Abstract
Objective
Hyporesponsiveness to erythropoiesis-stimulating agents (ESAs) is a prevalent problem affecting hemodialysis (HD) patients. Roxadustat is an oral hypoxia-inducible factor prolyl hydroxylase inhibitor that stimulates erythropoiesis and regulates iron metabolism. We explored the ability of roxadustat to increase the hemoglobin (Hb) concentration in ESA-hyporesponsive patients undergoing HD and assessed its effect on iron metabolism and inflammation.
Methods
This prospective study included 30 patients with ESA-hyporesponsive anemia who had been undergoing stable dialysis. All patients received roxadustat three times per week for 24 weeks. The primary endpoint was the mean change in Hb from baseline to the average level over weeks 20 to 24. Iron metabolism markers, C-reactive protein, interleukin (IL)-6, and safety were also assessed.
Results
At week 24, roxadustat treatment resulted in a 2.5 ± 1.3 g/dL increase in the Hb level. In total, 28 of 30 patients (93.3%) had an Hb level increase of more than 1.0 g/dL from baseline. Seventeen patients (56.7%) met the endpoint, with a mean Hb level of at least 10.0 g/dL. Iron metabolism and IL-6 levels were also improved.
Conclusions
Oral roxadustat is effective for ESA-hyporesponsive anemia in maintenance HD patients and may also improve iron metabolism and IL-6 levels.
Keywords
Introduction
Renal anemia is the most common complication in patients with chronic kidney disease (CKD). The incidence of anemia increases gradually with declining renal function, and the rate is as high as 98.2% in dialysis patients. 1 A relative erythropoietin (EPO) deficiency is the main cause of renal anemia. Most patients with CKD receive erythropoiesis-stimulating agents (ESAs); 2 however, hyporesponsiveness to ESAs occurs in 5% to 15% of patients who are undergoing hemodialysis (HD). 3 Hyporesponsiveness to ESAs, which is the inability to achieve or maintain target hemoglobin (Hb) levels despite higher than usual ESA doses, 4 is associated with increased hospitalizations and mortality 5 and frequent blood transfusions.
The pathogenesis of ESA hyporesponsiveness in CKD is complex and involves iron deficiency (absolute or functional), inflammation,
6
poor nutritional status, vitamin deficiency, inadequate dialysis, oxidative stress, hyperparathyroidism, and medications such as angiotensin-converting enzyme inhibitors and angiotensin receptor blockers.7–8 Among these factors, iron deficiency has been cited as the most common cause of hyporesponsiveness in maintenance hemodialysis (MHD) patients.
7
In a prospective study (TRAP study), Kuragano T
Hypoxia-inducible factors (HIFs) are transcriptional activators of hypoxia-inducible genes that play a key role in the physiologic adaptation to hypoxia by regulating the expression of multiple genes, including EPO and others involved in iron metabolism and erythropoiesis. 10 HIF expression is controlled by prolyl-4-hydroxylase domain proteins. Roxadustat is an oral HIF prolyl hydroxylase inhibitor (HIF-PHI), which is the latest medication used to treat anemia in CKD patients. As HIF stabilizers, HIF-PHIs can stimulate the production of endogenous EPO and increase iron mobilization and utilization by mimicking the natural response to hypoxia. 11 Several studies have reported that roxadustat was effective in correcting anemia in dialysis patients.12–14 However, limited research reports on roxadustat treatment for ESA-hyporesponsive-related anemia have been published.
This study aimed to explore the ability of roxadustat to increase the Hb concentration over a 24-week treatment period in ESA-hyporesponsive patients undergoing HD and assess the effect of roxadustat on iron metabolism and utilization and its influence on inflammation.
Materials and Methods
Study design and population
This prospective, single-center study explored the efficacy of roxadustat in 30 ESA-hyporesponsive anemia patients receiving chronic HD from August 2022 to February 2023. The reporting of this study conforms to the STROBE guidelines 15 and was approved by the Ethics Committee of the Second Xiangya Hospital, Central South University in August 2022 (2022C130). All patients signed informed consent forms after obtaining detailed information about the study protocol. We have de-identified all patient details.
The inclusion criteria were as follows: (1) patients aged 18 to 75 years with a body weight of 45 to 100 kg; (2) those receiving stable HD for at least 12 months with a single-pool Kt/Vurea ≥ 1.2; (3) Hb > 5.5 g/dL and <10.0 g/dL; (4) ESA-hyporesponsiveness with an EPO-α dose ≥12,000 IU/week in the 12 weeks before screening. Patients were required to be iron replete. Those with a TSAT level of <20% and ferritin level <100 ng/mL were excluded from the study. The exclusion criteria also included patients with myocardial infarction, acute coronary syndrome, stroke, class III/IV heart failure within 8 weeks, uncontrolled hypertension, inflammatory disease, hematological disease, and a history of cancer.
The study consisted of a 4-week recombinant human erythropoietin (rhEPO) run-in period followed by a 20-week roxadustat treatment period and a 4-week follow-up period. The starting dosage of roxadustat was either 100 mg (in patients weighing <60 kg) or 120 mg (in patients weighing ≥60 kg) three times per week. The dose was adjusted every 4 weeks on the basis of a predefined dose adjustment algorithm (according to the drug instructions) to achieve an Hb level within the target range of 10 to 12 g/dL. However, a dose decrease was permitted if the Hb level increased by >2.0 g/dL in 2 weeks in a patient whose Hb level was >9.0 g/dL. Dose increases and reductions were preset according to dose steps: 20, 40, 50, 70, 100, 120, 150, and 200 mg. The use of oral iron and intravenous iron therapy was prohibited.
Adverse events were monitored and reported by patients and treating physicians throughout the whole study.
Endpoints
The primary endpoints included the number and percentage of patients with at least a 1.0-g/dL increase in Hb from baseline at week 20 and the proportion of patients with an Hb level of at least 10.0 g/dL at week 20. The secondary endpoints included the mean change in the Hb level from baseline to the average level during weeks 20 to 24; changes in iron metabolism markers, such as hepcidin, ferritin, transferrin, and %TSAT, in serum from baseline; and evaluation of the inflammatory status of C-reactive protein (CRP) and interleukin (IL)-6 levels at week 20.
Laboratory measurements
Routine blood samples for the measurement of Hb, CRP, IL-6, and iron parameter levels were obtained at every visit and assayed at the central laboratory of the Second Xiangya Hospital. Serum hepcidin-25 was detected using an enzyme-linked immunosorbent assay (Mlbio, Shanghai, China). We calculated the ESA resistance index at baseline before the trial, which was defined as the weekly ESA dose per kg of body weight divided by the Hb level (g/dL).
Statistical analyses
Statistical analyses were performed using SPSS software (version 20.0; IBM Corp., Armonk, NY, USA). Data are expressed as the mean ± standard deviation for continuous variables and as percentages for categorical variables. The number and percentage of patients meeting the primary endpoint are summarized along with the 95% confidence intervals for the proportion of patients who met the primary endpoint definition. The secondary endpoints of observed change from baseline with at least a 1.0-g/dL increase in Hb and the Hb level over time are summarized. In addition, the endpoint Hb level is summarized using the average Hb values from weeks 20 to 24. Changes in iron parameters, CRP, and IL-6 from baseline are summarized by visit. Differences within changes from baseline were analyzed using paired t-tests. All adverse events during the study were recorded. A two-sided significance level of 0.05 was used to determine statistical significance.
Results
Baseline characteristics of the patients
Thirty patients, 17 men and 13 women, meeting the criteria were enrolled from August 2019 to February 2020. All patients completed the 24-week study. The average age of participants was 54.4 years (Table 1). All patients were treated with high-flux dialysis with a frequency of 2.5 to 3 times/week and using 1.5 mmol/L calcium dialysate for HD. Most patients (43%) had chronic nephritis. The mean baseline Hb level was 7.2 g/dL, and the mean dosage of prior rhEPO was 13,167 IU per week. Seven patients (23.3%) had a TSAT value (the percentage of transferrin, an iron-carrier protein occupied by iron) of more than 50%, indicating iron overload. Ten patients (33.3%) had a CRP level above the upper limit of the normal range. Seven patients underwent bone marrow biopsy because of long-term anemia, and the results were normal.
Baseline characteristics of patients in the study (n = 30).

SD, standard deviation; No., number; ERI, erythropoietin resistance index; Hb, hemoglobin; CRP, C-reactive protein; max, maximum; min, minimum; rhEPO, recombinant human erythropoietin; TSAT, transferrin saturation; IL-6, interleukin-6.
Hb levels
At weeks 20 to 24, roxadustat treatment resulted in an increase in the Hb level of 2.5 ± 1.3 g/dL from baseline. In total, 28 of 30 patients (93.3%) had an Hb level increase of more than 1.0 g/dL from baseline. Seventeen patients (56.7%) met the endpoint, with a mean Hb level of at least 10.0 g/dL. Two patients had an Hb level increase of only 0.2 to 0.3 g/dL, and their Hb levels remained between 6.4 and 7.0 g/dL throughout the entire study, even though the roxadustat dose was increased to 120 mg. Four patients had Hb increases of >2.0 g/dL within 4 weeks, and the roxadustat dose was reduced to 70 mg at weeks 4 and 8. The change in Hb level was observed at visits during the treatment period (Figure 1). Hb increases were greater at week 4 (1.2 g/dL) and week 8 (0.86 g/dL) than at the last visit.
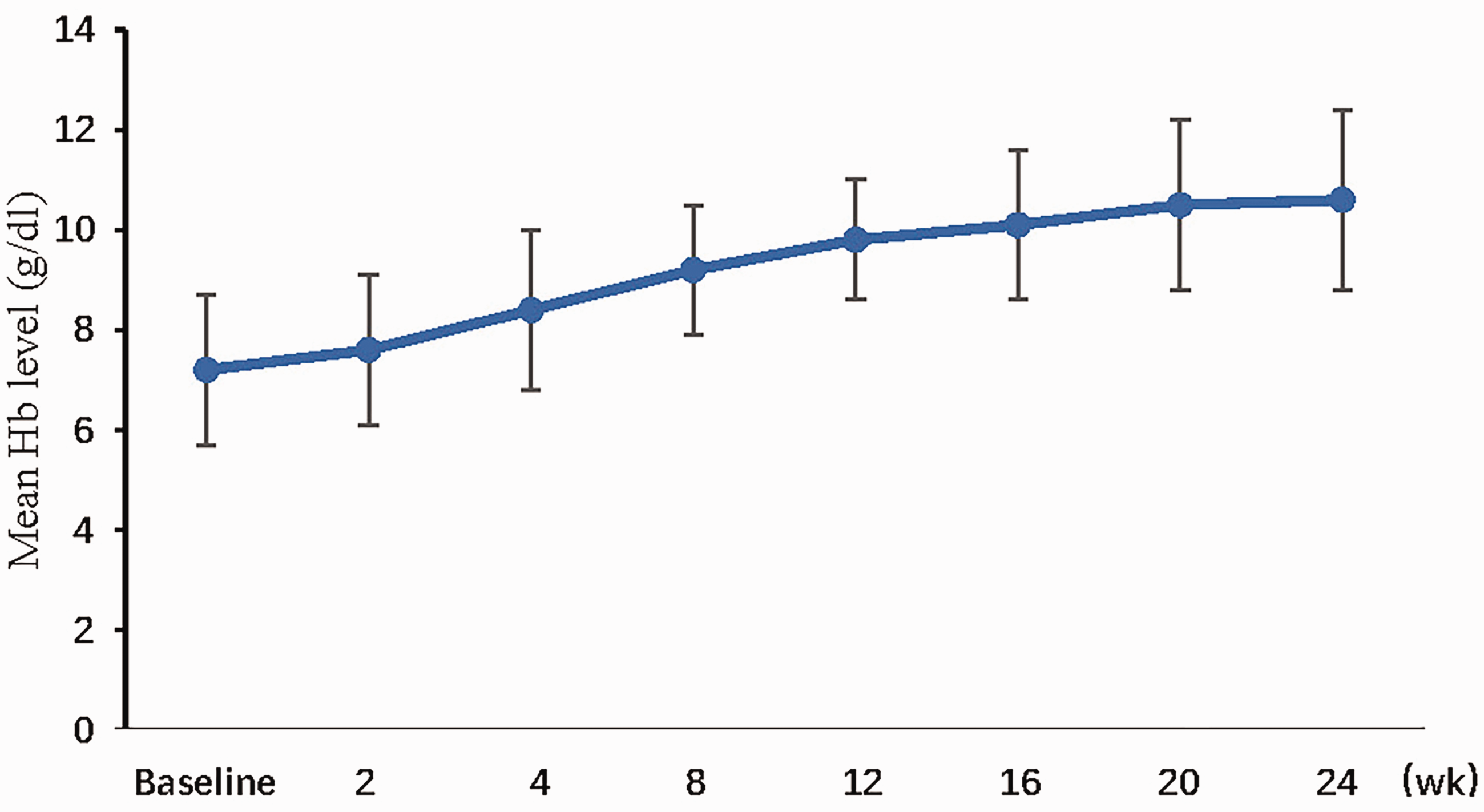
Mean hemoglobin (Hb) levels over time.
Changes in iron metabolism markers
Changes in iron metabolism are shown in Figure 2. At baseline, the mean hepcidin level was 270.2 ± 210.9 ng/mL. After 20 weeks of roxadustat treatment, the mean level was 201.3 ± 178.1, a decrease of 25.6% compared with the baseline level (
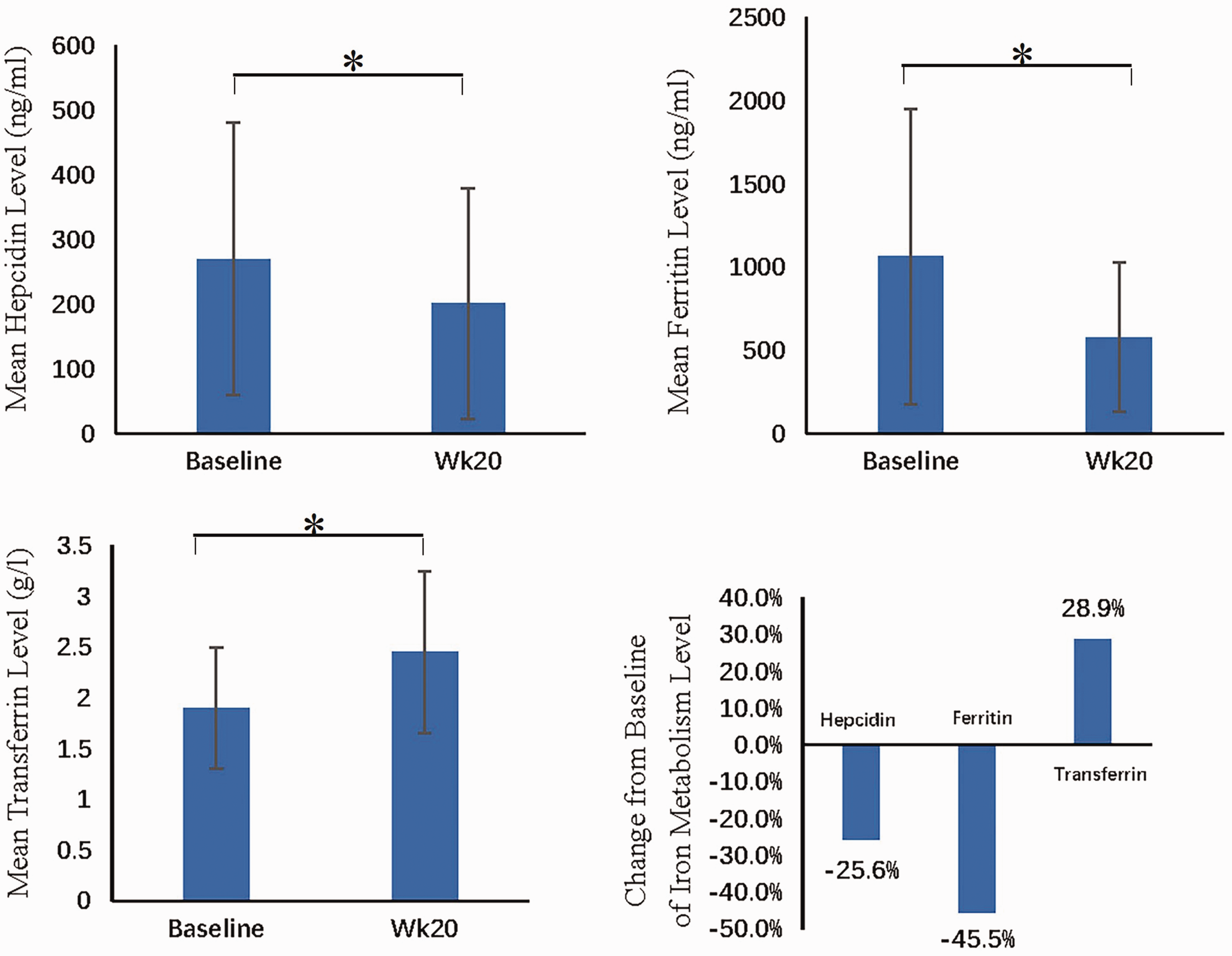
Mean levels and changes from baseline of iron metabolism markers at week 20.
Inflammation markers
At baseline, 50% of the patients had CRP levels above the upper limit of the normal range. The mean Hb level of the high CRP group at week 20 after roxadustat treatment was similar to that of the group with a normal CRP level (11.0 ± 2.0 g/dL vs 11.5 ± 1.9 g/dL) (Figure 3). The mean IL-6 level was decreased by 51% relative to baseline during the treatment period (12.4 ± 8.1 pg/mL vs. 7.2 ± 6.2 pg/mL,

Mean hemoglobin (Hb) levels at week 20 according to the C-reactive protein (CRP) subgroup (upper limit of the normal range (ULN) for CRP: 8 mg/L).
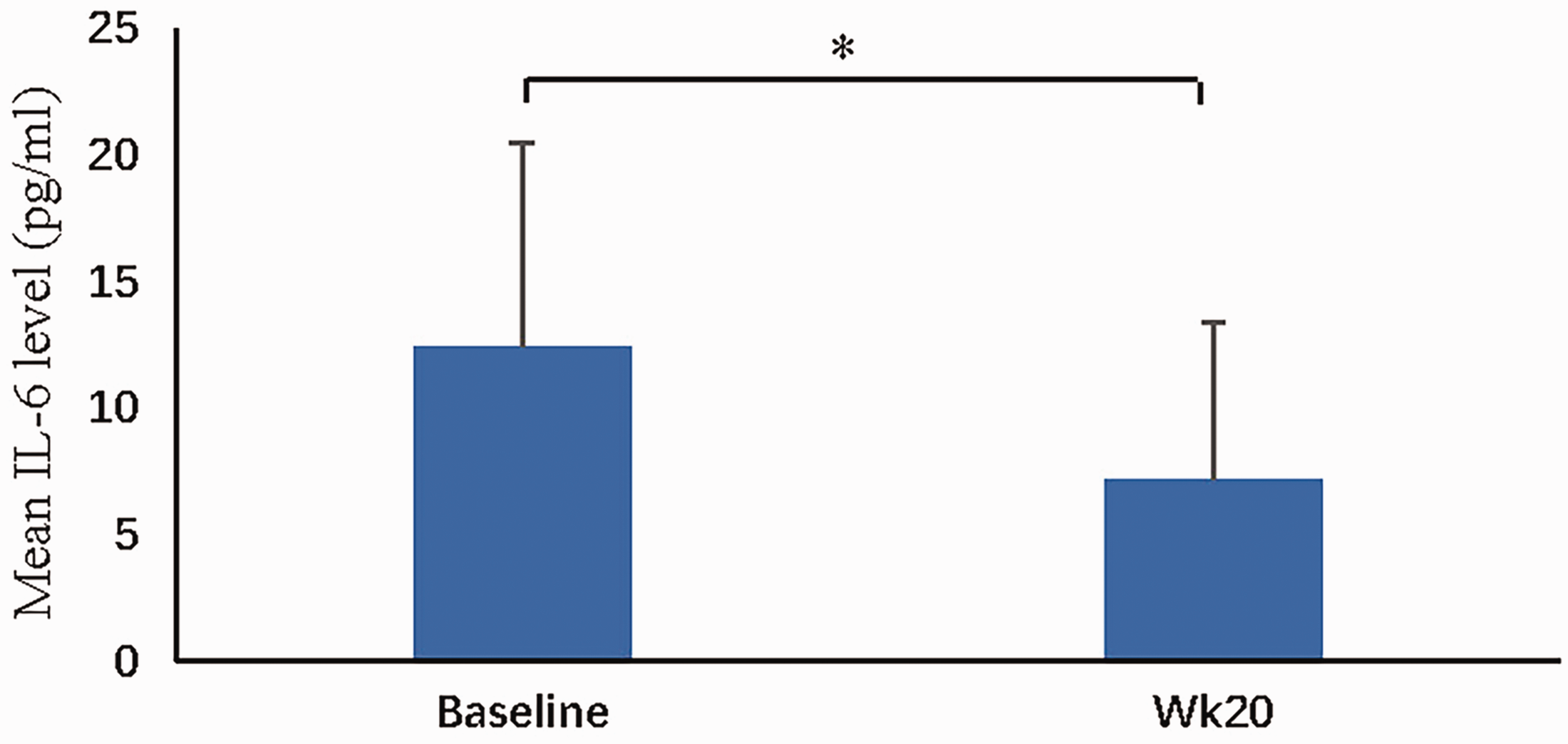
Change in mean interleukin (IL)-6 level after roxadustat treatment (
Safety aspects
All 30 patients completed the 24-week follow-up. The blood pressure levels in two patients were higher than the baseline levels. This symptom was transient and disappeared after adjusting the antihypertensive drug dosage. One patient reported arteriovenous fistula occlusion. There were no side effects involving the digestive system, nervous system, or liver function. No deaths occurred during the reporting period.
Discussion
At present, there are few reports on the treatment of ESA-hyporesponsive anemia with HIF-PHIs. In a subgroup analysis of the ESA-resistant population (baseline Hb, 9.0–10.0 g/dL) in a phase 3 roxadustat trial in Chinese HD patients, 16 after 26 weeks of treatment, 94.4% of patients had an increase in Hb of more than 1.0 g/dL, and 83.3% had an Hb level greater than 11 g/dL. In our study, after roxadustat treatment, 28 of 30 patients (93.3%) met the primary endpoint, with an Hb concentration increase of more than 1.0 g/dL from baseline, and 17 patients (56.7%) were within the target range (10.0–11.8 g/dL) at weeks 20 to 24. Roxadustat had similar effectiveness (93.3% vs. 94.4%) to that in a previous study, but the proportion of patients who reached an Hb level of 10 g/dL was much lower (56.7% vs. 83.3%), which may be because our study enrolled patients with much lower baseline Hb levels (7.2 ± 1.5 g/dL).
A clear definition of ESA-hyporesponsiveness has not yet been established, and guidelines have used different definitions to describe their findings. In 2012, the Kidney Disease: Improving Global Outcomes Clinical Practice Guidelines 17 defined ESA-hyporesponsiveness as not having an increase in Hb concentration from baseline after the first month of ESA treatment at appropriate weight-based dosing. Additionally, acquired ESA-hyporesponsiveness was defined as, after treatment with stable ESA doses, requiring two increases in ESA doses up to 50% beyond the dose at which patients had been stable to maintain a stable Hb concentration. In our study, all patients met the above criteria, and their rhEPO dosages were all greater than 12,000 IU/week before roxadustat treatment.
Functional iron deficiency may exist among patients with renal failure, which is characterized by the presence of adequate iron stores as defined by conventional criteria but with the inability to sufficiently mobilize this iron to support erythropoiesis with ESA administration. Because of the obstruction of iron transport from intestinal epithelial cells and macrophages to the blood, stored iron cannot be used effectively. Studies have shown that functional iron deficiency is often associated with inflammation, latent infection, malignancy, or liver disease. 18 In this study, most patients had elevated serum ferritin levels and low transferrin levels compared with those at baseline, indicating that these patients had functional iron deficiency.
Hepcidin is a small liver peptide, and many studies have shown that it is the master regulator of systemic iron homeostasis. As early as 2003, evidence from transgenic mouse models indicated that hepcidin is the predominant negative regulator of iron absorption in the small intestine, iron transport across the placenta, and iron release from macrophages.
19
Nicolas
MHD patients are in a chronic inflammatory state. In a study 21 of 754 HD patients, CRP and IL-6 were found to be strong and independent predictors of poor responsiveness to ESAs. In the present study, the effect of inflammation on anemia was also observed. Patients with elevated CRP levels did not exhibit an Hb hyporesponse with roxadustat treatment. After treatment, their IL-6 level decreased by 51%. Therefore, we speculate that roxadustat may improve the micro-inflammation state of MHD patients by regulating iron metabolism.
The limitations of this study include the small sample size, the short follow-up period, and its single-center, single-arm trial design without a placebo group. Therefore, some confounding factors may have affected the results.
In conclusion, oral roxadustat is effective for ESA-hyporesponsive anemia and may also improve iron metabolism and the micro-inflammatory state of MHD patients. Although the small sample size provided limited data, this study may provide important clinical information for future studies in subjects who are ESA-hyporesponsive. Further investigations of the relationship between roxadustat treatment and inflammation involving large-sample clinical trials are needed.
Footnotes
Acknowledgements
Author contributions
All authors have made substantial contributions to this work and have approved the final version of the manuscript. Concept and design: FY, HL. Acquisition of data: FY, XJC, LSZ, JS, WHY. Statistical analysis: FY, JS. Writing original draft: FY, WHY, JS. Writing, review, and editing: all authors.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
