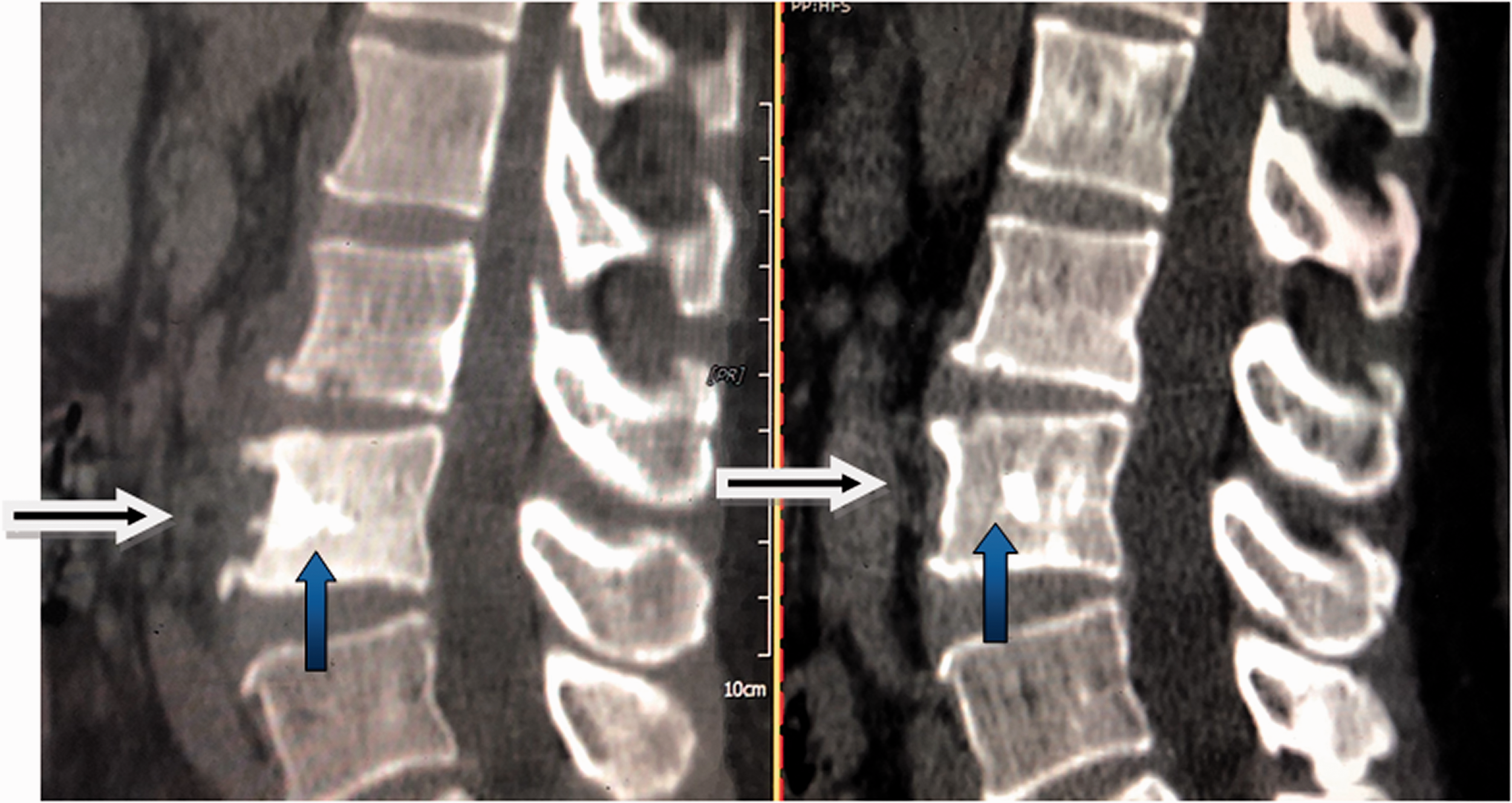
Lumbar computed tomography showing reduction of lesions and vertebral bone destruction (blue arrows) and reduced surrounding tissue swelling (black arrows).
Discussion
NTM are a large class of mycobacteria excluding members of the Mycobacterium tuberculosis complex (M. tuberculosis, M. bovis, M. africanum, M. microti, M. caprae, M. pinnipedii, M. suricattae, and M. mungi) and Mycobacterium leprae. NTM infection has a variety of clinical manifestations including lymphadenitis as well as pulmonary, skin, soft tissue, bone, joint, and disseminated infections.1,3,4 In 1956, Weed et al. reported the first case of NTM spinal osteomyelitis. 5 The most common organism responsible for NTM spondylitis is M. avium, followed by M. abscessus, M. xenopi, and M. kansasii.6–8 Kim et al. reviewed the English-language literature from 1961 to 2014 and identified 69 cases of NTM spondylitis; 10 patients were HIV positive, and M. chelonae was identified as the causative agent in only four patients. 7 As of 15 May 2020, Smimmo et al. systematically reviewed 91 cases of NTM spinal osteomyelitis, including HIV positive patients and patients with limited immune function. Only seven patients had M. cheloniae spondylitis. 8
M. chelonae is a fast-growing atypical mycobacterium belonging to Runyon’s fourth class. It was first isolated from a sea turtle by Friedman in 1903, and is widely distributed in soil, water, and dust particles. The organism can be cultured at 25 to 40°C and forms unpigmented colonies with rough or smooth surfaces.9–11 M. chelonae primarily causes lymph node, skin, and soft tissue infections, as well as bone and joint inflammation and disseminated disease in immunosuppressed patients. Less commonly, M. chelonae can cause lung disease. Other rarer infections include keratitis, thyroiditis, endocarditis, and spondylitis.12–14
NTM spondylitis is a chronic disease with non-specific clinical symptoms and signs. Patients with NTM spondylitis are prone to immunosuppression, but the condition also affects immunocompetent patients. Common causes of NTM spondylitis in immunocompetent patients are previous spinal surgery or epidural injection, previous tuberculosis infection, diabetes mellitus, and intravenous drug abuse.7,8 For patients without these risk factors, M. chelonae may be transmitted through the blood to spinal bone marrow. According to the literature, the average age of patients with NTM spondylitis ranges from 43 to 56, with no significant gender preference.6–8,15 The most common symptoms were low back pain, followed by lower limb weakness, neurologic deficits, fever, and weight loss.7,8 The most common involved regions are the lumbar spine, followed by the thoracic spine, the cervical spine, and the sacral vertebrae. Involvement of the whole spine is rare, with visible paravertebral abscess.8,15,16 In this case report, the patient had chronic onset of NTM spondylitis and her only symptom was low back pain. On imaging, multiple parts of the spine (thoracic, lumbar) and multiple segments of the vertebrae had bone destruction. The clinical and imaging features of NTM spondylitis require further study.
The diagnosis of NTM spinal infection requires bone marrow biopsy specimens and Mycobacterium culture. This is an invasive examination that requires a long culture period of about 4 to 8 weeks.1,6,8
The diagnostic techniques used for NTM isolation and culture and strain identification are varied. Traditional methods, such as liquid and solid medium culture, cannot identify NTM at the strain level and require long culture times. High pressure liquid chromatography is a practical and rapid diagnostic technique for slow-growing NTM, but cannot identify strains of fast-growing mycobacteria. Molecular biology technologies, including nucleic acid probe hybridization, probe analysis, polymerase chain reaction, and gene chips, are recommended to identify NTM.1,7 However, these methods cannot identify fungal and multiple microbial infections, and sensitivity is only approximately 70%. 17 Because the virulence of NTM is relatively weak, diagnosis of mycobacterial infection sometimes requires repeated sample culture. Blood cultures are often negative and up to 6 weeks of incubation are required to obtain positive cultures. 8 Righter et al. reported a case of M. chelonae septicemia in a patient with aplastic anemia; the patient was diagnosed only after eight blood cultures. 18 mNGS, as a new generation of sequencing technology, does not rely on microbial culture and can quickly and accurately detect pathogens. Compared with traditional NTM culture methods, mNGS can shorten the diagnosis time by at least 2 weeks, allowing treatment to be initiated earlier and improving prognosis. However, the high cost of this technique limits its large-scale clinical application. At present, there is limited information on the diagnostic value of mNGS for spondylitis. Some scholars have analyzed the application of mNGS in bone and joint infection. Zhao et al. found that only 48.4% of bone and joint specimens containing pathogens were detected by routine culture, while the detection rate of mNGS was 100%. 19 Huang et al. found that the sensitivity of mNGS in detecting bone and joint pathogens was significantly better than that of traditional culture (89.7% vs. 61.5%), and that the detection rate of NTM using mNGS was higher than that using traditional culture methods. 20 In the case presented here, several months elapsed from onset to the diagnosis of the disease. The pathogen was identified by blood mNGS, leading to a diagnosis and treatment that relieved the patient's pain. Regretfully, no blood culture and bone marrow biopsy specimens were sent for mycobacterial culture.
There are no consensus guidelines concerning the treatment of NTM spondylitis. Once diagnosed, antimicrobial therapy is often required to eradicate infection. Isolates of M. chelonae are susceptible or partially susceptible to tobramycin, clarithromycin, linezolid, imipenem, amikacin, clofazimine, doxycycline, and ciprofloxacin. 1 The optimal duration of treatment has not been determined. For bone infection, some scholars suggest a treatment duration of 6 months, while others believe a duration of more than 12 months is needed.1,6 Some patients need surgery in addition to antimicrobial therapy, but there is no unified opinion on surgical indicators. Some studies have suggested that patients with extensive disease, abscess formation, or difficult drug treatment should consider surgery.1,6,8 With timely treatment, the outcome for most patients is good, but a small number of patients will have complications including death. Smimmo et al. analyzed 80 cases of NTM spondylitis and found that 79.9% if patients were cured or improved, 10% were unresponsive to treatment, and 10% died; 33% experienced complications including neurological sequelae and spinal deformity. 8 In the case presented, the patient has a wide range of lesions and adopted a conservative treatment plan. At present, antimicrobial therapy appears to be effective.
Conclusion
Because NTM spondylitis is a rare and challenging diagnosis, morbidity may be underestimated. Whether in immunocompromised or immunocompetent patients, the etiology of spinal lesions routinely includes investigations of tuberculosis, tumors, bacteria, and fungi. However, NTM should be routinely investigated as well. In patients with complex features, mNGS may provide another choice for diagnosis and enable timely treatment. Further studies of the diagnostic value of mNGS in spondylitis are required.
Ethics statement
This study was approved by the Ethics Committee of the Dingli Clinical Institute of Wenzhou Medical University (Wenzhou Central Hospital) (approval number L 2021-01-050). The authors have no ethical conflicts to disclose.
Supplemental Material
sj-zip-1-imr-10.1177_03000605211047766 - Supplemental material for Spondylitis following bloodstream dissemination of Mycobacterium chelonae disseminated in an immunocompetent patient: a case report and literature review
Supplemental material, sj-zip-1-imr-10.1177_03000605211047766 for Spondylitis following bloodstream dissemination of Mycobacterium chelonae disseminated in an immunocompetent patient: a case report and literature review by Shuangxia Dong, Zhou Lin, Shenghao Wu and Leyi Cai in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contributions
S.X.D. and Z.L. designed research; Z.L. and W.S.H. performed research; Z.L. and L.Y.C. analyzed data; and S.X.D. and Z.L. wrote the paper. All authors read and approved the final manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
