Abstract
Objectives
Multiparametric flow cytometry (MFC) aids in the diagnosis and management of B-cell acute lymphoblastic leukemia (B-ALL) by establishing a baseline immunophenotype for leukemic cells and measuring minimal residual disease (MRD) throughout the course of treatment. Aberrant expression patterns of myeloid markers in B-ALL are also examined during long-term surveillance. Here, we investigated the utility of the newly described myeloid marker cluster of differentiation (CD)371 in MRD surveillance via MFC in patients with CD371-positive B-ALL.
Methods
Eight-color MFC with standard panels (including CD371) was used to evaluate 238 patients with newly diagnosed B-ALL. Expression levels of key markers were retrospectively assessed at diagnosis, as well as days 15 and 33 of therapy.
Results
CD371 was expressed in 8.4% of patients with B-ALL. CD371 positivity was associated with older age at diagnosis, higher expression levels of CD34 and CD38, and lower expression levels of CD10 and CD20. Residual leukemic cells demonstrated decreased CD10 expression and increased CD45 expression after therapy, whereas CD371 expression remained stable.
Conclusions
Patients with CD371-positive B-ALL exhibit a specific signature that merits further analysis, particularly because it has been associated with DUX4 rearrangement.
Keywords
Introduction
Acute lymphoblastic leukemia (ALL) constitutes a heterogeneous group of hematologic malignancies responsible for >80% of all acute leukemia cases; its cure rate is approaching 90%. 1 According to immunophenotype, ALL is classified as either B-cell lineage (B-ALL) or T-cell lineage (T-ALL); 85% of patients with ALL exhibit the B-ALL subtype.1–3 Although B-ALL can develop at any age, the incidence is highest between the ages of 2 and 5 years. 4 Genome-wide association studies have identified numerous genetic factors associated with an increased risk of B-ALL5,6; however, most patients have no recognized genetic factors. B-ALL is stratified into prognostically relevant genetic subtypes defined by chromosomal alterations, tumor-acquired DNA copy number variations, and leukemogenic sequence mutations. Chromosomal alterations include aneuploidy and chromosomal rearrangements that cause oncogene deregulation or the expression of chimeric fusion genes.3,7
Multiparametric flow cytometry (MFC) facilitates B-ALL diagnosis and management through immunophenotyping and minimal residual disease (MRD) analysis. Methodological advancements have led to increasing confidence in the clinical value of MRD analysis, such that this approach is preferred for assessing the treatment response and defining remission in patients with ALL.8,9 Several studies have identified a B-ALL subtype with a distinct immunophenotype and gene expression profile, characterized by deregulation of double homeobox 4 (DUX4) and the transcription factor E-26 transformation-specific-related gene (ERG).10–13 DUX4 deregulation is caused by enhancer element rearrangement, which leads to a C-terminally truncated DUX4 protein that is not normally expressed in B cells. This fusion commonly links the hypervariable region of the immunoglobulin heavy chain gene to the DUX4 gene in the subtelomeric region of chromosome 4. 14 Accurate identification of this subtype requires detection of DUX4 rearrangement (DUX4r).15,16 Recognition of the DUX4r subtype almost exclusively relies upon next-generation sequencing 13 ; however, this expensive approach is unsuitable for routine diagnosis. 14
Expression of the cell-surface antigen cluster of differentiation (CD)371, usually present on myeloid cells and easily detectable by flow cytometry, is a unique feature of DUX4r B-ALL. 17 The finding that this aberrant marker can be assessed by flow cytometry (rather than other complex and costly diagnostic techniques) has supported further investigation of DUX4r B-ALL.
Here, we analyzed the features of CD371 expression in patients with B-ALL from diagnosis until the end of induction therapy, then assessed the utility of this marker in MFC-based MRD monitoring of these patients. Furthermore, we investigated the immunophenotypic patterns of key antigens associated with this leukemia subtype, along with the impact of treatment on these patterns.
Materials and methods
This retrospective observational study included patients with newly diagnosed B-ALL who were assessed by the flow cytometry laboratory in King Hussein Cancer Center (KHCC), Jordan, from 1 October 2019 to 31 January 2022. Patient data were anonymized before analysis. Patients were diagnosed by morphology assessment and flow cytometry analysis using an eight-color BD FACSCanto™ II flow cytometer. Diagnostic phenotypes were determined using standard protocols. 18 At diagnosis, either bone marrow aspirates or peripheral blood samples were processed and analyzed. For MRD analysis, bone marrow samples were processed and assessed in accordance with our standard procedures.
At initial diagnosis, five combinations of eight monoclonal antibodies were used for immunophenotyping. MFC-based MRD monitoring (days 15 and 33) was performed with two combinations of eight monoclonal antibodies (Table 1); all leukemia-associated markers were monitored and at least 500,000 nucleated cells (events) were measured, allowing MRD detection with 0.01% sensitivity. 19 Pediatric patients underwent MRD monitoring on days 15 and 33 of induction therapy; adult patients underwent MRD monitoring solely on day 33. Some pediatric patients were referred from other clinics; thus, they did not undergo monitoring on day 15.
Flow cytometry markers used at diagnosis and during follow-up for patients with B-ALL.

Note: CD10, CD19, CD34, and CD45 are backbone markers in MRD analyses. Myeloid markers were also assessed during MRD analyses if patients initially exhibited positivity.
B-ALL, B-cell acute lymphoblastic leukemia; CD, cluster of differentiation; HLA-DR, human leukocyte antigen–DR isotype; MPO, myeloperoxidase; MRD, minimum residual disease; TdT, terminal deoxynucleotidyl transferase.
Patient characteristics were summarized by descriptive analysis. Categorical data (e.g., sex and marker presence/absence) are presented as counts and percentages. Continuous data (e.g., age and expression levels of key markers) are shown as medians and ranges.
Generally, categorical variables were compared using the χ2 test or Fisher’s exact test; continuous variables were compared using Student’s t-test or Mann–Whitney U test, depending on the assumptions required for each test. A significance threshold of P < 0.05 was used for all tests. Statistical analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA).
This retrospective study was observational and did not involve any contact with participants (i.e., it was low-risk). Therefore, the need for informed consent was waived by the Ethics Committee. The study protocol was approved by the Ethics Committee of KHCC (approval no. 21KHCC173F). The CARE checklist was followed when drafting this report. 20
Results
CD371 expression was assessed at diagnosis in 238 patients with B-ALL (median age, 7 years; range, 2 months to 83 years). Patient characteristics are summarized in Table 2. Among these patients, 20 (8.4%) were CD371-positive. Notably, CD371 positivity was associated with older age (P = 0.011, Table 2). At diagnosis, patients with CD371+ B-ALL displayed stronger expression of CD34 and CD38 (P < 0.001 and P = 0.026), compared with patients who had CD371-negative (CD371
Patient characteristics.

CD, cluster of differentiation.
Leukemia immunophenotype at diagnosis.

*Any of the following markers: CD4, CD11b, CD11c, CD13, CD15, CD33, and CD117.
**Pediatric patients were screened for all four rearrangements; adult patients were screened for t(9;22) only.
CD, cluster of differentiation.
There were no significant differences between the two groups in terms of aberrant myeloid marker expression. Additionally, MFC-based MRD monitoring revealed no significant differences in the response to induction therapy on days 15 and 33 (Table 4). However, a substantial decrease in CD10 expression was observed on days 15 and 33 among patients with CD371+ B-ALL who had positive MRD results (P < 0.001, Figure 1). At the end of induction therapy, 40% of patients had dim/partial CD10 expression, whereas 60% of patients did not show CD10 expression (Table 4).
Results of surveillance on day 15 and at end of induction therapy.

Note: Detection sensitivity threshold for MRD in acute lymphoblastic leukemia was 0.01%. 19
CD, cluster of differentiation; MFC, multiparametric flow cytometry; MRD, minimum residual disease.
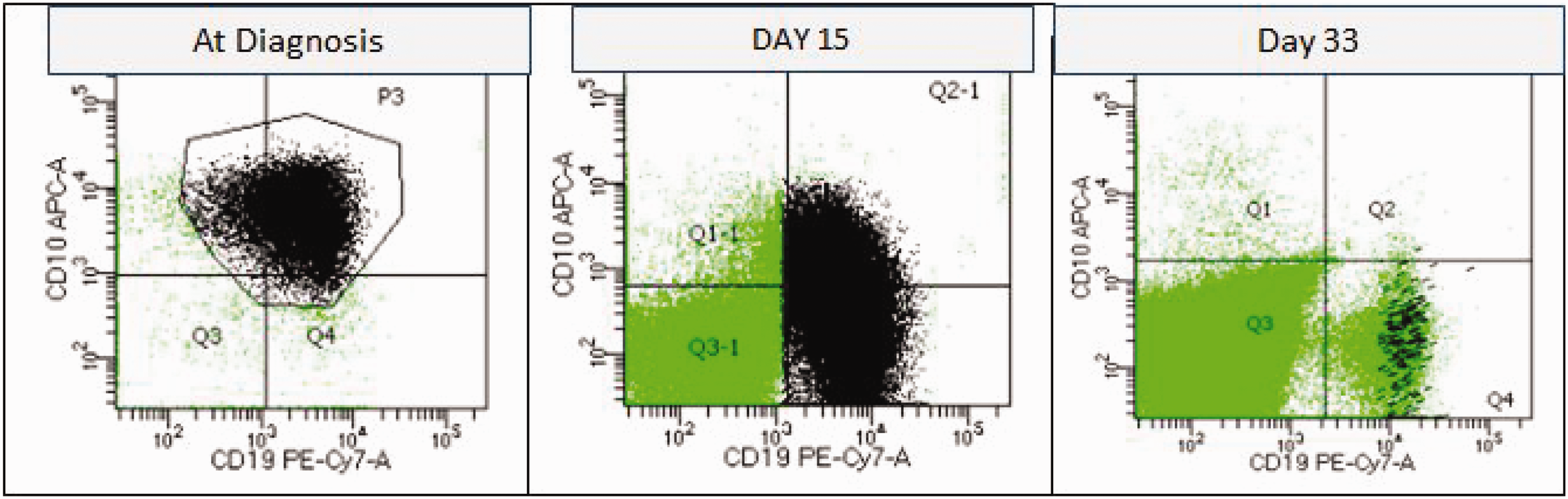
Patient with CD371+ B-ALL, with an obvious decrease in CD10 expression on days 15 and 33. APC, allophycocyanin; B-ALL, B-cell acute lymphoblastic leukemia; CD, cluster of differentiation; PE, phycoerythrin.
Increased CD45 expression was observed among most patients with CD371+ B-ALL on days 15 and 33, which shifted the blast population toward a monocytic phenotype (Figure 2). CD371 positivity persisted throughout the follow-up period among patients who initially displayed CD371 expression (Figure 3). The expression patterns of key antigens in all patients at baseline, day 15, and day 33 are illustrated in Figure 4.

B-lymphoblast shift toward a monocytic phenotype on day 15. CD, cluster of differentiation; SSC, side scatter.

Stability of CD371 expression after therapy. CD, cluster of differentiation; PE, phycoerythrin.

Expression patterns of key antigens among patients with CD371+ B-ALL at baseline, day 15, and day 33. B-ALL, B-cell acute lymphoblastic leukemia; CD, cluster of differentiation; MFI, mean fluorescence intensity.
Discussion
Over the past decade, there has been considerable progress in understanding the genomic features of B-ALL. New technologies have enabled detection of molecular changes, facilitating the identification of new B-ALL subtypes and biomarkers. A particularly useful finding is the aberrant expression of CD371 in DUX4r B-ALL.
In the present study, we used MFC analysis to examine the immunophenotypic features of patients with CD371+ B-ALL from initial diagnosis until the end of induction therapy (day 33). Consistent with the results of previous studies,13–15 we found that patients with CD371+ B-ALL comprised 8.4% of all patients with newly diagnosed B-ALL. The male predominance was slightly higher among patients with CD371+ B-ALL than among patients with CD371− B-ALL (1.86 vs. 1.27); patients with CD371+ B-ALL also exhibited an older median age at onset (13.5 years vs. 6 years).
At diagnosis, patients with CD371+ B-ALL showed stronger expression of CD34 and CD38, as well as weaker expression of CD10 and CD20, compared with patients who had CD371− B-ALL. We observed a substantial decrease in CD10 expression after induction therapy in patients with CD371+ B-ALL, such that 60% of patients showed complete loss of CD10 expression on day 33 (Figure 4). However, aberrant CD371 expression persisted in the blast population on days 15 and 33, which facilitated MRD detection via gating on the population of cells co-expressing CD19 and CD371 (Figure 3). This finding is important for the technical aspects of flow cytometry because it suggests an increased need to monitor all patients with CD19+/CD371+ B-ALL, regardless of CD10 positivity, to avoid missing MRD in these patients; we observed CD10 negativity in 33% of patients on day 15 and 60% of patients on day 33 (Table 4). Indeed, MFC-based MRD monitoring would have been technically challenging in these patients without the addition of CD371—its expression remained stable throughout the follow-up period, whereas other markers demonstrated variable expression (Figure 4). Furthermore, the presence of CD371 on the detected B-lymphoblasts aided in confidently distinguishing them from hematogones.
Although our MRD screening panels for patients with B-ALL lacked monocytic markers (e.g., CD14 and CD33), we observed a shift in the blast population toward a monocytic phenotype after induction therapy, consistent with the results of previous studies concerning the relationship between CD371 expression and early-switch ALL during corticosteroid treatment.14,21,22 Importantly, this “scatter” shift involves a change in scatter (i.e., higher CD45 mean fluorescence intensity and higher side scatter) rather than a change in lineage; a lineage shift requires confirmation using additional monocytic markers and validation with respect to genetic characteristics, which were not recorded in the present study. However, we emphasize the scatter shift from a technical perspective because facilitates gating of our target cell population. The assessment of B-ALL treatment response via MFC-based MRD monitoring is considered a powerful prognostic factor. 23 Some studies have demonstrated a favorable response to induction therapy in patients with MRD,15,24 whereas other studies have demonstrated an unfavorable response.14,21,22 In contrast, we did not find significant between-group differences in MRD results on days 15 and 33. Our results require validation in future studies that involve monitoring until the end of the therapy phase.
In conclusion, a greater difference in overall immunophenotypic profile between leukemic and normal cells facilitates the recognition of B-ALL cells and improves the sensitivity of MFC-based MRD monitoring. On the basis of this principle, and to enhance the recognition of leukemic cells, we strongly recommend routine inclusion of CD371 as a marker in the initial phenotype panel used for B-ALL diagnosis. If CD371 positivity is detected, this marker should be included in all subsequent MRD analyses. Furthermore, we recommend the use of distinct MFC-based MRD monitoring strategies for patients with CD371+ B-ALL, with emphases on blasts, the monocytic region, and corresponding markers, as well as CD19-positive events regardless of CD10 positivity.
An important limitation of this study was its lack of thorough genetic and molecular characterization mainly because of limited resources. However, we focused on immunophenotyping and the technical aspects of flow cytometry, which we considered the most feasible method.
In the future, because patients with CD371− B-ALL demonstrate considerable heterogeneity,1,5 it would be useful to compare the immunophenotypes of various antigens between patients with CD371+ B-ALL and patients with other distinct genetic subtypes of B-ALL.
Footnotes
Acknowledgments
The authors thank Ayat Taqash (KHCC Biostatistics and Clinical Research Department) for assistance with statistical analysis.
Author contributions
FB, AT, HS, and SK conceived and supervised the project. HS, SK, AA, and AK collected, analyzed, and interpreted multiparametric flow cytometry data. FB, AT, RA, and NH analyzed and interpreted clinical data. HS prepared tables and figures. FB and HS prepared the original draft and conducted statistical analysis. All authors contributed to manuscript writing, editing, and review. All authors read and agreed to the published version of the manuscript.
Data availability statement
All data used or generated in this study are included in the published article. Further inquiries can be directed to the corresponding author.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This study was supported by the Intramural Research Grant Program at King Hussein Cancer Center.
