Abstract
A rare but clinically important diagnostic dilemma arises when cases meet the criteria for both acute myeloid leukemia with myelodysplasia-related changes (AML-MRC) and mixed phenotype acute leukemia, especially those that evolve from myelodysplastic syndrome. We describe a 56-year-old male patient who presented with cytopenias and was initially diagnosed with myelodysplastic syndrome with single lineage dysplasia. Nearly 1 year later, this patient progressed to acute leukemia, and his blast cells simultaneously expressed T-lymphoid and myeloid antigens. Cytogenetic analysis showed a 20q deletion, and next-generation sequencing showed mutations of ASXL1, NRAS, PHF6, RUNX1, TP53, and PIGA. He was diagnosed with AML-MRC with blasts of the mixed T/myeloid phenotype according to the latest World Health Organization guidelines. In accordance with the treatment principles of AML-MRC, we chose an AML-like regimen for four cycles, but the patient did not achieve remission. Finally, we adhered to the treatment principles of mixed phenotype acute leukemia, and he achieved remission after a course of ALL-like regimen chemotherapy.
Keywords
Introduction
Mixed phenotype acute leukemia (MPAL) is a rare and aggressive leukemia, comprising 1.5% to 2.8% of newly diagnosed acute leukemias. 1 MPAL is defined as leukemia co-expressing certain antigens of more than one lineage of the same blast cells (biphenotypic leukemia) or that with separate populations of blasts of different lineages (bilineal leukemia). 2 However, a rare but clinically important diagnostic dilemma arises when cases evolve from myelodysplastic syndrome (MDS).
MDS is a group of heterogeneous hematological malignancies with a high risk of progression to acute myeloid leukemia (AML). AML with myelodysplasia-related changes (AML-MRC) represents a subgroup of AML defined by the presence of one or more of the following features: multilineage dysplasia (defined as the presence of ≥50% dysplastic cells in at least two cell lines), a history of MDS or MDS/myeloproliferative neoplasm (MPN), and specific MDS-defining cytogenetic abnormalities. 3 A small number of patients fit the diagnosis of both MPAL and AML-MRC, and questions remain as to which category such patients belong. The latest World Health Organization (WHO) guidelines classify such patients as having AML-MRC with a secondary notation of a mixed phenotype. 4 To date, only five AML-MRC cases with a mixed lineage have been reported.4–7 We report the first case of AML-MRC with the mixed T/myeloid phenotype in one population of blast cells, which evolved from MDS with single lineage dysplasia (MDS-SLD).
Case presentation
History of past illness
In January 2020, a 54-year-old man presented to an another hospital with repeated fever, cough and sputum. A computed tomography scan showed the presence of pneumonia. He had thrombocytopenia (40 × 109/L), and the white blood cell and hemoglobin counts were within the reference range. He went home after the pneumonia was cured, and did not undergo further examinations to clarify the etiology of the thrombocytopenia.
In February 2021, his complete blood cell count data showed cytopenias with a white blood cell count of 2.3 × 109/L, hemoglobin concentration of 95 g/L, and platelet count of 25 × 109/L. Bone marrow (BM) aspirate showed marked signs of dysplasia in granulocytes (10%), together with 3.5% myeloid blasts. Flow cytometric (FC) analysis of BM showed that myeloid blasts accounted for 4.16% (CD34+/CD117+/cMPO+/CD38+/CD13+/CD33+ and CD3−/CD5−/CD7−/CD8−/CD10−/CD19−). Karyotyping analysis showed 46, XY, del (20) (q11.2). A diagnosis of MDS-SLD with the revised international prognostic scoring system of the intermediate-risk group according to the 2016 WHO classification was made. He received only supportive care and refused hypomethylating agents for treatment.
In November 2021, he was hospitalized again for pneumonia, and a second BM analysis was performed. An increased number of blasts (21.5%) were observed. FC demonstrated one population of blasts (18.39%) with CD34+/HLA-DR+/CD117+/CD38+/cMPO+ (dim)/CD13+/CD7+/CD5+ (dim), part cCD3+ (dim), CD10+/TdT+/CD33+, and a lack of CD3/CD19/CD20.
A BM biopsy showed hypercellularity (90.0%) and an increased amount of immature granulocytes. Because cCD3 expression was low and immunohistochemical staining for cCD3 was negative, a diagnosis of MPAL was not established. Taken together, these results led to a diagnosis of AML-MRC with T cell antigen expression. Because of severe pulmonary infection, he was not able to have intensive induction chemotherapy. He received one cycle of decitabine (30 mg, intravenous infusion for 5 days) and one cycle of azacitidine (100 mg, subcutaneous infusion for 7 days) combined with venetoclax (100 mg on day 4, 200 mg on day 5, and 400 mg on days 6–28) treatment. He developed severe hematopoietic depression after chemotherapy, complicated by severe pneumonia and cerebral hemorrhage. Unfortunately, he did not achieve any remission. Another cycle of etoposide (100 mg, intravenous infusion for 5 days) combined with cytarabine (30 mg, subcutaneous infusion for 7 days) was then conducted. The patient still achieved no remission, and the immune phenotype of blast cells was not obviously reduced during this period.
History of the present illness
At 56 years old, the man had repeated fever with cough and sputum for 2 weeks, accompanied by dizziness, chest tightness, shortness of breath, and other symptoms. The man was then admitted to hospital. He was married and had one son. No additional family history was obtained.
Physical examination upon admission
A physical examination indicated skin and mucosal hemorrhage, and no hepatomegaly, splenomegaly, or lymphadenopathy.
Laboratory examinations
In April 2022, we performed another BM aspirate and observed that blast cells were increased to 40% with dysplastic features (Figure 1). FC analysis showed one population of aberrant blast cells (31.74%). These cells expressed CD34, CD117, HLA-DR, CD13, CD5, and CD7. Part of the cells expressed cMPO (57.41%), TdT (60.02%), cCD3 (50.32%), CD38 (58.62%), and CD10 (38.62%). All of the cells were negative for CD3, CD9, CD19, and cCD79a (Figure 2). Chromosomal karyotyping showed a 46, XY, del (20) (q11.2) karyotype. A reverse-transcriptase polymerase chain reaction test was performed to screen for 43 leukemia-causing chromosomal translocations. The patient tested negative for all 43 translocations. A panel of 72 genes that are frequently mutated in myeloid malignant neoplasms was examined using next-generation sequencing. The following mutations were found: ASXL1 (c.1927dupG:p.G642fs, variant allele frequency [VAF]: 78.33%), NRAS (c.G35A:p.G12D, VAF: 34.48%), PHF6 (c.C820T:p.R274X, VAF: 100%), RUNX1 (c.G497A:pR166Q, VAF: 30.14%), TP53 (c.G910T: p.E304X, VAF: 7.32%), and PIGA (c.C345A:p.Y115X, VAF: 8.62%).
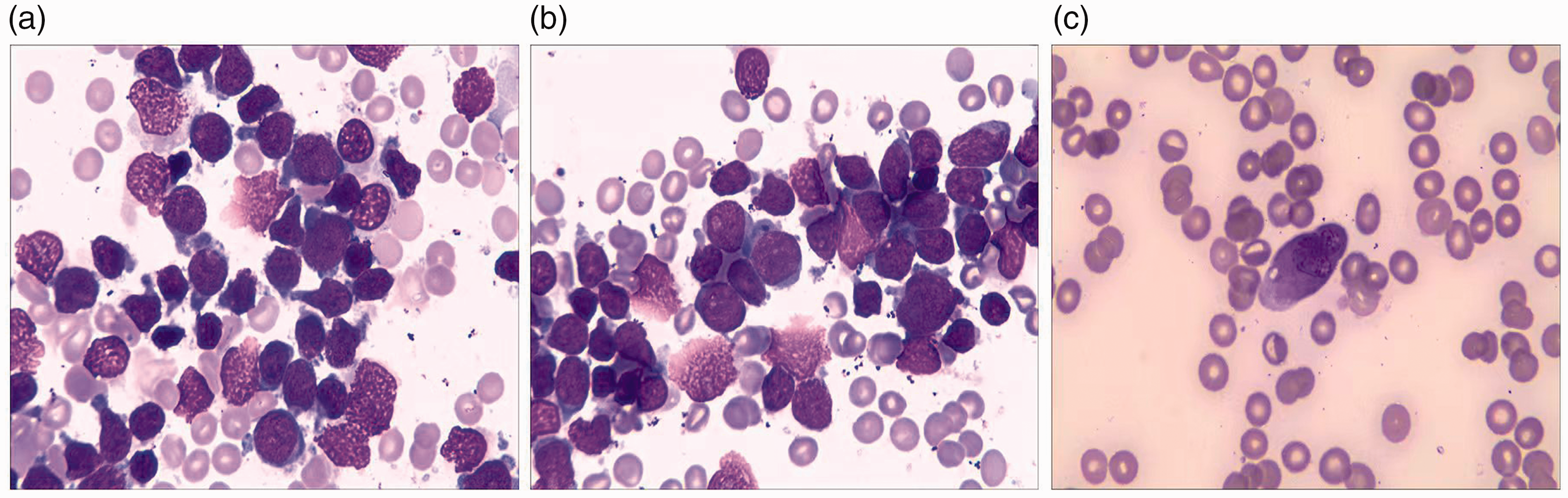
Acute myeloid leukemia with myelodysplasia-related changes with blasts of the mixed T/myeloid phenotype. Bone marrow aspirate shows one population of blasts with dysplastic features (a–c).

Flow cytometry shows a population of blasts with acute myeloid leukemia with myelodysplasia-related changes with T cell antigen expression of CD7 and low cCD3 in November 2021 (a–f). Flow cytometry shows a population of blasts with mixed T/myeloid lineage as shown by expression of CD34, HLA-DR, CD117, CD13, CD5, CD7, cMPO, and cCD3, and negativity for CD79a in April 2022 (g–m).
Imaging examinations
A computed tomography scan showed the presence of pneumonia, pleural effusion, and pericardial effusion.
Final diagnosis and treatment
On the basis of our findings, we established the diagnosis of AML-MRC with blasts of the mixed T/myeloid phenotype.
The Eastern Cooperative Oncology Group Performance Status was 3. We chose a hybrid regimen consisting of dexamethasone (7.5 mg, days 1–4) in combination with azacitidine (100 mg, days 1–7) and HAA (homoharringtonine 2 mg, days 1–5; aclacinomycin, 20 mg, days 1, 3, and 5; and cytarabine 50 mg, days 1–10). However, he failed to achieve remission again. Finally, an ALL-based therapy (vinorelbine 30 mg, days 1, 8, 15, and 22; doxorubicin hydrochloride liposome 20 mg, days 1–3; cyclophosphamide 1 g, days 1 and 15; and dexamethasone 7.5 mg on days 1–15, and 5 mg on days 16–28) was applied intravenously for one cycle. After this time, complete remission with an incomplete blood count recovery was achieved with 3% residual abnormal blasts.
Outcome and follow-up
The patient achieved complete remission with an incomplete recovery of the blood count and is still alive. He is ready to undergo an allogeneic transplant. The reporting of this study conforms to the CARE guidelines. 8
Discussion
AML-MRC accounts for 24% to 48% of all AML cases and is associated with worse outcomes than AML not otherwise specified.9,10 MPAL represents an uncommon, but heterogenous, disease and is associated with an inferior outcome relative to AML and ALL cohorts. 1 Some cases may have the overlapping features of AML-MRC and MPAL, which lead to difficulties in classification. To date, there is no independent WHO diagnostic category for such patients with both AML-MRC and MPAL features. Although the WHO classification suggests classifying this entity as AML-MRC with blasts of a mixed phenotype, whether it can accurately reflect the characteristics of such patients is questionable.
One issue is whether gene mutation profiles in such patients are similar to AML-MRC or similar to MPAL. As previously reported,
Another issue is whether the current diagnostic classification facilitates patients’ treatment options. The treatment options for patients with AML-MRC are still similar to those for patients with non-AML-MRC, including the 3 + 7 regimens, hypomethylating agents, venetoclax, liposomal daunorubicin–cytarabine (CPX-351), and allogenic hematopoietic stem cell transplantation, namely AML-like regimens. 3 However, most of the current reports have shown that patients with MPAL benefit from beginning with ALL-like therapies.1,13,14 Therefore, if such patients are classified as a subtype of AML-MRC, the AML-like regimen is the first choice. However, if they are classified as a subtype of MPAL, an ALL-like regimen is preferred. Unfortunately, in our patient and a patient reported by Kim et al., 7 AML-like regimens were chosen, and both patients failed to achieve remission. However, our patient achieved an incomplete blood count recovery after receiving an ALL-like regimen. Therefore, such patients being classified as MPAL with MRC appears to be more appropriate, and ALL-like regimens are preferred.
Currently, a diagnosis of AML-MRC can be established on the basis of cytogenetic abnormalities, such as complex karyotypes. The frequency of complex karyotypes in MPAL was reported to be between 24% and 32%.14,15 Therefore, AML-MRC with mixed lineage blasts may occur more frequently than previously reported. Further studies are required to clarify the clinical and genetic characteristics of such patients to facilitate more accurate diagnostic classification systems while helping to guide treatment options.
Conclusion
We describe a patient with MDS-SLD who progressed to acute leukemia with blasts of the mixed T/myeloid phenotype. The current WHO classification suggests classifying this entity as AML-MRC with blasts of a mixed phenotype. Further studies are required to summarize the characteristics of such patients, and perhaps classifying them as MPAL with MRC will be applied in the near future.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221122741 - Supplemental material for Acute myeloid leukemia with myelodysplasia-related changes and blasts of the mixed T/myeloid phenotype: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605221122741 for Acute myeloid leukemia with myelodysplasia-related changes and blasts of the mixed T/myeloid phenotype: a case report by Xian-Fu Sheng, Li-Li Hong, Fei-Yan Huang and Hai-Feng Zhuang in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Data availability statement
The data used to support the findings of this study are available from the corresponding author upon request.
Ethics statement
This study did not require approval by an ethics committee or institutional review board because the study did not involve human or drug experiments, and did it involve the use of human biological specimens. Written informed consent was obtained from the patient for treatment and publication of this case report.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Zhejiang Province (No. LY19H290003); the Zhejiang Provincial Medical and Health Science and Technology Project (No. 2020KY196); and the Foundation of Zhejiang Province Chinese Medicine Science and Technology Planes (No. 2020ZA04).
Author contributions
Zhuang HF conceived and designed the study; Sheng XF and Hong Lili wrote the manuscript; Huang FY provided the images and material support.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
